Abstract
Background: Overlap between diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS) is associated with significantly higher mortality than either condition alone. While hypertonic (trans locational) hyponatremia is common in hyperglycemic crises, clinically significant hypernatremia in mixed DKA/HHS presents with certain treatment challenges due to rapid osmotic shifts during insulin therapy.
Case presentations: A 40-year-old woman with non-significant past medical history presented with vomiting, polyuria, polydipsia, and confusion for two days. Laboratory evaluation revealed serum glucose 1280 mg/dL, arterial pH 7.214, bicarbonate 13.1 mmol/L, anion gap 24, β-hydroxybutyrate 6.0 mmol/L, corrected sodium 152 mmol/L (measured sodium 133 mmol/L), and calculated effective osmolality 337 mOsm/kg. Initial treatment with isotonic saline and intravenous insulin led to improvement in hyperglycemia but worsening corrected hypernatremia to 160 mmol/L after 3 liters of normal saline. Free water deficit was calculated at 6.3 L. Fluid therapy was transitioned to hypotonic solutions with titration to limit sodium correction to ≤10 mmol/L per 24 hours. Sodium levels gradually normalized with resolution of ketoacidosis, and improvement in mental status without significant neurologic complications.
Conclusion: In mixed DKA/HHS, early assessment of corrected sodium and effective osmolality is critical. Insulin-mediated intracellular water shifts may unmask or worsen hypernatremia. Prompt reassessment of corrected sodium and individualized fluid management is needed to avoid treatment-related exacerbation of hypertonicity.
Keywords
Diabetic ketoacidosis, Hyperosmolar hyperglycemic state, Hypernatremia
Introduction
Diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS) represent two ends of the hyperglycemic crisis spectrum. Combined DKA/HHS presentations occur in up to 30% of cases and are associated with nearly double the mortality compared with isolated syndromes [1,2]. Despite this significant clinical burden, current management algorithms largely extrapolate from isolated DKA or HHS data and provide only limited guidance for overlap presentations. In hyperglycemic crises, hyponatremia is typically hypertonic (trans locational) hyponatremia, reflecting osmotic water movement from intracellular to extracellular space due to elevated glucose [3]. Corrected sodium must therefore be calculated to accurately assess tonicity. While measured hypernatremia (>145 mmol/L) is seen only in a minority of cases, corrected hypernatremia is predominant in HHS and reflects excessive free water deficit [4], Importantly, insulin therapy reverses glucose-driven extracellular dilution, causing water to shift intracellularly and potentially increasing serum sodium concentration. As a result, hypernatremia may worsen during treatment even when guideline-based therapy is followed appropriately.
We present a case of mixed DKA/HHS in which corrected hypernatremia significantly worsened during initial isotonic fluid resuscitation and insulin therapy, requiring early shift away from the standardized protocols and individualized fluid management based on sodium levels. This case challenges the prevailing practice of routine initial isotonic fluid administration in mixed presentations and calls for a need for further research on evaluating the role of hypotonic fluid therapy as an alternative early management strategy in selected patients.
Gap
There is limited literature addressing optimal fluid strategies in DKA/HHS overlap with hypernatremia.
Objective
We present a case of mixed DKA/HHS with significant hypernatremia that worsened during standard therapy, highlighting the need for individualized fluid management.
Significance
This case underscores the importance of early recognition of corrected sodium and reconsideration of standard isotonic fluid protocols in select patients.
Case Description
A 40-year-old female with no prior medical history and no known diagnosis of diabetes presented to the emergency room (ER) with complaints of generalized weakness, nausea, vomiting, increased thirst, and urinary frequency lasting two days. Over the past few hours, she also had become drowsy. No other complaints were reported. Family history was significant for type 2 diabetes in the father.
Her vital signs in the ER had shown a temperature of 37.5°C, pulse rate of 110/min, respiratory rate of 22/min, blood pressure of 126/81mmHg, and oxygen saturation of 98%. Physical exam revealed an ill-appearing female with generalized weakness, lethargy, and altered mental status without focal neurologic deficit or abdominal tenderness. Point-of-care glucose levels obtained in the ER were found to be elevated. Initial chemistry panel was revealed glucose level of 1280 mg/dL (N: 70–140 mg/dL), potassium of 5.8 mmol/L (N: 3.6–5.2 mmol/L), calcium of 10.5 mg/dL (corrected calcium was 9.7 mg/dL for an albumin concentration of 5 g/dL; N: 8.6-10.5 mg/dL), lactate of 3.3 mmol/L (N: 0.5-2.2 mmol/L), and widened anion gap of 24 (N: 7-15). Arterial blood gas analysis demonstrated pH 7.214, pCO2 33.8 mm Hg, paO2 95.8 mm Hg, and bicarbonate 13.1 mmol/L. Of note, her serum sodium level of 133 mmol/L, roughly translated to a corrected sodium level of 152 mmol/L (N: 135–145 mmol/L). Corrected Na (mmol/L) for hyperglycemia was calculated as: measured Na + 1.6 x [(Glucose – 100) / 100]. Corrected Na for this patient was (133 + 1.6 x [(1280 – 100) / 100]) = 152 mmol/L. Additional testing had revealed markedly elevated β-hydroxybutyrate levels of 6.0 mmol/L (N: <0.4 mmol/L), and a high serum osmolality of 337 mOsm/kg (N: 275–295 mOsm/Kg). Effective osmolality was calculated as: [2 x measured Na (mmol/L)] + [glucose mg/dL)/18]. Blood urea nitrogen (BUN) is excluded as it is an ineffective osmole that crosses cell membranes freely and does not contribute significantly to transcellular water shifts in HHS. The patient was thus admitted with an overlap diagnosis of hyperosmolar hyperglycemic state and diabetic ketoacidosis (DKA/HHS) with hypernatremia, complicated by metabolic encephalopathy.
Treatment was promptly initiated per DKA/HHS protocols, beginning with aggressive IV hydration with 0.9% normal saline (NS) and an insulin drip. Blood glucose levels were monitored every 1–2 Hr, and serum chemistry profiles were monitored every four hours. A few hours into the treatment, there was no appreciable change in her clinical status, and her corrected sodium level rose to 160 mmol/L. By that time, a total of 3 liters of NS had already been infused. To address her worsening hypernatremia and total calculated free water deficit of 6.26 L, subsequent fluid hydration was calculated to ensure slow correction of hypernatremia in 24 Hrs. Free water deficit was calculated as:(% total body water) x [(current Na/ideal Na) - 1], and percent total body water for an adult female as 0.5 x weight (kg). The free water deficit for this patient was 6.26 L (0.5 x 86.7 kg x [(160/140)-1]). She was made NPO (nothing by mouth) and transferred to the Progressive Care Unit (PCU) for close monitoring. She was transitioned to hypotonic fluids with 0.45% NaCl in sterile water until her glucose levels reached less than 250. Then fluids were switched to 5% dextrose & 0.45% NaCl in water per protocol. The goals of management were to treat DKA/HHS with insulin infusion while simultaneously calculating free water deficit to titrate fluids so that serum sodium was reduced by no more than 8–12 mmol/L in 24 Hr to prevent cerebral edema.
After 24 hours of treatment, her condition began to stabilize. Blood glucose levels had improved significantly to 235 mg/dL, bicarbonate to 24 mmol/L, serum osmolality decreased to 314 mOsm/kg, and β-hydroxybutyrate to 0.4 mmol/L. She was then transitioned off the DKA/HHS protocol insulin drip to a lower-dose insulin infusion. Her sodium levels slowly corrected to 152 mmol/L with subsequent improvement in mentation. She was then transitioned to 5% dextrose in water (D5W) to target further reduction in hypernatremia, followed by initiation of liberal oral fluid intake.
By 48 hours, her clinical condition had improved further with return to baseline mentation. Her blood glucose further decreased to 179 mg/dL, osmolality to 302 mOsm/kg, and bicarbonate stabilized at 22 mmol/L. Subcutaneous insulin therapy was introduced with a regimen of 15 units of insulin glargine (long-acting insulin) twice daily and 10 units of insulin as part (short acting insulin) three times daily with meals. Her corrected sodium levels had reduced to 146 mmol/L - meeting the established goals of treatment. At 60 Hr, the corrected sodium levels had normalized to 143 mmol/L. She continued to improve clinically over the next day, with minimal further change in serum sodium levels and optimal glucose level control. Insulin glargine dose was increased to 20 units twice daily. An overview of the timeline correlating with blood glucose and sodium levels has been shown in Figure 1.
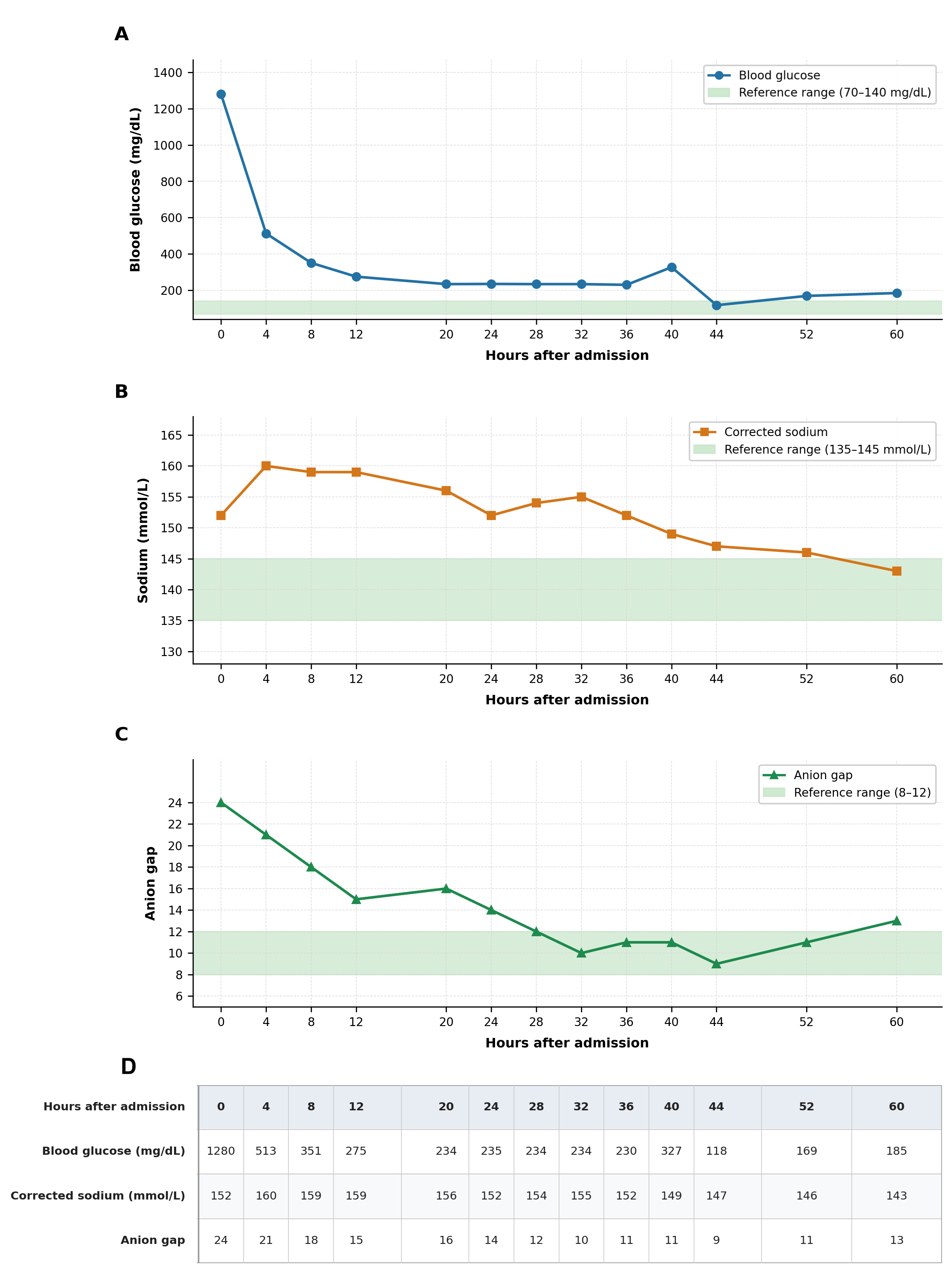
Figure 1. Serial metabolic trends in mixed DKA/HHS complicated by hypernatremia. Temporal trends in blood glucose (A), corrected sodium (B), and anion gap (C) over first 60 hours following hospital admission. Green shaded bands denote reference ranges for each parameter (blood glucose 70–140 mg/dL; sodium 135–145 mmol/L; anion gap 8–12). Initial severe hyperglycemia and high anion gap metabolic acidosis improved with insulin therapy, while corrected sodium transiently increased (peaking at 160 mmol/L) after isotonic fluid resuscitation before gradually normalizing following transition to hypotonic fluids. Raw values underlying all three panels are presented in the data table (D).
At Ninety-six hours, her lab results were within normal range, except for elevated blood glucose levels of 236 mg/dL. She was educated on diabetes and strategies for the prevention of similar episodes in the future. Before discharge, she was transitioned to a long-term insulin regimen consisting of 50 units of Tresiba (insulin degludec) nightly and 15 units of Novolog (insulin as part) three times daily with meals, along with a correctional scale for additional glucose control. At a follow-up visit one week later, the patient was doing well and reported no concerns. Her pre-meal glucose levels ranged between 160–180 mg/dL, which were slightly elevated but not concerning. A 30-day follow-up visit was arranged, along with referrals to a primary care physician and an endocrinologist for continued diabetes management. A diagnosis of type 2 diabetes was established upon follow-up visits.
Discussion
Here we presented a case of a middle-aged female with no prior diagnosis of diabetes who had nausea, vomiting, and altered mental status on presentation. Further investigations revealed a mixed diagnosis of HHS and DKA. Notably, this was accompanied by overt hypernatremia in the absence of correction–an unusual finding. Interestingly, the patient lacked any identifiable risk factors predisposing to hypernatremia. This was unlike most other previously reported cases, which were seen in the pediatric population and associated with certain predisposing factors such as excessive consumption of carbohydrate-rich soft drinks or herbal supplements [5,6]. The initial management of our patient was done with isotonic fluids as per the DKA and HHS guidelines; however, her mental status failed to improve, along with worsening of measured hypernatremia. Subsequently, hypotonic D5W was administered to correct the sodium imbalance with total volume calculated based on free water deficit. This case was complex from the outset, marked by an atypical diagnosis along with worsening of metabolic derangements following the guideline-based treatment. HHS and, less often, DKA are associated with trans locational hyponatremia, which occurs due to transcellular shift of water from the intracellular space into the extracellular space secondary to high serum levels of osmotically active glucose. Calculating sodium levels with glucose correction provides a more accurate assessment. However, hypernatremia as a presenting feature in DKA/HHS is exceptionally rare. Multiple mechanisms have been proposed for this, all of which share a common underlying general principle: an exacerbated free water loss that exceeds electrolyte losses through polyuria, vomiting, or diarrhea [7]. Moreover, in the setting of severe hyperglycemia, the renal absorption threshold for glucose is surpassed, leading to glucosuria, which in turn causes osmotic diuresis and dramatic water loss. The continuation of this over a prolonged period resulted in hypernatremia and an increased serum osmolarity, additive to hyperosmolar state from hyperglycemia [8].
Hypernatremia in HHS and DKA is associated with numerous complications, some of which can be life-threatening. Mortality rates for severe hypernatremia (serum sodium >150 mmol/L) range from 50% during hospitalization to approximately 90% in the long term [8]. A well-known complication of severe hypernatremia is osmotic demyelination syndrome, characterized by intracellular fluid depletion in vulnerable pontine neurons, leading to cell death. At extremely high serum sodium levels (>200 mmol/L), rare but devastating complications such as rhabdomyolysis and acute caudate infarcts have been reported [9]. Additionally, rapid correction of plasma osmolality and hypernatremia poses a risk of cerebral edema, though this has been predominantly observed in pediatric DKA cases and only rarely in adults [10]. Although our patient did not develop any of these complications, she did have persistent confusion even after the correction of hyperglycemia and ketoacidosis. Nevertheless, careful management of hypernatremia in the setting of overlapping DKA and HHS remains critical requiring special attention to corrected serum sodium levels and free water deficit, to prevent such outcomes [4]. Most cases of hypernatremia in the setting of overlapping DKA and HHS reported so far have been managed with initial normotonic fluids to correct fluid deficit, followed by a transition to hypotonic fluids to address hypernatremia. For instance, one case study described the management of a 50-year-old woman with DKA/HHS and an initial corrected sodium of 158 mmol/L, using an initial infusion of isotonic saline at 15-20 mL/kg/Hr for the first hour, followed by hypotonic half-normal saline [11]. Similarly, a 50-year-old woman with severe hypernatremia of 169 mmol/L upon presentation was treated with isotonic fluids initially, followed by D5W for gradual sodium correction [9]. However, a major concern with this approach is the potential for unexpected worsening of hypernatremia, deterioration in mental status, and hemodynamic instability unresponsive to cardiopulmonary resuscitation, particularly when serum sodium exceeds 180 mmol/L. This was notably seen in a case involving a 17-year-old male with severe DKA-HHS following excessive consumption of high-fat milk and carbonated beverages [12]. Another area of debate is free water replacement via the enteral route, which was also done in our case during the third day of treatment. However, a recent retrospective study from a critical care setting reported limited effectiveness of this approach for hypernatremia correction [13]. In our case, the patient's corrected sodium levels rose from 152 to 160 after treatment of hyperglycemia. Given lack of clinical improvement after 3L of NS administration, shifting to hypotonic fluids later led to the resolution of hypernatremia. This is explained physiologically as the initial severe hyperglycemia causes water to shift from the intracellular space to the extracellular space, diluting serum sodium. Electrolyte derangements are further compounded by osmotic diuresis driven by glucosuria. As hyperglycemia is treated with insulin, water shifts back into cells reversing the dilutional effect and increasing the serum sodium concentration. The goal is to facilitate a gradual decline in sodium and glucose to avoid rapid changes in plasma osmolality and mitigate risk of cerebral edema, with studies recommending sodium reduction by no more than 0.5 mEq/L/Hr and glucose reduction at a rate of 75–100 mg/dL/Hr [14]. Thus, rather than rigidly adhering to existing protocols, there is a need to tailor management based on individual patient presentations. Most cases of hypernatremia in the setting of overlapping DKA and HHS reported so far have been managed with initial normotonic fluids to correct fluid deficit, followed by a transition to hypotonic fluids to address hypernatremia. For instance, one case study described the management of a 50-year-old woman with DKA/HHS and an initial corrected sodium of 158 mmol/L, using an initial infusion of isotonic saline at 15–20 mL/kg/Hr for the first hour, followed by hypotonic half-normal saline [11]. Similarly, a 50-year-old woman with severe hypernatremia of 169 mmol/L upon presentation was treated with isotonic fluids initially, followed by D5W for gradual sodium correction [9]. However, a major concern with this approach is the potential for unexpected worsening of hypernatremia, deterioration in mental status, and hemodynamic instability unresponsive to cardiopulmonary resuscitation, particularly when serum sodium exceeds 180 mmol/L. This was notably seen in a case involving a 17-year-old male with severe DKA-HHS following excessive consumption of high-fat milk and carbonated beverages [12]. Another area of debate is free water replacement via the enteral route, which was also done in our case during the third day of treatment. However, a recent retrospective study from a critical care setting reported limited effectiveness of this approach for hypernatremia correction [13]. In our case, the patient's corrected sodium levels rose from 152 to 160 after treatment of hyperglycemia. Given lack of clinical improvement after 3L of NS administration, shifting to hypotonic fluids later led to the resolution of hypernatremia. This is explained physiologically as the initial severe hyperglycemia causes water to shift from the intracellular space to the extracellular space, diluting serum sodium. Electrolyte derangements are further compounded by osmotic diuresis driven by glucosuria. As hyperglycemia is treated with insulin, water shifts back into cells reversing the dilutional effect and increasing the serum sodium concentration. The goal is to facilitate a gradual decline in sodium and glucose to avoid rapid changes in plasma osmolality and mitigate risk of cerebral edema, with studies recommending sodium reduction by no more than 0.5 mEq/L/Hr and glucose reduction at a rate of 75–100 mg/dL/hr [14]. Thus, rather than rigidly adhering to existing protocols, there is a need to tailor management based on individual patient presentations.
Conclusion
Mixed HHS and DKA complicated by hypernatremia are an uncommon presentation in adults without a prior diagnosis of diabetes. The presence of hypernatremia upon presentation to the ED has been associated with significantly increased mortality rates. Therefore, early and appropriate management of hypernatremia and plasma osmolality is as crucial as addressing DKA and HHS to prevent poor clinical outcomes. Current management strategies typically involve initial fluid resuscitation with isotonic saline, followed by a transition to hypotonic solutions. Transitioning to hypotonic fluids earlier may offer greater benefit and help avoid exacerbation of hypernatremia and prevent rapid shifts in osmolality. Ultimately, clinical judgment should remain essential, and fluid management should be individualized based on each patient's physiological status. In the future, as this overlap of DKA/HHS is further studied, guidelines should be updated to add DKA/HHS overlap protocols to improve patient outcomes and decrease length of hospital stays. While our case report may provide directions for physicians facing similar challenges with the clinical management of hypernatremia in DKA/HHS overlap, there were certain limitations. For one, serum osmolality measurements were infrequent thereby limiting accurate insights into treatment response. Furthermore, this is a single-patient case report and thus findings may not necessarily be applied to similar future cases. More evidence is indeed needed for the findings to be established into management protocols.
References
2. Pasquel FJ, Tsegka K, Wang H, Cardona S, Galindo RJ, Fayfman M, et al. Clinical Outcomes in Patients With Isolated or Combined Diabetic Ketoacidosis and Hyperosmolar Hyperglycemic State: A Retrospective, Hospital-Based Cohort Study. Diabetes Care. 2020;43(2):349–57.
3. Dhatariya K, Mustafa O, Stathi D. Hyperglycemic Crises. In: Feingold KR, Adler RA, Ahmed SF, et al, eds. Endotext. MDText.com, Inc.; 2000. http://www.ncbi.nlm.nih.gov/books/NBK279052/. Accessed Feb 14, 2026.
4. Cao S, Cao S. Hyperglycemic Hypernatremic Hypertonic State: A Predominant HHS Subtype and Its Clinical and Diagnostic Features. J Clin Endocrinol Metab. 2026 Jan 21;111(2): e406–13.
5. Kim HJ, Kim DH, Jun YH, Lee JE. A rare diabetes ketoacidosis in combined severe hyponatremic hyperosmolarity in a new-onset Asian adolescent with type I diabetes. BMJ Case Rep. 2014;2014: bcr2014208016.
6. Choo SJ, Lee HG, Kim CJ, Yang EM. Severe hypernatremia in soft drink ketoacidosis and hyperglycemic hyperosmolar state at the onset of type 2 diabetes mellitus: a case series of three adolescents. Clin Pediatr Endocrinol. 2022;31(2):81–6.
7. Ibarra G, Majmundar MM, Pacheco E, Zala H, Chaudhari S. Hypernatremia in Diabetic Ketoacidosis: Rare Presentation and a Cautionary Tale. Cureus. 2020 Dec 2;12(12):e11841.
8. Wagner B, Ing TS, Roumelioti ME, Sam R, Argyropoulos CP, Lew SQ, et al. Hypernatremia in Hyperglycemia: Clinical Features and Relationship to Fractional Changes in Body Water and Monovalent Cations during Its Development. J Clin Med. 2024 Mar 28;13(7):1957.
9. Sohal RJ, Joshi S. Salty, Sweet and Difficult to Treat: A Case of Profound Hypernatremia in the Setting of Hyperosmotic Hyperglycemic State. Cureus. 2020;12(3): e7278.
10. Glaser NS, Wootton-Gorges SL, Marcin JP, Buonocore MH, Dicarlo J, Neely EK, et al. Mechanism of cerebral edema in children with diabetic ketoacidosis. J Pediatr. 2004 Aug;145(2):164–71.
11. Kumar V, Nanavati SM, Melki G, Upadhyaya M, Dhillon R, Gibiezaite S, et al. A Case of Diabetic Ketoacidosis Presenting with Hypernatremia, Hyperosmolarity, and Altered Sensorium. Case Rep Endocrinol. 2018 Jul 16; 2018:4806598.
12. Choo SJ, Lee HG, Kim CJ, Yang EM. Severe hypernatremia in soft drink ketoacidosis and hyperglycemic hyperosmolar state at the onset of type 2 diabetes mellitus: a case series of three adolescents. Clin Pediatr Endocrinol. 2022;31(2):81–6.
13. de Vos EAJ, van der Voort PHJ. ICU acquired hypernatremia treated by enteral free water – A retrospective cohort study. J Crit Care. 2021; 62:72–5.
14. Palmer BF, Clegg DJ. Electrolyte and Acid-Base Disturbances in Patients with Diabetes Mellitus. N Engl J Med. 2015;373(6):548–59.
