Abstract
Insulinoma is a rare and potentially life-threatening tumor if not treated promptly. The time from initial presentation to diagnosis may be prolonged in patients who have impaired awareness of hypoglycemia. A 34-year-old female with daytime hypoglycemia awareness and nocturnal hypoglycemia unawareness was initially treated for reactive hypoglycemia until continuous glucose monitoring (CGM) data revealed nocturnal hypoglycemia. Outpatient CGM was used to help establish the diagnosis of insulinoma and to help expedite treatment. Given the potential for hypoglycemia unawareness in patients with life-threatening causes of hypoglycemia and given the potential for missed diagnosis in these patients, CGM should be considered as an adjunctive diagnostic tool in all patients presenting for hypoglycemia evaluation.
Introduction
Insulinomas are insulin secreting tumors arising from spontaneous mutations of the ductal, acinar, or islet cells of the pancreas [1,2]. They are rare, having an incidence of only four cases per million people per year [3]. Patients typically present with fasting hypoglycemia, experiencing neurologic symptoms like confusion, changes in vision, or abnormal behavior and autonomic symptoms like palpitations, diaphoresis, or tremulousness [4]. These hypoglycemic episodes are due to decreased release of hepatic glucose. There is a slight predilection for females (57%) and those of older age (median of 50 years) [2,5]. Insulinomas can also be seen in MEN1 (multiple endocrine neoplasia 1) [2].
Diagnosis is made by finding elevated insulin levels during a hypoglycemic episode. This is usually achieved by monitoring glucose during a 72 hour fast in the hospital. However, patients with hypoglycemia unawareness often have an atypical presentation and can be difficult to diagnose. Their hypoglycemia unawareness is due to recurrent hypoglycemic episodes, which can desensitize the patient, especially in those with increased exposure to hypoglycemia and frequent episodes of hypoglycaemia [6-8]. In these patients, using a continuous glucose monitor in the outpatient setting can aid in the diagnosis of an insulinoma.
Case Description
A 34-year-old female was referred to our outpatient endocrinology clinic for evaluation and treatment of hypoglycemia that had been present for 3 years. The hypoglycemic episodes had become more frequent 3 months prior to her presentation, which prompted her to seek medical evaluation. She is an elementary school teacher and reported daytime hypoglycemic episodes that had started to impact her job performance. She had access to a glucometer at work, through the school nurse, and had confirmed blood sugars of less than 70 mg/dL on multiple occasions. The patient reported both hypoglycemic episodes and symptoms after eating meals high in non-complex carbohydrates. Symptoms and hypoglycemic episodes resolved after carbohydrate intake but would return a few hours after food intake. She denied nocturnal hypoglycemic symptoms and denied awareness of nocturnal hypoglycemic episodes. Her social habits were unremarkable; no history of alcohol consumption or restrictions in diet. There was no history of gastric-bypass or pancreatic surgery, no history of diabetes or impaired glucose tolerance, and the patient was otherwise in good health. Her physical exam was unremarkable and vital signs were stable. At the time of her initial consultation, she was advised to purchase a glucometer, check blood sugars fasting and when symptomatic, and she was referred to a dietitian for implementation of a reactive hypoglycemia diet.
The hemoglobin A1c value on the day of her initial consultation was 3.9%. Labs were also obtained 1 week later after a 12 hour overnight fast (Table 1). These labs were remarkable for undetectable beta-hydroxy butyrate level (<0>60 mL/min/1.73sq.m), normal ACTH (19 pg/ml) and cortisol (16.7 ug/dL) levels, and undetectable human insulin antibody (<0>Table 2). After seeing the dietitian, she commenced a reactive hypoglycemia diet which initially resulted in improved blood sugars and fewer episodes of hypoglycemia. However, a few weeks later, the patient reported that the reactive hypoglycemia diet was no longer working. A Dexcom G6 continuous glucose monitor (CGM) was ordered for the patient to wear for personal use due to the inappropriate response to the reactive hypoglycemia diet and due to laboratory evidence of inappropriate insulin secretion (10.6 mU/L) for a blood glucose level of 71 mg/dL.
| GLUCOSE 70-110 mg/dL |
71 | |
| BUN 6.0-24.0 mg/dL |
19.0 | |
| CREATININE 0.55-1.30 mg/dL |
0.99 | |
| BUN/CREAT RATIO 6-30 ratio |
19 | |
| SODIUM 136-145 mEq/L |
138 | |
| POTASSIUM 3.5-5.1 mEq/L |
4.4 | |
| CHLORIDE 98-107 mEq/L |
103 | |
| CO2 CONTENT 21.0-32.0 mEq/L |
27.1 | |
| CALCIUM 8.4-10.1 mg/dL |
9.2 | |
| EGFR (ACTUAL) mL/min/1.73sq.m |
>60 | |
| HEMOGLOBIN A1C, POC 4.3-5.7% |
3.9 | |
| INSULIN 3.0-28.0 mU/L |
10.6 | |
| C-PEPTIDE 0.78-5.19 ng/ml |
1.88 | |
| PROINSULIN <=8.0 pmol/L |
2.9 | |
| CORTISOL 3.1-22.4 ug/dL |
16.7 | |
| ADRENOCORTICOTROPIC HORMONE 6-58 pg/ml |
19 | |
| BETA-HYDROXYBUTYRATE <=0.3 MMOL/L |
<0> | |
| HUMAN INSULIN ANTIBODY 0.0-0.4 U/ML |
<0> |
| WBC 4.0-11.0 K/uL |
7.8 | |
| LYMPHOCYTES - REL (DIFF) 20.0-45.0% |
27.4 | |
| NEUTROPHILS - REL (DIFF) 35.0-75.0% |
64.4 | |
| MONOCYTES RELATIVE PERCENT 4.0-15.5% |
6.6 | |
| EOSINOPHILS, DIFF 0.5-5.0% |
1.2 | |
| BASOPHILS - REL (DIFF) 0.1-2.0% |
0.4 | |
| ABSOLU LYMPHOCYTES 1.1-4.4 K/uL |
2.1 | |
| NEUTROPHILS ABSOLUTE COUNT 2.2-7.9 K/uL |
5.1 | |
| MONOCYTES ABSOLUTE COUNT 0.0-1.1 K/uL |
0.5 | |
| EOS (ABSOLUTE) 0.00-0.50 K/uL |
0.10 | |
| BASO (ABSOLUTE) 0.0-0.2 K/uL |
0.0 | |
| RBC 3.63-4.92 M/uL |
4.55 | |
| NUCLEATED RED BLOOD CELLS /100 WBC |
0.0 | |
| HEMOGLOBIN 11.0-15.0 gm/dL |
14.0 | |
| HEMATOCRIT 33.0-47.0% |
40.5 | |
| MCV 77.0-106.0 fL |
89.1 | |
| MCH 26.0-35.0 pg |
30.8 | |
| MCHC 32.0-36.0 g/dL |
34.6 | |
| RDW 11.6-16.4% |
12.6 | |
| PLATELET COUNT, CITRATED 140-440 K/uL |
343 | |
| MPV, CITRATED 7.3-10.3 fL |
9.1 | |
| GLUCOSE 70-110 mg/dL |
67 (L) | |
| BUN 6.0-24.0 mg/dL |
16.0 | |
| CREATININE 0.55-1.30 mg/dL |
1.01 | |
| BUN/CREAT RATIO 6-30 ratio |
16 | |
| SODIUM 136-145 mEq/L |
140 | |
| POTASSIUM 3.5-5.1 mEq/L |
4.6 | |
| CHLORIDE 98-107 mEq/L |
105 | |
| CO2 CONTENT 21.0-32.0 mEq/L |
26.6 | |
| ALBUMIN 3.40-5.00 gm/dL |
3.50 | |
| TOTAL PROTEIN 6.4-8.2 gm/dL |
7.9 | |
| GLOBULIN, TOTAL 1.4-4.8 gm/dL |
4.4 | |
| MP A/G RATIO 1.0-2.5 ratio |
0.8 (L) | |
| BILIRUBIN, TOTAL 0.2-1.3 mg/dL |
0.3 | |
| ALKALINE PHOSPHATASE 46-116 U/L |
62 | |
| ALANINE AMINOTRANSFERASE (ALT)(SGPT) 12-78 U/L |
19 | |
| ASPARTATE AMINOTRANSFERASE (AST)(SGOT) 8-34 U/L |
17 | |
| CALCIUM 8.4-10.1 mg/dL |
9.4 | |
| EGFR (ACTUAL) mL/min/1.73sq.m |
>60 | |
| CHOLESTEROL 0-200 mg/dL |
198 | |
| TRIGLYCERIDES 30-150 mg/dL |
139 | |
| HDL 30-60 mg/dL |
48 | |
| VLDL 7-24 mg/dL |
28 (H) | |
| LDL CHOLESTEROL CALCULATED mg/dL |
122 | |
| NON HDL CHOL | 150.0 | |
| CHOL/HDL RATIO 0.00-5.00 ratio |
4.13 | |
| HEMOGLOBIN A1C, POC 4.3-5.7% |
4.4 | |
| OUT OF RANGE RESULT REPORTED TO? **REQ IF ABNORMAL** |
No | |
| CORTISOL 3.1-22.4 ug/dL |
23.7 (H) | |
| TSH 0.358-3.74 uIU/mL |
2.943 | |
| INSULIN 3.0-28.0 mU/L |
10.0 |
CGM data available for physician interpretation was limited to 72 hours due to close proximity of insurance approval for the device and her scheduled follow-up appointment with endocrinology (Figure 1). Glucometer data was available for a longer time period and was used for clinical correlation (Figure 2). Review of Dexcom CGM data revealed nocturnal hypoglycemia, despite her unawareness while sleeping. The patient was admitted to the hospital for a supervised 72 hour fast. She was advised to wear her CGM while hospitalized as her hypoglycemia awareness was impaired and we felt this would help facilitate more accurate timing of hypoglycemic episodes. Hypoglycemia was noted 9 hours into the fast and the patient was asymptomatic. Point of care (POC) glucose was 55 mg/dL, which correlated with plasma glucose of 55 mg/dL. Additional labs were drawn at the time of the episode and confirmed endogenous insulin secretion which was not suppressed by hypoglycemia (Table 3A). One milligram of glucagon was administered at the end of the fast and the plasma glucose level rose >25 mg/dL (Table 3B).
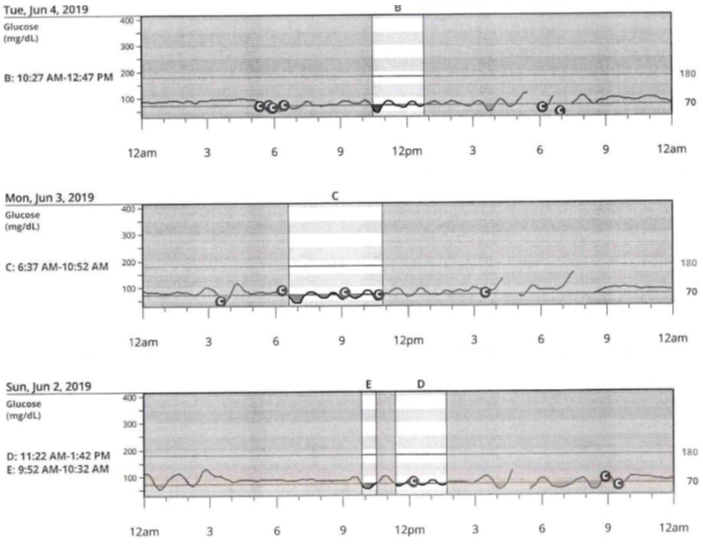
Figure 1. 72-hour CGM data confirming the presence of nocturnal and daytime hypoglycemia.
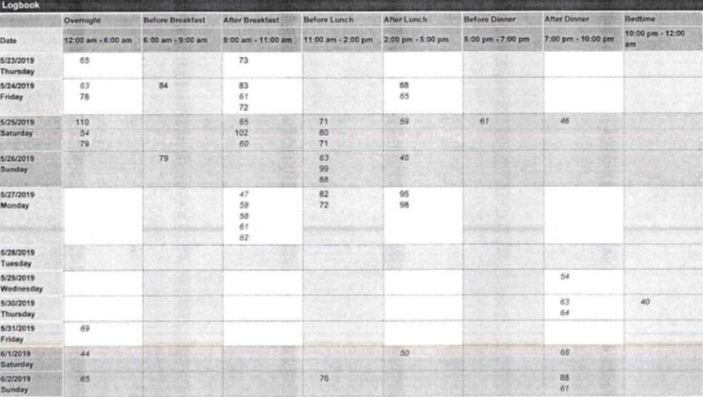
Figure 2. Glucometer data confirming the presence of nocturnal and daytime hypoglycemia.
| GLUCOSE 70.0-100.0 mg/dL |
55.0 (L) |
| CREATININE 0.6-1.1 mg/dL |
0.8 |
| CALCIUM 8.4-10.3 mg/dL |
9.7 |
| EGFR (ACTUAL) mL/min/1.73sq.m |
>59 |
| HEMOLYSIS INDEX | 0 |
| CHLORPROPAMIDE (GROUP) Not established UG/ML |
NEGATIVE |
| TOLAZAMIDE (GROUP) Not established UG/ML |
NEGATIVE |
| TOLBUTAMIDE (GROUP) Not established UG/ML |
NEGATIVE |
| GLYBURIDE (GROUP) Not established ng/ml |
NEGATIVE |
| GLIPIZIDE (GROUP) Not established ng/ml |
NEGATIVE |
| ACETOHEXAMIDE (GROUP) Not established UG/ML |
NEGATIVE |
| GLIMEPIRIDE Not established ng/ml |
NEGATIVE |
| NATEGLINIDE Not established ng/ml |
NEGATIVE |
| REPAGLINIDE Not established ng/ml |
NEGATIVE |
| BETA-HYDROXYBUTYRATE <=0.3 MMOL/L |
<0> |
| C-PEPTIDE 0.78-5.19 ng/ml |
1.67 |
| INSULIN <=24.6 uU/mL |
10.3 |
| PROINSULIN <=8.0 pmol/L |
4.8 |
| 1:05 AM | 2:25 AM | 2:35 AM | 2:45 AM | |
| GLUCOSE 70-100 MG/DL |
55 (L) | |||
| GLUCOSE 70.0-100.0 MG/DL |
106.0 (H) | 112.0 (H) | 57.0 (L) |
Abdominal helical computed tomography (CT) was then performed and revealed a 2.4 x 1.6 cm arterially hyper-enhancing mass in the tail of the pancreas, suspicious for neuroendocrine tumor, specifically insulinoma in the setting of hypoglycemia (Figure 3). Chest CT was then performed and showed no evidence of metastatic disease.
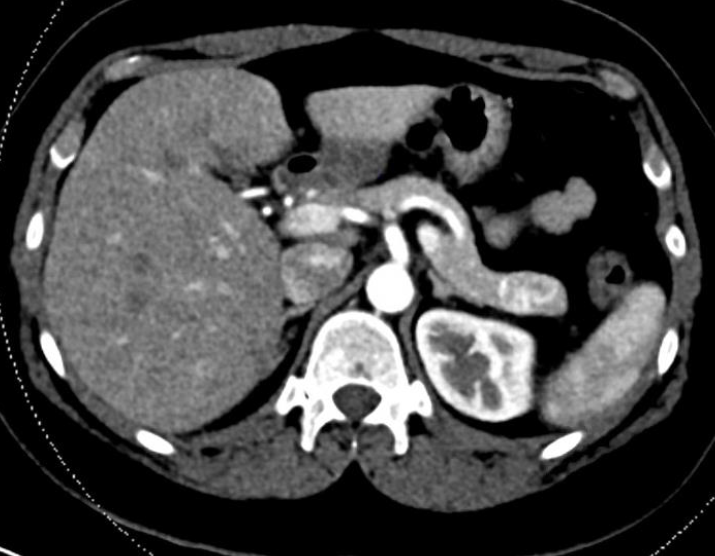
Figure 3. Contrast enhanced computed tomography (CT) image of the abdomen showing a 2.4 x 1.6 cm arterially hyper-enhancing tumor at the tail of the pancreas.
Surgical consultation was placed, given the clinical context, and laparoscopic distal pancreatectomy and splenectomy were recommended. Our patient remained hospitalized until the time of surgery 4 days later. In the interim, diazoxide (3 mg/kg/day) and 10?xtrose (D10) intravenous infusion were initiated for medical stabilization prior to surgery. The last dose of diazoxide was administered approximately 8 hours prior to surgery and D10 was discontinued immediately following tumor resection. The pathological examination confirmed a well-differentiated neuroendocrine tumor measuring 2 cm in diameter at the body and tail of the pancreas. Immunohistochemical staining was positive for synaptophysin and the KI-67 proliferative index was 1%. Pathologic Stage Classification (pTNM, AJCC 8th Edition): pT2 pN0. Staining for insulin and chromogranin A were not available at our institution.
Intraoperative POC blood glucose testing 20 minutes after tumor resection confirmed a stable blood glucose level of 137 mg/dL. Testing 60 minutes after surgery showed an insulin level of 7.0 mU/L, C-peptide level of 0.99 ng/ml and plasma glucose level of 226 mg/dL.
Our patient required transient insulin therapy for 72 hours post-op and was discharged home without insulin. She was advised to continue wearing her CGM. Six weeks after surgery the patient presented for outpatient endocrinology follow-up. No additional episodes of hypoglycemia were reported and was confirmed via review of CGM data (Figure 4). Fasting c-peptide and glucose levels 8 weeks after surgery were 2.31 ng/ml and 99 mg/dL, respectively.
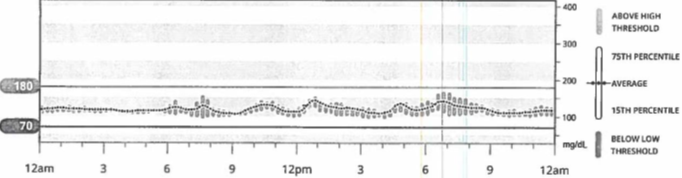
Figure 4. 14-day CGM summary from post-operative endocrinology follow-up visit. Confirming absence of hypoglycemia post-operatively.
Discussion
Insulinomas are rare neuroendocrine tumors of the pancreas. The hallmark of an insulinoma is fasting hypoglycemia. This can present with autonomic symptoms such as diaphoresis, tremor, palpitations, or neuroglycopenic manifestations which include confusion, visual changes, seizures, or coma. Insulinomas are difficult to diagnose. One study reports an average of 18 months of symptoms before an insulinoma diagnosis is made [1]. The gold standard for diagnosis of insulinoma remains measurement of plasma glucose, insulin, c-peptide, and proinsulin during a 72 hour fast.
CGM allows for the ongoing measurement of interstitial glucose. CGM can provide insight into the daily fluctuations of a patient’s glucose, providing the clinician with more data points to assist in diagnosis or management. Most of the literature related to CGM is regarding its use in diabetes management, although with improving technologies more applications of CGM are being researched.
Our case report demonstrates the clinical utility of CGM in diagnosing insulinomas. This case highlights the benefit of outpatient CGM in patients where hypoglycemia unawareness is suspected or present. Our patient was asymptomatic at night, despite having several hypoglycemic episodes, and she was symptomatic during the day. She was initially thought to have reactive hypoglycemia based on her history and symptoms. While hypoglycemia unawareness has been documented in patients with insulinoma, our patient’s daytime hypoglycemia awareness and nocturnal hypoglycemia unawareness are unique to this case. There are reports of CGM being used as diagnostic adjuncts in patients hospitalized for hypoglycemia secondary to insulinoma [9]. However, this case is unique because it highlights the clinical utility of CGM prior to hospitalization for 72-hour fast and emphasizes the strength of this technology when diagnostic ambiguity is present. Use of ambulatory CGM enabled us to detect the asymptomatic nocturnal hypoglycemic episodes and expedite the diagnosis of insulinoma.
Limitations to widespread use of outpatient CGM include cost and insurance coverage, both of which largely determine which patients are eligible for monitoring. The CGM data in this case was limited to 72 hours but was enough to lead to the appropriate diagnostic evaluation and treatment. Thus, there may be a role for professional CGM monitoring in which practices would purchase their own CGM devices that patients presenting with hypoglycemia could wear for 3 to 7 days. This would eliminate the need for insurance approval and would reduce barriers associated with the cost of personal CGM devices
Although insulinomas are very rare and difficult to diagnose, our case is an example of how ambulatory CGM may aid in quicker diagnosis and therefore a potential reduction in morbidity and mortality.
References
2. Placzkowski KA, Vella A, Thompson GB, Grant CS, Reading CC, Charboneau JW, Andrews JC, Lloyd RV, Service FJ. Secular trends in the presentation and management of functioning insulinoma at the Mayo Clinic, 1987–2007. The Journal of Clinical Endocrinology & Metabolism. 2009 Apr 1;94(4):1069-73.
3. SERVICE FJ, McMAHON MM, O'BRIEN PC, BALLARD DJ. Functioning insulinoma—incidence, recurrence, and long-term survival of patients: a 60-year study. In: Mayo Clinic Proceedings 1991 Jul 1 (Vol. 66, No. 7, pp. 711-719). Elsevier.
4. Dale AJ, Elveback LR, Jiang NS. Insulinoma: clinical and diagnostic features of 60 consecutive cases. InMayo Clinic Proceedings 1976 Jul (Vol. 51, No. 7, pp. 417-429).
5. Shao S, Zeng Z, Hu S. An observational analysis of insulinoma from a single institution. QJM: An International Journal of Medicine. 2018 Jan 8;111(4):237-41.
6. Halfdanarson TR, Rubin J, Farnell MB, Grant CS, Petersen GM. Pancreatic endocrine neoplasms: epidemiology and prognosis of pancreatic endocrine tumors. Endocrine-related cancer. 2008 Jun;15(2):409.
7. Cryer PE. Symptoms of hypoglycemia, thresholds for their occurrence, and hypoglycemia unawareness. Endocrinology and metabolism clinics of North America. 1999 Sep 1;28(3):495-500.
8. Martín-Timón I, del Cañizo-Gómez FJ. Mechanisms of hypoglycemia unawareness and implications in diabetic patients. World journal of diabetes. 2015 Jul 10;6(7):912.
9. Sugawa T, Murakami T, Yabe D, Kashima R, Tatsumi M, Ooshima S, Joo E, Wada K, Yoshizawa A, Masui T, Nakamoto Y. Hypoglycemia unawareness in insulinoma revealed with flash glucose monitoring systems. Internal Medicine. 2018 Dec 1;57(23):3407-12.
