Abstract
Background: Nimesulide, a preferential Cyclooxygenase-2 (COX-2) inhibitor, is frequently prescribed for musculoskeletal conditions in middle-aged (45–64 years) and older adults (≥65 years). Given age-associated alterations in drug metabolism, polypharmacy, and comorbidities, its safety and efficacy in adults >45 years warrants critical re-evaluation.
Objectives: To systematically synthesize and appraise evidence on the safety, tolerability, and therapeutic efficacy of oral nimesulide in adults >45 years across diverse pain-related indications, with emphasis on gastrointestinal, hepatic, renal, and cardiovascular risk domains.
Methods: A comprehensive systematic search of PubMed and Google Scholar in accordance to Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) 2020 guidelines, was conducted from inception through August 2025. Eligible studies included randomized controlled trials, observational studies, and pharmacovigilance datasets. Safety and efficacy outcomes were assessed.
Results: Thirty-four studies encompassing osteoarthritis, lower back pain, musculoskeletal disorders, and oral surgical pain were analyzed. Nimesulide in controlled trials demonstrated gastrointestinal tolerability. Hepatic adverse events were uncommon in interventional studies; however, pharmacovigilance signals indicated a low but measurable incidence of drug-induced liver injury, consistent with nonsteroidal anti-inflammatory drugs (NSAIDs) class effects. Renal and cardiovascular safety outcomes were comparable to other NSAIDs, with no consistent evidence of excess risk. Fixed-dose combinations with paracetamol exhibited favorable tolerability. Analgesic efficacy was consistent, with rapid onset and pain reduction.
Conclusion: Nimesulide demonstrated a favorable short-term safety profile in adults >45 years, with low incidences of serious gastrointestinal, hepatic, renal, and cardiovascular abnormalities. Pharmacovigilance data indicated monitoring of hepatic abnormalities. Evidence supports short-term tolerability, while emphasizing the importance of age-specific safety studies.
Keywords
Oral nimesulide, Nonsteroidal anti-inflammatory drugs, Older adults, Safety, Efficacy, Systematic review
Introduction
Pain is an often-debilitating symptom among older adult patients, and significantly impacts their quality of life, physical function, emotional well-being, and independence [1,2]. With increasing age, the risk of developing chronic pain conditions, such as osteoarthritis, musculoskeletal pain, low back pain, and neck pain, increases [2]. The global demographic shift towards aging populations and healthy ageing, effective and safe pain management in older adults is now a public health priority [3].
Nonsteroidal anti-inflammatory drugs (NSAIDs) effectively treat osteoarthritis, post-traumatic and postoperative pain, and other degenerative joint disorders [4]. However, NSAIDs can cause adverse events across multiple systems, such as gastric mucosa damage, renal complications such as hypertension, acute renal dysfunction and papillary necrosis, nephrotic syndrome; cardiovascular risks including myocardial infarction (MI), thromboembolism, and atrial fibrillation, most notably with diclofenac; hepatic effects ranging from aminotransferase level elevation to rare severe hepatotoxicity. Serious events like jaundice, necrosis of liver and its failure have been noted. Hematologic risks due to antiplatelet activity (nonselective NSAIDs) becomes a risk factor in patients with bleeding disorders or ulcers; and occasional hypersensitivity reactions like urticaria or aspirin-exacerbated respiratory disease [5].
NSAID use in the older adult population is not without risk. Advancing age is associated with pharmacokinetic and pharmacodynamic changes such as reduced renal clearance, altered hepatic metabolism, and changes in drug receptor sensitivity, which can increase the susceptibility to adverse effects [6]. The presence of age-related comorbidities, including hypertension, diabetes, cardiovascular disease, and chronic kidney disease, alongside frequent polypharmacy, elevates the risk of complications associated with NSAID therapy, such as gastrointestinal bleeding [7].
Despite these concerns, NSAIDs remain a cornerstone of pain management in older adults. Therefore, choosing an appropriate NSAID for use in the older adult population requires a careful balance between efficacy and safety. Nimesulide has been widely used for its analgesic and anti-inflammatory effects [8].
Nimesulide is classified as a preferential COX-2 inhibitor at low doses and a weak COX-1 inhibitor at high doses, belonging to the pyridinic sulfonamide NSAID class/group [9]. It is indicated for the treatment of painful inflammatory conditions, available in oral, dispersible, and topical formulations, offering flexibility in administration. Its widely increasing prescribing pattern is attributed to its rapid onset of action (15 minutes), short half-life (4 hours), and perceived lower gastrointestinal toxicity compared to traditional non-selective NSAIDs, such as ibuprofen, diclofenac, and piroxicam [10,11]. These features may be especially relevant in older adults seeking fast relief with a minimal risk of sedation and with simplified treatment regimens.
Pharmacologically, nimesulide exerts its effects primarily through selective inhibition of the COX-2 enzyme, reducing the synthesis of prostaglandins involved in inflammation and pain. This COX-2 selectivity is believed to spare COX-1–mediated gastroprotective prostaglandins, potentially offering a better gastrointestinal safety profile. Furthermore, its fast absorption and short plasma half-life make it suitable for short-term symptomatic relief [11]. Given these properties, the use of nimesulide may be considered in older adults when clinically appropriate, especially those seeking rapid pain relief with minimal sedation and manageable dosing schedules.
The safety profile of nimesulide, especially concerning hepatic, renal, and gastrointestinal systems, has been under scrutiny. Nimesulide has a safety profile that appears comparable to other commonly used NSAIDs in short-term use in certain populations, though concerns regarding hepatotoxicity persist in global literature [11]. In the SAFE-1 and SAFE-2 prospective multicentric studies conducted in India, which together included over 1,000 patients with acute pain conditions, found no clinically significant changes in liver or renal function parameters after treatment with nimesulide/paracetamol combinations [12,13]. Similarly, gastrointestinal tolerability has been favorable, with fewer adverse events reported compared to other NSAIDs in the elderly population.
As far as hepatotoxicity is concerned, at high concentrations, nimesulide can cause oxidative stress due to bio reductive metabolism, leading to mitochondrial dysfunction, glutathione depletion and ultimately hepatocellular necrosis [14]. Pharmaco-epidemiological reviews and meta-analyses have demonstrated that the incidence of drug-induced liver injury (DILI) with nimesulide is relatively low and comparable to agents like ibuprofen and diclofenac [15].
Meunier and Larrey examined cases of NSAID-related hepatotoxicity and used the Roussel-claf Causality Assessment Method to determine causality which demonstrated that 5.8% of NSAID-related DILI cases were attributed to nimesulide, significantly lower than diclofenac (34.1%) and ibuprofen (14.6%) [16].
Despite efficacy and safety comparable to traditional NSAIDs and lower gastrointestinal toxicity, increasing use of nimesulide in aging populations necessitates systematic evaluation of its safety and effectiveness in patients over 45 years, considering global demographic shifts and age-appropriate prescribing priorities.
This systematic review aims to consolidate and systematically evaluate current evidence on the efficacy, safety, and tolerability of nimesulide in patients >45 years of age, across a range of pain-related indications. The review will assess therapeutic outcomes, incidence of adverse events, particularly hepatic, gastrointestinal, cardiovascular, and renal and treatment patterns, including dosage and duration.
Methods
This study was carried out in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) standards. The PRISMA checklist is provided as Supplementary File 2.
Search strategy and study selection
An extensive review of the literature was conducted in PubMed and Google Scholar from inception to August 2025, particularly with a focus on the English language. Medical Subject Headings (MeSH) and free-text phrases related to “nimesulide”, “elderly”, “older adults”, “pain”, “osteoarthritis”, “musculoskeletal pain”, “dental pain”, “post-operative dental surgery”, “low back pain”, “pain relief”, “analgesia”, and “safety”, were combined in the search strategy. The Supplementary file 1 includes the detailed search strings for every database.
Additionally, conference abstracts from major rheumatology, pain medicine, and older adult medicine congresses, published up to August 2025, were also reviewed.
Eligibility criteria
A full-text review was conducted after two reviewers (X.X and X.X) independently screened the titles and abstracts. A third reviewer (X.X) was consulted to reconciliation of any discrepancies. The eligibility criteria were as follows:
a. Population: Patients >45 years of age (but studies may include wider age groups, 18–80 years).
b. Intervention: majorly Oral nimesulide (any dosage, monotherapy or combination therapy).
c. Comparator: Placebo, other NSAIDs, paracetamol, opioids, or non-pharmacological interventions.
d. Outcomes: Safety (adverse events, discontinuations due to adverse events, laboratory changes), tolerability, and Efficacy (pain intensity reduction, functional improvement), safety.
e. Study design: Randomized controlled trials (RCTs), prospective and retrospective cohort studies, and case-control studies. Case series, pharmacovigilance reports, and post-marketing surveillance data were included for safety analysis only.
The choice of parameters was guided by clinical relevance and prescribing patterns. The age threshold of 45 years was selected to capture middle-aged and older adults, reflecting the most frequently prescribed nimesulide demographically. Dose was selected according to the standard recommended dose prescribed. Comparators represent commonly used alternative medications. Outcomes were selected based on the systems most commonly affected by NSAIDs (gastrointestinal, hepatic, renal and cardiovascular), alongside efficacy measures relevant to pain-related conditions in older adults and diverse study designs included to capture both efficacy and real-world safety.
Short-term safety and tolerability were described as the incidence of adverse events and treatment-related discontinuations during therapy for around ≤15 days. Long-term safety and tolerability were described as adverse events or cumulative toxicities during treatment for around ≥15 days, including studies with a follow-up period extending to one year.
Classification of interventions
a.Dosage: standard dose (100 mg twice daily) vs. other dosing regimens.
b.Treatment duration: ≤15 days vs >15 days.
c.Comparator class: placebo, non-selective NSAIDs, selective COX-2 inhibitors, paracetamol, opioids, and others.
Outcomes of interests
The primary outcome was to evaluate nimesulide, with respect to the following systems: renal, gastrointestinal, hepatic, cardiac, and other systems. The secondary outcome was to assess the efficacy of nimesulide with respect to the following conditions: Osteoarthritis (OA), lower back pain (LBP), other musculoskeletal pain and conditions.
Data extraction
Two or more independent reviewers (X.X and X.X) used Microsoft Excel to extract the data. Data on participants, concepts, context, methods of study, and noteworthy results pertinent to the review question were all extracted. Comprehensive extraction forms had author information, their countries of origin, study characteristics (year, study design, age specific inclusion criteria, and treatment duration), patient characteristics (mean age, sec, indication and baseline pain intensity), and regimen characteristics (dose, comparator, and dose), and outcomes of interest (Western Ontario and McMaster Universities Arthritis Index (WOMAC), Visual Analogue Scale (VAS), Numeric rating scale (NRS), Quality of Life (QOL), rescue medication) were extracted. As required, draft extraction tool was modified and improved. Conflicts were settled by discussion or by consulting with another reviewer (X.X).
Assessing the risk of bias
For randomized studies, using the Risk of Bias 2 (RoB2) tool, and the Risk of Bias in Non-Randomized Studies of Interventions (ROBINS-I) tool, two reviewers (X.X and X.X) independently assessed the risk of bias [17,18]. A third reviewer (X.X) was involved, or through a conversation, the disagreements were settled.
Results
After a comprehensive investigation of two databases, a total of 98 articles were scanned and searched. Out of these, 34 articles were included. Literature search and flowchart for screening are presented in Figure 1.
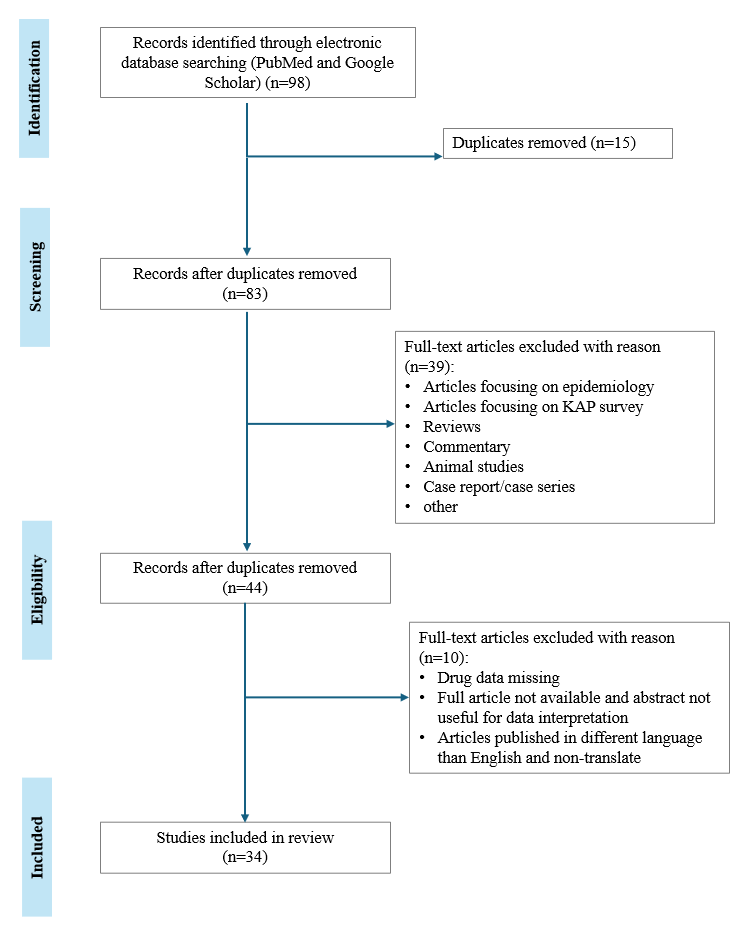
Figure 1. PRISMA flowchart of the search strategy and results.
Study characteristics
The main features of the included studies, such as authors, years, country of origin of the lead author, study design, sample size, dose of nimesulide, comparator, and demographical data of the participants, are compiled in Table 1.
|
Sr. No. |
Author, Year, Country |
Study Design |
Sample Size |
Indication |
Age Inclusion Criteria |
Study Drug (Name, Dose) |
Comparator (Name, Dose) |
Treatment Duration |
Mean Age |
|
1 |
Wang W et al. [19] |
Non- Randomized Controlled Trial (RCT) |
100 |
Traumatic knee osteoarthritis |
Not reported |
Nimesulide sustained-release capsule + Injection of Sodium Hyaluronate into the knee cavity |
Oral nimesulide sustained-release capsules |
Not reported |
Not reported |
|
2 |
Omololu B, et al. [20], 2005, Nigeria |
Double blind RCT |
67 |
Osteoarthrosis of the hip and knee joints |
35–80 years |
100 mg of nimesulide tablet |
Placebo for seven days and diclofenac 50 mg tablets twice daily |
7 weeks |
59.6 years (Nimesulide group) |
|
3 |
Kriegel W, et al. [21], 2001, Germany |
Double blind RCT |
370 |
Osteoarthritis |
Not Reported |
Nimesulide 100 mg twice daily |
Naproxen 250 mg morning, 500 mg evening |
One year |
Not Reported |
|
4 |
Edward C. Huskisson, et al. [22], 1999, United Kingdom and Ireland |
Randomized, double -masked, parallel-group study |
279 |
Osteoarthritis |
45 to 79 years |
Nimesulide 100 mg twice daily |
Diclofenac 50 mg three times daily |
24 weeks |
Nimesulide= 64.6 ± 9.1 |
|
5 |
Pochobradsky MG, et al. [23], 1991, Switzerland |
Multicentric trial |
22,938 |
Osteoarthritis |
Not reported |
Nimesulide tablets (40%) or granules (60%) from 100 to 400 mg/day |
Not reported |
1 to 3 weeks |
Not reported |
|
6 |
Alexeeva LI, et al. [24], 2009, Russia |
RCT |
40 |
Osteoarthritis |
60 to 74 years |
Nimesulide 200–100 mg/day |
Paracetamol till 3 g/day, in combination with local application of ketoprofen |
One Year |
67.1 ± 4.3 Group 1) |
|
7 |
M Quattrini, et al. [25], 1995, Italy |
RCT |
120 |
Osteoarthritis |
Not Reported |
Nimesulide 100 mg twice daily for 4 weeks |
Naproxen 500 mg twice daily for 4 weeks |
4 weeks |
66.37 years (Nimesulide group) |
|
8 |
V Fossaluzza, et al. [8], 1989, Italy |
Double-blind, randomized trial |
40 |
Osteoarthritis |
Not Reported |
100 mg Nimesulide granules |
250 mg Naproxen granules |
28 days |
Nim: 72.80 ± 0.54 |
|
9 |
Bianchi M, et al. [26], 2003, Italy |
Randomized, double-blind, clinical trial |
31 |
Osteoarthritis |
Aged 18 years or older |
Oral nimesulide (100 mg) |
Celecoxib (200 mg) and rofecoxib (25 mg) |
3 weeks |
69.0 years |
|
10 |
P W Lücker, et al. [27], 1994, Germany |
Double-blind, randomized, multi-center clinical study |
200 |
Osteoarthritis |
Not reported |
Nimesulide 200 mg/day |
Etodolac 600 mg/day |
3 months |
Not reported |
|
11 |
Dreiser RL, et al. [28], 1993, Switzerland |
Double-blind RCT |
174 |
Osteoarthritis |
Not reported |
Oral nimesulide (100 mg twice daily) |
Placebo |
Placebo: 2 weeks |
Not reported |
|
12 |
Bianca F E S, et al. [29], 2022, Brazil |
Randomized placebo-controlled split-mouth clinical trial |
40 |
Open flap periodontal surgery |
Between 18 and 60 years |
Nimesulide 100 mg |
Ibuprofen 600 mg |
Pre-operative dose administered 1 hour before procedure |
43.25 ± 11.78 (Ibuprofen group) |
|
13 |
Fábio Andrey da Costa Araújo, et al. [30], 2012, Brazil |
Prospective, randomized, controlled, paired trial |
47 |
Third molar surgery |
15–45 years |
100 mg nimesulide |
100 mg Tramadol chlorhydrate |
Pre-operative dose administered 1 hour before procedure |
Not reported |
|
14 |
Bocanegra M, et al. [31], 2005, Venezuela |
24-hour, double-blind, randomized, double-dummy, parallel-group study |
86 |
Surgical extraction of an impacted third |
12 to 60 years |
Nimesulide 300 mg tablet |
Ibuprofen 400 mg tablets |
24-hour duration of study |
25.2 years (Nimesulide group) |
|
15 |
Bracco P, et al. [32], 2004, Italy |
Randomized comparative study |
130 |
Extraction pain in oral surgery |
Not reported |
Nimesulide 200 mg/day |
Rofecoxib 50 mg/day |
7 days |
Rofecoxib group: 22.7 years |
|
16 |
K℩l℩ç E, et al. [33], 2014, Turkey |
RCT |
90 |
Endoscopic sinus surgery |
18–70 years |
Nimesulide 100 mg: Group III |
Oral placebo capsules: Group I |
Dose given 1 hour before procedure |
35.5 ± 12.9 (Group 1) |
|
17 |
Aho M, et al. [34], 2003, Finland |
Double-blind, randomized, active comparator-controlled clinical trial |
78 |
Postoperative tonsillectomy pain |
aged 14–58 years |
Nimesulide 100 mg |
Ibuprofen 800 mg |
7 days |
23 (14–40) = Nimesulide |
|
18 |
Ragot JP, et al. [35], 1993, France |
Double blind RCT |
159 |
Oral surgery |
between 12 and 60 years old. |
Nimesulide 100 mg (one 100 mg tablet and 1 placebo capsule); |
Niflumic acid (1 placebo tablet and one 250 mg capsule); |
Single dose study |
Nimesulide 100 mg: 22.3 years |
|
19 |
Chaudhary B, et al. [36], 2020 Nepal |
Non-RCT |
42 |
Surgical extraction of the |
18–59 years |
Nimesulide 100 mg |
Ketorolac 10 mg |
5 days |
30.83 ± 9.85 |
|
20 |
Pohjolainen T, et al. [37], 2000, Finland |
Randomized double-blind comparative trial |
104 |
Acute low back pain |
Not reported |
Oral nimesulide 100 mg twice daily plus placebo |
Ibuprofen 600 mg |
10 days |
Not reported |
|
21 |
Dhole S, et al. [38], 2025, India |
Multicenter, retrospective study analyzed records |
1,873 |
Acute painful conditions |
Aged 18 years |
Nimesulide-paracetamol combination |
Not reported |
707 patients (37.7%) received therapy for up to 5 days, 563 patients (30.1%) for 6–7 days, and 603 patients (32.2%) for 8–10 days |
45.54 ± 21.22 years |
|
22 |
Samartsev IN, et al [39], 2024, Russia |
Open observational study with a retrospective analysis |
80 |
Non-specific musculoskeletal pain in the lower back (ANBP) |
Not reported |
Group 3 nimesulide 200 mg/day 10 days; |
Group 1 aceclofenac 200 mg/day 10 days; |
10 days |
52.6 years |
|
23 |
Patil S, et al. [12], 2024, India |
Randomized, prospective, multicenter, active-controlled study |
600 |
Acute painful conditions |
≥18 years |
FDC of nimesulide (100 mg) + paracetamol (325 mg) [NP] |
Ketorolac (10 mg) [Kt] |
10 days |
38.6 ± 9.86 years (NP group) |
|
24 |
Tiwaskar M, et al. [13], 2023, India |
Prospective, open-label, and multicenter study |
464 |
Acute pain management |
Not reported |
Nimesulide + paracetamol (100 + |
Not reported |
Not reported |
Not reported |
|
25 |
U Marini, et al. [40], 1993, Italy |
Double-blind RCT |
30 |
To check gastric tolerability of nimesulide |
Not reported |
Nimesulide 100 mg, 200 mg |
Placebo |
7 days |
Not reported |
|
26 |
Scheinberg M, et al. [41], 2018, Brazil |
Multi-center, double-blind, double-dummy, active-controlled, parallel-group, noninferiority phase III study |
490 |
Osteoarticular diseases and dyspeptic symptoms |
>18 years of age |
Nimesulide/Pantoprazole (one tablet; 100 mg/20 mg) twice daily Placebo |
Naproxen/Esomeprazole Magnesium (one tablet; 500 mg/20 mg) plus placebo twice daily |
14 days |
Mean ± SD |
|
27 |
Alberto Pilotto , et al. [42], 2003, Italy |
Non-RCT |
Overall: 2251 |
The role of gastroprotective drugs |
Older than 65 years of age |
Nimesulide |
Diclofenac |
Not reported |
Overall: 80.18 ± 6.74 |
|
28 |
E Cunietti , et al. [43] 1993, Italy |
Double-blind RCT |
39 |
Pyrexia |
≥65 years |
Nimesulide 200 mg |
Paracetamol 500 mg suppositories |
2 days |
Not Reported |
|
29 |
Ashraf SM, et al. [44], 2025, India |
Prospective, multicenter, comparative, and observational study |
303 |
Fever or fever with pain |
≥18 to ≤60 years |
Group -nimesulide |
Group II-ibuprofen (400 mg) + |
10 days |
Groups I, II, and III were 33.90 ± |
|
30 |
Calligaris A et al. [45], 1993, Italy |
Randomized double-blind trial |
660 |
Minor sport injuries |
16–60 years |
Granular formulation of nimesulide (100 mg 3 times daily) |
Naproxen (250 mg 3 times daily) |
7 days
|
27 ± 7 years (Nimesulide group) |
|
31 |
Bavbek S, et al. [46], 2004,Turkey |
Single-blind placebo controlled oral challenges |
140 |
Comparison of |
Not reported |
Nimesulide (Mesulid 100 mg) |
Meloxicam (Mobic 7.5 mg) |
1 day |
Nimesulide: 39.46 ± 10.44 |
|
32 |
Monteiro C, et al. [47], 2022, Portugal |
Observational and retrospective analysis |
14 articles |
Safety of NSAIDs in the elderly: an analysis |
Age ≥ 65 |
Ibuprofen + nimesulide
|
Aceclofenac |
Variable |
Not reported |
|
33 |
Kim, et al. [48], 2015, Korea |
Nationwide cohort study |
117,610 |
GI bleeding and cardiovascular events due to NSAIDs |
Not reported |
Nimesulide |
Loxoprofen Sodium, Aceclofenac, Talniflumate, and Dexibuprofen consisting of 69.1% of the total NSAIDs |
Variable |
Initial cohort |
|
34 |
Conforti A et al. [49], 2001, Italy |
Pharmacovigilance study based on spontaneous reporting |
10, 608 reports describing 16, 571 ADRs |
Adverse drug reactions related to the use of NSAIDs |
Not reported |
Nimesulide |
Diclofenac |
Variable |
Not reported |
|
Abbreviations: NSAIDs: Non-Steroidal Anti-Inflammatory Drugs; RCT: Randomized Controlled Trial |
|||||||||
Study designs
After carrying out the systematic review of all the included articles, the study design of various studies comprised of double blind clinical trials, active controlled equivalence study, post-marketing survey, open controlled study, randomized study, multi-center study, split-mouth study, double dummy parallel group study, active comparator-controlled clinical trials, retrospective/prospective real-world evidence study, open observational study, and cohort study.
Sample size description
The sample sizes in all 34 studies had a broad range, ranging from a minimum of 30 patients [40] to as many as 117,610 patients [48]. The study population included both males and females. There were 11 studies with a mean age of their study population above 45 years, and based on available data from articles, 5 studies included only the older adult population.
Indications analyzed
The indications analyzed included osteoarthritis, lower back pain, musculoskeletal pain, oral/head and neck conditions, such as open flap periodontal surgeries, post-tooth extraction pain, third molar surgeries, endoscopic sinus surgeries and postoperative tonsillectomy pain. Other conditions included: osteoarticular diseases and dyspeptic symptoms, and minor sport injuries.
Common comparators of nimesulide
The common comparators of nimesulide include diclofenac, aceclofenac, naproxen, paracetamol, celecoxib, rofecoxib, piroxicam, meloxicam, lornoxicam, ketoprofen, dexketoprofen, ibuprofen, tramadol chlorhydrate, gabapentin, niflumic acid, ketorolac, and aspirin.
Regimens of nimesulide used: route of administration, dosage and treatment duration
The characteristics of the nimesulide regimen in this systematic review consist of many variations. The route of administration of nimesulide was majorly oral tablets. The medication administration was mainly twice a day (BID) and three times a day (TID). The dose of nimesulide varied from 100–300 mg/day. The duration of treatment ranged from a day [31,46] to as long as 1 year [24].
Follow-up durations
The included studies had follow-up durations ranging from as short as seven days to as long as three years.
Risk of bias in studies
The risk of bias in randomized controlled trials (RCT) was assessed using the Cochrane Risk of Bias 2 (ROB-2) tool. Most RCTs were rated at high risk in at least one domain, resulting in an overall high-risk of bias. Bias related to randomization, deviations from intended interventions, and missing outcome data were frequently rated as high or unclear risk. These issues stemmed from inadequate reporting of sequence generation and allocation concealment, limited details on blinding and adherence, and poor documentation of attrition. Additional concerns included reliance on subjective endpoints without standardized or blinded assessment, and the absence of trial protocols or registrations which prevented verification of prespecified outcomes and complete safety reporting. The detailed RoB2 assessment outcomes for individual RCTs are presented in Figure 2.
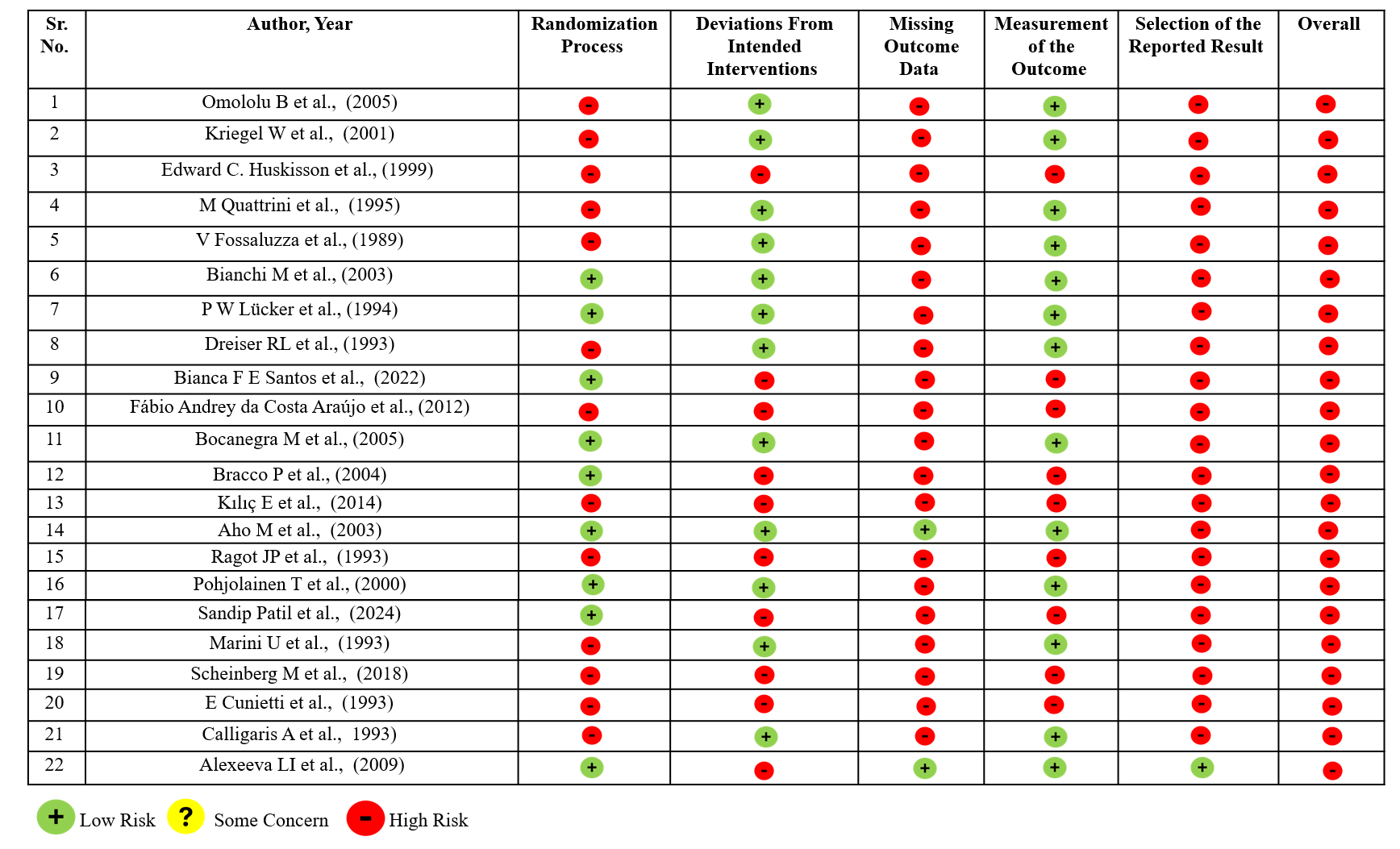
Figure 2. Summary of Cochrane Risk of Bias (ROB-2) assessments for individual Randomized Controlled Trial (RCT) studies.
Non-randomized studies were assessed using the ROBINS-1 framework, and most showed moderate to serious and, in some cases, critical risk of bias. The main issues were inadequate adjustment for baseline variables such as comorbidities and concurrent NSAID use, resulting in elevated confounding risk. Inconsistent documentation of dose and treatment duration, and incomplete reporting of adverse events. Intervention classification was limited by inconsistent documentation of Nimesulide dose, duration, and exposure. Missing data and selective reporting led to serious or critical bias ratings, while outcome measurement risks were lower. Overall, most studies showed serious bias, with some critical. Figure 3 displays the ROBINS-I evaluation outcomes for non-RCT studies.
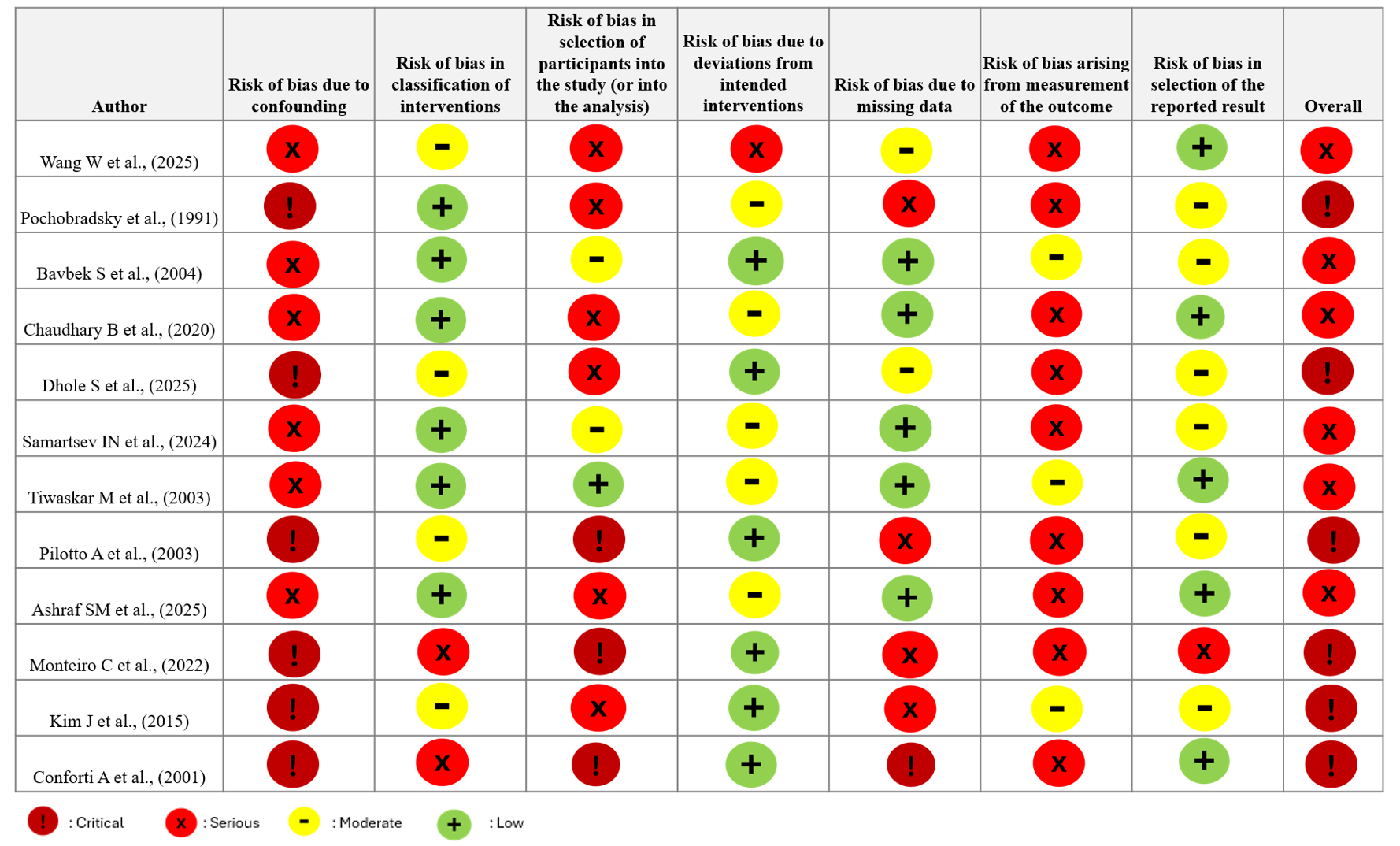
Figure 3. Summary of Cochrane Risk of Bias in Non-Randomized Studies of Interventions (ROBINS-I) assessments for individual non-Randomized Controlled Trial (non-RCT) studies.
Safety profile of nimesulide
Safety, both short-term and long-term was assessed for the studies. Table 2 summarizes the safety profile of Nimesulide across all 34 articles that are a part of this systematic review.
|
Sr. No. |
Author |
Year |
Serious Adverse Event |
Discontinuation Due To Nimesulide Adverse Event |
Mortality (All-Cause/Drug-Related) |
Gastro-Intestinal |
Hepatic |
Renal |
Cardio |
|
1 |
Wang W et al. [19] |
2025 |
Not reported |
Not reported |
Not reported |
Not reported |
Not reported |
Not reported |
Not reported |
|
2 |
Omololu B, et al. [20] |
2005 |
Nil |
Yes |
Nil |
Yes |
Nil |
Yes |
Nil |
|
3 |
Kriegel W, et al. [21] |
2001 |
Nil |
Nil |
Nil |
Yes |
Nil |
Nil |
Nil |
|
4 |
Huskisson, et al. [22] |
1999 |
Yes |
Yes |
Yes |
Yes |
Nil |
Nil |
Nil |
|
5 |
Pochobradsky MG, et al. [23] |
1991 |
Not reported |
Not reported |
Not reported |
Yes |
Nil |
Nil |
Nil |
|
6 |
Alexeeva LI, et al. [24] |
2009 |
Nil |
Yes |
Nil |
Yes |
Nil |
Nil |
Yes (Not TEAE) |
|
7 |
M Quattrini, et al. [25] |
1995 |
Nil |
Nil |
Nil |
Yes |
Nil |
Nil |
Nil |
|
8 |
V Fossaluzza, et al. [8] |
1989 |
Yes |
Yes |
Nil |
Yes |
Yes |
Nil |
Nil |
|
9 |
Bianchi M, et al. [26] |
2003 |
Nil |
Nil |
Nil |
Not reported |
Not reported |
Not reported |
Not reported |
|
10 |
P W Lücker, et al. [27] |
1994 |
Nil |
Not reported |
Nil |
Yes |
Nil |
Nil |
Nil |
|
11 |
Dreiser RL, et al. [28] |
1993 |
Nil |
Yes |
Nil |
Yes |
Nil |
Nil |
Nil |
|
12 |
Bianca F E S, et al. [29] |
2022 |
Not reported |
Not reported |
Not reported |
Not reported |
Not reported |
Not reported |
Not reported |
|
13 |
Araújo, et al. [30] |
2012 |
Nil |
Nil |
Nil |
Yes (In tramadol group only) |
Nil |
Nil |
Nil |
|
14 |
Bocanegra M, et al. [31] |
2005 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
15 |
Bracco P, et al. [32] |
2004 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
16 |
K℩l℩ç E, et al. [33] |
2014 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Yes |
|
17 |
Aho M, et al. [34] |
2003 |
Nil |
Nil |
Nil |
Yes |
Nil |
Nil |
Nil |
|
18 |
Ragot JP, et al. [35] |
1993 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
19 |
Chaudhary B, et al. [36] |
2020 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
20 |
Pohjolainen T, et al. [37] |
2000 |
Nil |
Yes |
Nil |
Yes |
Nil |
Nil |
Nil |
|
21 |
Dhole S, et al. [38] |
2025 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
22 |
Samartsev IN, et al. [39] |
2024 |
Not reported |
Not reported |
Not reported |
Yes |
Nil |
Nil |
Yes |
|
23 |
Patil S, et al. [12] |
2024 |
Nil |
Nil |
Nil |
Yes |
Yes |
Yes |
Yes |
|
24 |
Tiwaskar M, et al. [13] |
2023 |
Nil |
Nil |
Nil |
Nil |
No |
Nil |
Nil |
|
25 |
U Marini, et al. [40] |
1993 |
Nil |
Nil |
Nil |
Yes |
Nil |
Nil |
Nil |
|
26 |
Scheinberg M, et al. [41] |
2018 |
Nil |
Yes |
Nil |
Yes |
Nil |
Nil |
Nil |
|
27 |
Alberto Pilotto, et al. [42] |
2003 |
Nil |
Nil |
Nil |
Yes |
Nil |
Nil |
Nil |
|
28 |
E Cunietti, et al. [43] |
1993 |
Nil |
Yes |
Nil |
Nil |
Nil |
Nil |
Yes |
|
29 |
Ashraf SM, et al. [44] |
2025 |
Nil |
Nil |
Nil |
Yes |
Nil |
Nil |
Nil |
|
30 |
Calligaris A et al. [45] |
1993 |
Not reported |
Not reported |
Nil |
Yes |
Nil |
Nil |
Nil |
|
31 |
Bavbek S, et al. [46] |
2004 |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
Nil |
|
32 |
Monteiro C, et al. [47] |
2022 |
Yes |
Not reported |
Yes |
Yes |
Not reported |
Yes |
Yes |
|
33 |
Kim, et al. [48] |
2015 |
Yes |
Not reported |
Yes |
Yes |
Nil |
Nil |
Yes |
|
34 |
Conforti A et al. [49] |
2001 |
Yes |
Not reported |
Nil |
Yes |
Yes |
Yes |
Yes |
|
†: Nil: Active monitoring with no adverse events identified ‡: Not Reported: Adverse event assessment not conducted or documented |
|||||||||
Nimesulide and gastrointestinal (GI) safety
For quite some time, nimesulide has been renowned for its capacity to manage pain and inflammation. Its safety profile is also advantageous, particularly regarding a decreased likelihood of gastrointestinal adverse events. Nimesulide’s pharmacokinetics are also favorable, as it is absorbed quickly and completely [12].
In reviewing the literature for this systematic review, it was observed that earlier comparative studies (before 2010) consistently suggested that Nimesulide was better tolerated than diclofenac and naproxen, with fewer GI-related withdrawals and adverse events [8,20–22,25,45]. Large observational cohorts also reported overall good tolerability, and decreased GI adverse event incidence in comparison to comparison to diclofenac, ketoprofen, and piroxicam [23,49], while mixed results were seen against paracetamol and ibuprofen [24,34], and largely similar tolerability was noted with etodolac, piroxicam, and ketoprofen [27,28]. No significant differences in gastric mucosal injury were observed between nimesulide and placebo after short-term treatment [40]. At the same time, evidence indicated that nimesulide contributes to GI bleeding risk, with odds comparable to other NSAIDs but lower than naproxen [42].
However, in a 1999 study, one patient treated with Nimesulide, unfortunately passed away due to rectal bleeding, however that was not considered a drug related adverse event. In the same study, the treatment discontinuations were, [Total N (%): discontinuation due to GI-AEs n (%)]: nimesulide group: 31 (23%); 13 (9.6%), and diclofenac group: 35 (24.3%); 17 (11.8%) [22].
Araújo et al. (2012) reported no GI adverse events with nimesulide as a pre-emptive analgesic in dental surgery, while tramadol was associated with frequent nausea and vomiting. However, large pharmaco-epidemiological studies highlight a signal for GI bleeding risk [30]. Kim et al. (2015) observed that nimesulide use was connected to increased risk of upper GI bleeding, particularly in older adult patients having diabetes. Thus, precaution is warranted in these patients [48].
In a study conducted by Scheinberg et al. (2018) it was found that naproxen/esomeprazole magnesium (NE) and nimesulide/pantoprazole (N/P) resulted in similar GI event frequencies. However, there were five cases of treatment discontinuations due to adverse effects in the N/P group and six in NE group. The adverse events noted in this study were [NE; N/P n (%) (in that order)] abdominal discomfort 31 (8.4) ; 21 (5.7); abdominal distension 47 (12.8) ; 36 (9.8); abdominal pain 13 (3.5) ; 6 (1.6); upper abdominal pain 19 (5.2) ; 13 (3.6); constipation 8 (2.2) ; 2 (0.5); diarrhoea 11 (3.0) ; 8 (2.2); dysgeusia 1 (0.3) ; 9 (2.5); dyspepsia 42 (11.4) ; 39 (10.7); nausea 50 (13.6) ; 42 (11.5) [41].
Monteiro et al. (2022), observed in elderly patients, that there were few severe GI symptoms linked to nimesulide compared to diclofenac. Ibuprofen + nimesulide was suspected to cause Hematochezia in one of the studies they evaluated. Fixed-dose combinations (FDCs) have also been studied [47].
In a comparative study between nimesulide (100 mg) + paracetamol (325 mg) tablets[NP]; diclofenac (50 mg) + paracetamol (325 mg) tablets[DP]; ketorolac (10 mg) [Kt], and aceclofenac (100 mg) + paracetamol (325 mg) tablets [AP] conducted by Patil et al. (2024) showed that NP FDC had a favorable safety profile compared with DP or AP, with fewer GI adverse reactions. Adverse events reported in the study for NP (n = 150), n (%): nausea 2 (1.3%); gastritis 2 (1.3%); abdominal pain 3 (2%); dyspepsia 2 (1.3%) and diarrhea 1(0.6%), for DP (n = 150), n (%): nausea 5 (3.3%); vomiting 2 (1.3%); gastritis 4 (2.6%); abdominal pain 5 (3.3%); dyspepsia 1 (0.6%); diarrhea 1 (0.6%); for Kt (n = 150), n (%): nausea 4 (2.6%); vomiting 1 (0.6%); gastritis 2 (1.3%); abdominal pain 4 (2.6%); dyspepsia 2 (1.3%) and for AP (n = 150), n (%): nausea: 3 (2%); vomiting: 1 (0.6%); gastritis: 2 (1.3%); abdominal pain 2 (1.3%); dyspepsia 3 (2%) and diarrhea 2 (1.3%) [12].
Multiple gastric mucosa erosions, gastralgia and heartburn are some of the symptoms that have resulted in treatment discontinuations due to nimesulide in some studies [20,24,28]. Other common GI adverse events observed in all the studies include heartburn, nausea, diarrhea, constipation, dyspepsia, vomiting, abdominal pain, discomfort, distension and cramps, flatulence, isolated erosions, asymptomatic duodenal ulcer (DU), gastric acidity and intolerance, and epigastric pain.
Supplementary File 1 (Supplementary Table 1) presents an overview of the safety profile of nimesulide regarding the gastrointestinal safety found during this systematic review.
Nimesulide and hepatic safety
Approximately, 15% of NSAID users can develop transient mild serum aminotransferase elevation, with lower rates for nimesulide, while clinically significant increases occur in fewer than 1% individuals [50].
This systematic review demonstrated that Cunietti et al. (1993) found no alterations in hepatic, renal, or hematopoietic function on comparing nimesulide with paracetamol, supporting its short-term safety [43] .
However, larger observational datasets and pharmacovigilance analyses suggest that nimesulide carries a risk of drug-induced liver injury (DILI), although comparable to other NSAIDs. Pharmacovigilance data from Conforti et al. (2001), covering over 16,000 adverse drug reactions, indicated that liver and biliary system events accounted for 2.9% of reports for nimesulide, which was higher than diclofenac (1.4%), ketoprofen (0.4%), and piroxicam (0.5%). Serious reaction includes four reports of hepatitis in the patients who consumed nimesulide. These results emphasize the significance of careful patient selection and monitoring [49].
Whereas, in small-controlled studies, no evidence of hepatotoxicity was observed. Alexeeva et al. (2009), in a cohort of 40 women with osteoarthritis, stated that tests of hepatic function did not worsen with long-term nimesulide management, and no cases of hepatotoxicity were observed [24].
A study by Tiwaskar et al. (2023), conducted in 464 individuals receiving nimesulide–paracetamol FDC, found no significant rise in serum bilirubin, Serum Glutamate Pyruvate Transaminase (SGPT), or Serum Glutamic-Oxaloacetic Transaminase (SGOT) after two weeks of therapy, except only ALT levels showed a significant rise in patients with dental pain [13].
Patil et al. (2024) further confirmed that liver function test (LFT) variations in the NP group were statistically insignificant compared to Kt, DP, and AP groups. Elevated liver enzymes n (%) was 3 (2.0%) in the NP group; 4 (4.0%) in the DP group; 4 (2.6%) in the Kt group and 5 (3.3%) in the AP group [12].
Supplementary File 1 (Supplementary Table 2) presents a summary of nimesulide’s safety profile regarding hepatic safety found during this systematic review.
Nimesulide and renal safety
NSAID dose and duration influence organ toxicity; kidneys are commonly affected, and older adults face increased risk of reversible or permanent renal injury [51].
In this systematic review, we observed that in older studies, such as, Cunietti et al. (1993) [43], compared paracetamol and nimesulide in 39 patients and found both drugs to be well tolerated, with no changes in renal, hepatic or hematopoietic function, however Huskisson et al. (1999) [22], demonstrated a significant decrease from baseline for uric acid levels in the nimesulide group. Furthermore, Conforti et al. (2001), analyzed over 10,000 reports of adverse events to several NSAIDs, noting that nimesulide had a slightly higher proportion of urinary system toxicity reports compared to others, and cautioning against its use in patients with severe liver or kidney impairment. Serious reactions noted were nine reports of acute renal impairment for nimesulide and five reports of acute renal failure in diclofenac receiving patients [49].
A study conducted by Omolulu et al. (2005), on 67 osteoarthritis patients treated with diclofenac or nimesulide, observed reversible deep yellow urine in the nimesulide group but no biochemical or hematological abnormalities [20].
The recent study by Patil et al. (2024), assessed 600 patients with acute painful conditions across four treatment groups (NP, DP, AP and Kt). Abnormal renal function n (%) was 1 (0.6%) in NP group; 7 (4.7%) in DP group; 3 (2%) in Kt group and 3 (2%) in AP group. Changes in renal function tests, serum creatinine and blood urea nitrogen (BUN), from baseline to days 7 and 14 were not statistically significant in the NP or Kt groups. The DP group showed a statistically significant rise in serum creatinine at day 14, though the increase was not clinically relevant. The NP and DP groups showed a statistically significant difference in serum creatinine on days 7 and 14, whereas no statistically significant difference was observed between the NP and Kt groups[12].
Supplementary File 1 (Supplementary Table 3) presents an overview of the safety profile of nimesulide regarding renal safety found during this systematic review.
Nimesulide and cardiac safety
Although effective, NSAIDs require careful cardiovascular risk evaluation, as both COX inhibitors and traditional NSAIDs show varying cardiovascular effects across patient populations [52] .
In this systematic review, we found that in older studies, such as, Cunietti et al. (1993), found paracetamol and nimesulide to have stable cardiovascular parameters with only minor clinically insignificant blood pressure changes [43]. Then, Conforti et al. (2001) reported small differences in cardiovascular toxicity across NSAIDs, suggesting broadly comparable safety [49].
Later, Alexeeva et al. (2009) observed equal hypertension changes in both paracetamol and nimesulide groups, but noted coronary heart disease occurred only in patients receiving nimesulide (four cases), pointing to a potential cardiovascular risk [24].
K℩l℩ç et al. (2014), found postoperative tachycardia and hypertension were more frequent with placebo and nimesulide while gabapentin showed fewer cardiovascular events. Additionally, there were no cases of bradycardia and hypotension observed in both gabapentin and nimesulide group. Observed nimesulide group adverse events [n (%)]: tachycardia:10 (33%), hypertension: 6 (20%). There were no cases of bradycardia and hypotension [33].
Kim et al. (2015), in a large cohort of over 117,000 patients reported that nimesulide increased the likelihood of gastrointestinal bleeding, while ketorolac raised the risk of cardiovascular events when compared to celecoxib, in elderly diabetics [48]. In a study conducted by Scheinberg et al. (2018) it was found that naproxen/esomeprazole magnesium (NE) and nimesulide/pantoprazole (N/P) resulted in similar cardiac event frequencies. The adverse events noted in this study were [NE; N/P n (%) (in that order)] hypertension 4 (1.1) ; 6 (1.6) [41].
In 2024, Samartsev et al. [39], overall 41.3% of patients experienced adverse events, with aceclofenac showing the best safety profile and lornoxicam linked to higher cardiovascular risk, whereas Patil et al. (2024) [12] found no abnormal ECG changes across the four groups (NP, AP, DP and Kt). However, since NSAIDs carry dose and duration-dependent cardiovascular risk, it was suggested that patient factors and existing conditions must be evaluated for a safe medication choice.
Supplementary File 1 (Supplementary Table 4) presents an overview of nimesulide’s cardiac safety profile found during this systematic review.
Nimesulide and other systems safety
Adverse effects of NSAIDs also include hematologic adverse effects and other adverse events including, anaphylaxis, urticaria, and other skin and pulmonary and exacerbated respiratory reactions [53] .
NSAID-induced drug hypersensitivity reactions (DHRs) is the second most common drug reaction after antibiotics, with a reported prevalence of 0.6–2.5% in the general population, higher in women and in patients with chronic urticaria or asthma [54]. A United States (US) study found that, from around 1.8 million patients, NSAID-induced anaphylaxis was observed in 13 cases per 10,000 patients, underscoring its clinical significance [55]. In a retrospective study conducted by Bonadonna et al. (2022), showed that cohort comparisons demonstrated variations in NSAIDs incidence. In Sweden cohort, diclofenac was most suspected (44% of reactions, 75% of anaphylaxis), while in Italy acetylsalicylic acid (ASA) accounted for 30% of cases. In contrast, Nimesulide was linked to 43% of anaphylactic reactions, followed by diclofenac and ibuprofen (29% each). These differences likely reflect national prescribing and availability patterns, suggesting that consumption trends influence hypersensitivity prevalence [54].
This systematic review evaluation resulted in showing that studies occurring before 2010, reported nimesulide to be generally well tolerated with only isolated mild allergic reactions [43], and adverse event rates comparable to diclofenac in osteoarthritis patients [22]. In acute low back pain, nimesulide produced fewer withdrawals and slightly fewer adverse events than ibuprofen [37]. Large pharmacovigilance data also confirmed a toxicity profile similar to other NSAIDs [49]. Trials in tonsillectomy patients found no meaningful differences between nimesulide and ibuprofen [34]. However, a study in 2004 by Bavbek et al. reported a higher incidence of cutaneous reactions with nimesulide compared to meloxicam and rofecoxib [46].
In a study conducted by Scheinberg et al. (2018) it was found that naproxen/esomeprazole magnesium (NE) and nimesulide/pantoprazole (N/P) resulted in similar event frequencies. The adverse events noted in this study were [NE; N/P n (%) (in that order)] arthralgia 8 (2.2) ; 8 (2.2); back pain 6 (1.6) ; 8 (2.2); dizziness 9 (2.5) ; 9 (2.5); headache 26 (7.1) ; 29 (7.9); somnolence 15 (4.1) ; 8 (2.2) [41]. More recent evidence by Ashraf et al. (2025), showed nimesulide to have a comparable safety profile to paracetamol and ibuprofen-paracetamol combination. Common adverse events included, headache and somnolence, one case each, in the nimesulide group [44].
Symptoms such as facial oedema, vertigo, insomnia, broncho-obstructive symptoms, cutaneous erythema resulted in treatment discontinuations [8,28,37,43]. Other common adverse events observed in these studies include headache, petechiae, micro erosions, fatigue, pruritis, skin rash, dizziness, blurry vision, respiratory issues, bleeding, tiredness, emotional lability, fever, itching, asthmatic reactions, cutaneous reactions, rhinitis symptoms, urticaria, angioedema, nasal discharge, laryngeal oedema, Stevens-Johnson syndrome and erythema multiforme.
Supplementary File 1 (Supplementary Table 5) presents a summary of nimesulide’s safety profile relating to other systems’ safety found during this systematic review.
Efficacy of nimesulide: osteoarthritis (OA), lower back pain, and other musculoskeletal pain
The efficacy of nimesulide included assessing the following conditions: osteoarthritis, lower back pain, and other musculoskeletal pain, and several other conditions such as tooth extractions and oral surgeries. Osteoarthritis manifests itself in the form of joint pain and functional loss, affecting about 3.3 to 3.6% of the global population. The primary objective of treating osteoarthritis is to alleviate the pain and to increase functional abilities [56]. Back and musculoskeletal pain are extremely common among adults globally, several due to idiopathic causes. The treatment includes nonpharmacological and pharmacological management plans [57] .
According to this systematic review, a study conducted by Lücker et al. (1994), found nimesulide and etodolac equally effective in knee osteoarthritis, though physicians favored nimesulide [27]. Pohjolainen et al. (2000) showed that nimesulide demonstrated greater effectiveness than ibuprofen in improving bending movements in acute low back pain [37]. Omolulu et al. (2005) reported better pain control with nimesulide than diclofenac in hip and knee osteoarthritis [20]. Bianchi et al. (2003) demonstrated faster and sustained relief with nimesulide compared to celecoxib and rofecoxib in knee osteoarthritis [26]. Alexeeva et al.(2009) found nimesulide to be more effective than paracetamol in reducing osteoarthritis symptoms over nine months [24]. Samartsev et al. (2024) observed that aceclofenac and nimesulide were found to be better than dexketoprofen and lornoxicam in low back pain outcomes, though nimesulide carried a higher chronic pain risk [39]. Patil et al. (2024), showed that the fixed-dose nimesulide-paracetamol combination provided better pain relief than ketorolac and was non-inferior to diclofenac-paracetamol or aceclofenac-paracetamol combination [12].
Table 3 demonstrates a comparison of the articles included in this systematic review relating to nimesulide and its efficacy in treating osteoarthritis, lower back, and musculoskeletal pain.
|
Study |
Sample Size and Population Details |
Intervention |
Efficacy |
Inference |
|
Omololu B et. al (2005) [20] |
N=67 patients with osteoarthrosis of the hip and knee joints |
Diclofenac 50 mg and nimesulide 100 mg |
14.7% of patients in the nimesulide group experienced moderate pain, whereas 38.5% in the diclofenac group reported the same level of pain. Additionally, only 2.1% of individuals in the nimesulide group indicated severe pain at the end of the trial, in contrast to 18.4% of patients in the diclofenac group. |
A lower proportion of patients in the nimesulide group reported moderate or severe pain by the end of the trial, indicating better overall pain control compared with diclofenac. |
|
Alexeeva et al. (2009) [24] |
N=40 women having osteoarthritis |
Nimesulide 100–200 mg/day and paracetamol till 3 g/day |
Nimesulide group displayed statistically significant decrease in the overall WOMAC index after the 9th month of therapy (p<0.05). |
This shows that nimesulide was more effective than paracetamol in reducing the osteoarthritis symptoms |
|
Lücker PW et al. (1994) [27] |
N=200 patients with knee osteoarthritis |
Nimesulide 200 mg/day, and etodolac 600 mg/day |
While spontaneous pain demonstrated a noteworthy improvement throughout the study, there was no significant difference in the effectiveness of either compound. Likewise, there was a progressive and significant decrease in the Lequesne functional index, though no statistical difference was observed between nimesulide and etodolac. |
The physician's overall evaluation of efficacy was significantly more favorable towards nimesulide, but there was no such bias in the assessment for patients who completed all 12 weeks. |
|
Bianchi M et al. (2003) [26] |
N=31 Symptomatic Knee Osteoarthritis patients. |
Oral nimesulide (100 mg), celecoxib (200 mg) and rofecoxib (25 mg) |
Results showed that administering a single 100 mg dose of nimesulide offered a more significant therapeutic effect compared to celecoxib 200 mg or rofecoxib 25 mg over a 3-hour time frame in knee osteoarthritis patients. This difference was apparent on both the initial and final days of a week-long therapy. |
The enhanced therapeutic effect was consistent both at the start and end of the week-long treatment period, indicating that nimesulide provided more effective and sustained pain relief. |
|
Pohjolainen T et al. (2000) [37] |
N=104 patients to test the efficacy of COX-2-selective anti-inflammatory drugs on acute low back pain. |
Nimesulide 100 mg and Ibuprofen 600 mg |
The ability of patients to perform daily activities showed enhancements in both groups (P<0.001), but a statistically significant difference was observed between the two groups, favoring the nimesulide group (P<0.05) after 10 days. Nimesulide demonstrated greater effectiveness than ibuprofen in improving lateral bending measurements (P=0.026). |
Both nimesulide and ibuprofen resulted in similar improvements in the modified Schober tests, as well as in pain intensity and back stiffness scores. |
|
Samartsev IN et al. (2024) [39] |
N=80 patients (acute non-specific low back pain) |
Aceclofenac 200 mg/day; dexketoprofen 50 mg/day; nimesulide 200 mg/day; lornoxicam 16 mg/day. |
Notably higher percentage of patients in all assessed groups considered the therapy's improvement as "significant", with no differences observed among the groups. The relative risk of chronic low back pain was considerably greater with nimesulide (OR=4.378 (0.95% CI 1.022-18.765)) and lornoxicam (OR=4.426 (0.95% CI (1.014-19.316)), while the rate of recurrence was increased with dexketoprofen (OR=2.743 (0.95% CI 1.076 - 6.994)) in comparison to aceclofenac. |
Aceclofenac and Nimesulide were found to be more effective than Dexketoprofen and lornoxicam regarding functional activity linked to low back pain and overall health-related quality of life. |
|
Patil S et al. (2024) [12] |
N=600 patients with acute painful conditions |
Fixed-dose combination (FDC) of nimesulide 100 mg + paracetamol 325 mg [NP], ketorolac 10 mg [Kt] alone, diclofenac 50 mg + paracetamol 325 mg [DP], and aceclofenac 100 mg + paracetamol 325 mg [AP] |
The NP group experienced a significantly greater reduction in pain (p<0.001) when compared to the Kt group, and their results were non-inferior to those of the DP and AP groups on days seven and 14 |
NP provided better pain relief to Kt and performed at least as effectively as DP and AP over the treatment period. |
|
Abbreviations: Abbreviation: AP: Aceclofenac+Paracetamol; CI: Confidence Interval; COX-2: Cyclooxygenase-2; DP: Diclofenac+Paracetamol; FDC: Fixed-Dose Combination; Kt: Ketorolac; NP: Nimesulide+Paracetamol; OR: Odds Ratio; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index |
||||
Efficacy of nimesulide with respect to other conditions
The other efficacy-related conditions included in this review are oral surgeries, post-operative tonsillectomy pain control, and wisdom tooth extractions. As far as other conditions are concerned, this systematic review demonstrated that a study done by Ragot et al. (1993), showed that nimesulide provided significantly greater analgesia than placebo in oral surgery patients [35]. In 2003, Aho et al. [34] found that nimesulide offered faster pain relief and earlier return to daily activities compare to ibuprofen in tonsillectomy patients, and in 2012, da Costa Araújo et al. [30] reported that nimesulide and tramadol chlorhydrate had comparable analgesic effects in third molar extractions, with no significant difference in pain scores.
Supplementary File 1 (Supplementary Table 6) demonstrates a comparison of the articles included in this systematic review relating to nimesulide’s efficacy in treating other conditions.
Discussion
Safety of nimesulide
Nimesulide has been widely studied for both its safety and efficacy. The systematic review conducted here demonstrated that in terms of GI safety, many studies reported that nimesulide is better tolerated than diclofenac and comparable to naproxen and ibuprofen, with some evidence suggesting fewer toxicities [8,20–22,25,34,37,45,47]. However, results are not entirely consistent, as a study highlighted increased risks of GI bleeding in elderly diabetic patients [48]. One of the studies also noted that aceclofenac demonstrated a more favorable overall safety profile compared to dexketoprofen, nimesulide, and lornoxicam [39].
Hepatic safety is the most critical concern, with rare but serious cases of liver injury reported, sometimes even after short-term use of just 3–5 days [34]. While some studies did not find any significant changes in liver function tests with nimesulide or its combinations, the potential for hepatotoxicity remains a limiting factor in its use.
With prolonged use, NSAIDs may cause nephrotoxicity. The older adult age group, along with comorbidities, are often the contributing factors in decreasing the glomerular filtration rate and this increases the hazard of kidney dysfunction among NSAID users [51].Renal safety outcomes are generally reassuring, with studies showing no major alterations in renal function and better safety compared to diclofenac and aceclofenac combinations, though caution is advised in patients with severe kidney impairment [12].
In 2006 a meta-analysis was published to assess traditional NSAIDs effect on cardiovascular health. Their review showed that the risk of cardiovascular toxicity was increased with high doses of ibuprofen and high doses of diclofenac, but high dose naproxen related risks were lower [58]. Another meta-analysis in 2013, yielded similar results [59]. While the cardiovascular risks related to NSAIDs vary, the Food and Drug Administration (FDA) currently states that the evidence is not robust enough to conclude the degree of risk relating to a specific NSAID in comparison to others [52,60]. In this systematic review, cardiovascular risks are variable across NSAIDs. One study noted coronary heart disease indicators in nimesulide users [24], whereas another study found no abnormal ECG changes [12], leaving the evidence inconclusive. Mild adverse effects such as rash, itching, dizziness, and fatigue were reported but were not clinically significant.
Efficacy of nimesulide
As far as efficacy is concerned, this systematic review reported that for the treatment of osteoarthritis, Nimesulide has shown better efficacy compared to diclofenac [20], paracetamol [24] and celecoxib or rofecoxib [26], though results were similar to etodolac [27]. In lower back pain, nimesulide was better than ibuprofen in improving daily activity performance [37] and in postoperative pain, it was more effective than ibuprofen following tonsillectomy [34], while showing comparable outcomes to tramadol in dental surgery [30]. Combination therapy with paracetamol demonstrated strong pain reduction, proving non-inferior to DP and AP combinations, and superior to ketorolac in some measures [12]. Studies also suggest that nimesulide and aceclofenac, may contribute to a better overall quality of life compared to other NSAIDs such as dexketoprofen and lornoxicam [39] .
In summary, nimesulide offers effective pain relief with generally favorable GI tolerability and promising efficacy across a range of conditions. However, its rare but serious risk of hepatotoxicity remains the most significant safety concern, warranting careful monitoring and judicious use. Renal and cardiovascular safety profiles are largely comparable to other NSAIDs. Overall, nimesulide can be considered a useful analgesic and anti-inflammatory agent, but its safety profile requires cautious prescribing, particularly in populations at risk for liver or kidney complications.
Strengths and Limitations
This systematic review synthesizes a substantial body of clinical and real-world evidence, incorporating randomized trials, prospective and retrospective cohort studies, and pharmacovigilance datasets to provide a comprehensive, multi-domain characterization of nimesulide safety in adults >45 years. The systematic extraction of both clinical adverse events and laboratory parameters strengthens the internal consistency of safety interpretation across diverse study designs. While the included studies varied in sample size, monitoring intensity, and reporting granularity, most provided sufficient data to evaluate gastrointestinal, hepatic, renal, and cardiovascular safety outcomes, enabling meaningful synthesis of safety signals. Variability in age stratification and the predominance of short-duration treatment windows reflect typical real-world prescribing patterns for nimesulide and do not detract from the overall conclusion of its favorable short-term tolerability. Nonetheless, harmonized safety endpoints and contemporary, methodologically robust studies would further refine the precision of age-specific risk estimates and strengthen long-term safety characterization.
The inclusion of studies enrolling adults ≥18 years introduces age-related heterogeneity that may constrain the interpretability of results intended for a >45-year target population. Although the methodological and clinical variability across the 34 included studies on nimesulide precluded formal meta-analysis, this systematic review reflects a wide range of indications in which oral nimesulide's safety and efficacy have been studied. Grading of Recommendations Assessment, Development, and Evaluation (GRADE) was not performed due to heterogeneity and sparse arm-level data. The studies included in this review have their inherent limitations. Their findings may not be generalizable to elderly populations, as many trials exclude individuals with cognitive decline or renal and hepatic impairments. Long-term safety outcomes are often not captured, and clinical heterogeneity extends beyond age differences to include variations in comorbidities and baseline risk profiles. Moreover, inconsistencies in outcome definitions across studies, the absence of quantitative synthesis, and reliance on subjective interpretation further constrain the robustness of the evidence. Hence, these are limitations of the current systematic review as well.
Conclusion
Across all the studies in this systematic review, including both middle-aged and broader adult populations, nimesulide demonstrated a safety profile characterized by low frequencies of serious adverse events and generally mild, transient gastrointestinal, hepatic, renal, and cardiovascular events. Nimesulide provides effective analgesia and favorable tolerability in adults, with evidence suggesting fewer gastrointestinal adverse events compared to several traditional NSAIDs. However, concerns regarding hepatotoxicity and the methodological limitations of existing studies such as small sample sizes, heterogenous populations, and a high risk of bias, necessitate cautious interpretation.
Clinically, nimesulide may be considered for short-term use in selected older adult patients, provided hepatic function is monitored and comorbidities are carefully evaluated. Its potential role in multimodal pain management strategies, especially where gastrointestinal safety is a priority, is noteworthy. Future research should prioritize large, well-designed randomized controlled trials specifically targeting older populations, with standardized outcome measures and long-term follow-up to better define gastrointestinal, hepatic, renal and cardiovascular risks. Comparative effectiveness studies against newer COX-2 inhibitors and other analgesics, alongside pharmaco-epidemiological surveillance, will be essential to clarify real-world safety profile.
Acknowledgements
The authors sincerely thank IntelliMed Healthcare Solutions Pvt. Ltd. (Mumbai, India) for editorial assistance in literature search, drafting the manuscript, and journal submission.
Author's Contribution Statement
All listed authors satisfy the International Committee of Medical Journal Editors (ICMJE) criteria for authorship, accept accountability for the accuracy and integrity of the work, and approved the final manuscript version for publication. Author contributions were as follows: Dr. Anil Jain: Conceptualized and designed the systematic review, and manuscript writing-review and editing. Dr. Mansij Biswas: Data extraction, data synthesis, data interpretation, and manuscript writing- review and editing. Mr. Prince Bakshi: Validation, project administration, and manuscript writing-review and editing. All authors have read and approved the final version of the manuscript.
Conflict of Interest
All authors are permanent salaried employee of Dr Reddy’s Laboratories Limited, Hyderabad, India, which commercially manufactures and markets nimesulide in several countries. The authors do not have any other financial or personal relationships that could have influenced the content of this article.
Funding Statement
Dr Reddy’s Laboratories Limited funded the editorial and publication processing charges.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Disclaimer
The views/ opinion expressed and/or the information provided in this article are those of the respective author(s) and are meant for use by registered healthcare professionals only. They do not purport to reflect the opinion or views of Dr. Reddy’s Laboratories Limited and its affiliates (collectively “Dr. Reddy’s”) in any manner whatsoever and accordingly, Dr. Reddy’s does not recommend or make any representation about the appropriateness of the views/ opinion/ information contained herein. The article may discuss uses and dosage for therapeutic products that may not have been approved by the relevant regulatory agencies. Dr. Reddy’s does not support, endorse and/ or encourage any off-label usage.
References
2. Domenichiello AF, Ramsden CE. The silent epidemic of chronic pain in older adults. Prog Neuropsychopharmacol Biol Psychiatry. 2019 Jul 13;93:284–90.
3. Zubiashvili T, Zubiashvili N. Population Aging – A Global Challenge. ECOFORUM. 2021 oct;10(20):25.
4. Dilleban J, Sundaresan T. Role of non-steroidal anti-inflammatory drugs in orthopedics. Sys Rev Pharm. 2023 May 1;14:375–9.
5. Ghlichloo I, Gerriets V. Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). Treasure Island (FL): StatPearls Publishing; 2025 [cited 2026 Mar 4]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK547742/.
6. Ngcobo NN. Influence of Ageing on the Pharmacodynamics and Pharmacokinetics of Chronically Administered Medicines in Geriatric Patients: A Review. Clin Pharmacokinet. 2025 Mar;64(3):335–67.
7. Uskur T, Kurt BF. Polypharmacy and Drug-Drug Interactions in Elderly Patients With Gastrointestinal Bleeding: A Single-Center Retrospective Study. Cureus. 2025 Jan 23;17(1):e77866.
8. Fossaluzza V, Montagnani G. Efficacy and tolerability of nimesulide in elderly patients with osteoarthritis: double-blind trial versus naproxen. J Int Med Res. 1989 May-Jun;17(3):295–303.
9. Leong CS, Man CC, Kan AK, Li PH. Review of NSAID Hypersensitivity Reactions Based on Clinical Phenotyping. J Clin Rheumatol Immunol. 2024 Jul 22;24(01):1–10.
10. Kulkarni SV, Ahmed J, Pal M, Dharewa BK, Veer V, Petare AU, et al. Narrative Review of Nimesulide in Adults: Current Scenario. Indian Journal of Clinical Practice. 2022 oct;33(5):13–15.
11. Tiwaskar M, Charde S, Balankhe N, Hegde A, Deodhar M, Gondane A, et al. Nimesulide: Critical Appraisal of Safety and Efficacy in Acute Pain. J Assoc Physicians India. 2025 Mar;73(3):e22–8.
12. Patil S, Nadaf N, Gupta S, Barai P, Makhija S, Lodha P, et al. A Comparative Analysis of the Efficacy and Safety of Nimesulide/Paracetamol Fixed-Dose Combination With Other NSAIDs in Acute Pain Management: A Randomized, Prospective, Multicenter, Active-Controlled Study (the SAFE-2 Study). Cureus. 2024 Apr 23;16(4):e58859.
13. Tiwaskar M, Muruganathan A, Gondane A, Pawar D. An Open-label, Prospective, Multicentric, Cohort Study of Nimesulide/Paracetamol Fixed Drug Combination for Acute Pain Management: Sub-group Analysis. J Assoc Physicians India. 2023 Apr;71(4):11–2.
14. Boelsterli UA. Mechanisms of NSAID-induced hepatotoxicity: focus on nimesulide. Drug Saf. 2002;25(9):633–48.
15. Low EXS, Zheng Q, Chan E, Lim SG. Drug induced liver injury: East versus West - a systematic review and meta-analysis. Clin Mol Hepatol. 2020 Apr;26(2):142–54.
16. Meunier L, Larrey D. Recent Advances in Hepatotoxicity of Non Steroidal Anti-Inflammatory Drugs. Ann Hepatol. 2018 Mar 1;17(2):187–91.
17. Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019 Aug 28;366:l4898.
18. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016 Oct 12;355:i4919.
19. Wang W, Du Y, Zhang X. Nimesulide and sodium hyaluronate ameliorate the inflammatory process and pain severity in traumatic knee osteoarthritis. Inflammopharmacology. 2025 Jun;33(6):3135–42.
20. Omololu B, Alonge TO, Ogunlade SO, Aduroja OO. Double blind clinical trial comparing the safety and efficacy of nimesulide (100mg) and diclofenac in osteoarthrosis of the hip and knee joints. West Afr J Med. 2005 Apr-Jun;24(2):128–33.
21. Kriegel W, Korff KJ, Ehrlich JC, Lehnhardt K, Macciocchi A, Moresino C, et al. Double-blind study comparing the long-term efficacy of the COX-2 inhibitor nimesulide and naproxen in patients with osteoarthritis. Int J Clin Pract. 2001 Oct;55(8):510–4.
22. Huskisson EC, Macciocchi A, Rahlfs VW, Bernstein RM, Bremner AD, Doyle DV, et al. Nimesulide versus diclofenac in the treatment of osteoarthritis of the hip or knee: an active controlled equivalence study. Curr Ther Res Clin Exp. 1999 May 1;60(5):253–65.
23. Pochobradsky MG, Mele G, Beretta A, Montagnani G. Post-marketing survey of nimesulide in the short-term treatment of osteoarthritis. Drugs Exp Clin Res. 1991;17(3):197–204.
24. Alekseeva LI, Karateev AE, Popkova TV, Novikova DS, Sharapova EP, Markelova EP, et al. Efficacy and safety of long-term use of nimesulide in patients with osteoarthritis: results of a 12-month open-label controlled study DINAMO (Long-Term Use of Nimesulide in Osteoarthritis Multifactorial Assessment). Scientific and practical rheumatology. 2009(4):64–72.
25. Quattrini M, Paladin S. A Double-Blind Study Comparing Nimesulide with Naproxen in the Treatment of Osteoarthritis of the Hip. Clin Drug Investig. 1995 Sep;10(3):139–46.
26. Bianchi M, Broggini M. A randomised, double-blind, clinical trial comparing the efficacy of nimesulide, celecoxib and rofecoxib in osteoarthritis of the knee. Drugs. 2003;63 Suppl 1:37–46.
27. Lücker PW, Pawlowski C, Friedrich I, Faiella F, Magni E. Double-blind, randomised, multi-centre clinical study evaluating the efficacy and tolerability of nimesulide in comparison with etodalac in patients suffering from osteoarthritis of the knee. Eur J Rheumatol Inflamm. 1994;14(2):29–38.
28. Dreiser RL, Riebenfeld D. Nimesulide in the treatment of osteoarthritis. Double-blind studies in comparison with piroxicam, ketoprofen and placebo. Drugs. 1993;46 Suppl 1:191–5.
29. Santos BFE, Costa FO, Vasconcelos AMA, Cyrino RM, Cota LOM. Preemptive effects of ibuprofen and nimesulide on postoperative pain control after open flap periodontal surgeries: A randomized placebo-controlled split-mouth clinical trial. J Periodontol. 2022 Feb;93(2):298–307.
30. da Costa Araújo FA, de Santana Santos T, de Morais HH, Laureano Filho JR, de Oliveira E Silva ED, Vasconcellos RJ. Comparative analysis of preemptive analgesic effect of tramadol chlorhydrate and nimesulide following third molar surgery. J Craniomaxillofac Surg. 2012 Dec;40(8):e346–9.
31. Bocanegra M, Seijas A, González Yibirín M. Effectiveness and tolerability of once-daily nimesulide versus ibuprofen in pain management after surgical extraction of an impacted third molar: A 24-hour, double-blind, randomized, double-dummy, parallel-group study. Curr Ther Res Clin Exp. 2005 May;66(3):172–80.
32. Bracco P, Debernardi C, Coscia D, Pasqualini D, Pasqualicchio F, Calabrese N. Efficacy of rofecoxib and nimesulide in controlling postextraction pain in oral surgery: a randomised comparative study. Curr Med Res Opin. 2004 Jan;20(1):107–12.
33. Kılıç E, Mızrak A, Göksu S, Cesur M. Fonksiyonel endoskopik sinüs cerrahisinde gabapentin ve nimesulidin preemptif analjezik etkinliği [Preemptive analgesic efficacy of gabapentin and nimesulide in the functional endoscopic sinus surgery]. Agri. 2014;26(2):73–81. Turkish.
34. Aho M, Kokki H, Nikanne E. Nimesulide versus Ibuprofen for Postoperative Tonsillectomy Pain : A Double-Blind, Randomised, Active Comparator-Controlled Clinical Trial. Clin Drug Investig. 2003;23(10):651–60.
35. Ragot JP, Monti T, Macciocchi A. Controlled clinical investigation of acute analgesic activity of nimesulide in pain after oral surgery. Drugs. 1993;46 Suppl 1:162–7.
36. Chaudhary B, Gurung D, Dahal S, Twyana R, Khanal P, Adhikari P. Comparison of nimesulide and ketorolac in control of pain, swelling, and trismus following extraction of impacted third molar. J Nepal Dent Assoc. 2020 Jul;20(31):79–87.
37. Pohjolainen T, Jekunen A, Autio L, Vuorela H. Treatment of acute low back pain with the COX-2-selective anti-inflammatory drug nimesulide: results of a randomized, double-blind comparative trial versus ibuprofen. Spine (Phila Pa 1976). 2000 Jun 15;25(12):1579–85.
38. Dhole DS, Darbastwar DS, Mendhe DC, Shirbhate DA, Gondane DA, Pawar DD, et al. A Multicenter, Retrospective, Real-World Evidence Study to Evaluate the Effectiveness and Safety of Nimesulide-Paracetamol Combination Therapy for the Management of Acute Painful Conditions. Annals of Medicine and Medical Sciences. 2025 Aug 16;923–7.
39. Samartsev IN, Zhivolupov SA, Parshin MS, Magomedov KR. Otkrytoe nablyudatel'noe issledovanie s retrospektivnym analizom effektivnosti i perenosimosti atseklofenaka v lechenii patsientov s ostroi nespetsificheskoi skeletno-myshechnoi bol'yu v nizhnei chasti spiny [Open observational study with a retrospective analysis of the efficacy and tolerability of aceclofenac in patients with acute non-specific low back pain]. Zh Nevrol Psikhiatr Im S S Korsakova. 2024;124(12):99–105. Russian.
40. Marini U, Spotti D. Gastric tolerability of nimesulide. A double-blind comparison of 2 oral dosage regimens and placebo. Drugs. 1993;46 Suppl 1:249–52.
41. Scheinberg M, Pott Júnior H, Macêdo EA, Bocchi de Oliveira MF, Ecclissato C, Amazonas RB. Efficacy and safety of a fixed-dose combination of nimesulide/pantoprazole compared to naproxen/esomeprazole for pain relief in patients with osteoarticular diseases and dyspeptic symptoms. Drug Des Devel Ther. 2018 Sep 6;12:2775–83.
42. Pilotto A, Franceschi M, Leandro G, Paris F, Niro V, Longo MG, et al. The risk of upper gastrointestinal bleeding in elderly users of aspirin and other non-steroidal anti-inflammatory drugs: the role of gastroprotective drugs. Aging Clin Exp Res. 2003 Dec;15(6):494–9.
43. Cunietti E, Monti M, Viganò A, D'Aprile E, Saligari A, Scafuro E, et al. A comparison of nimesulide vs paracetamol in the treatment of pyrexia in the elderly. Drugs. 1993;46 Suppl 1:124–6.
44. Ashraf SM, Shejole VS, Kadam NR, Chandrappa AM, Toke SK, Yadagiri SK, et al. Prospective Real-World Study Comparing the Safety and Effectiveness of Nimesulide with Available Antipyretic and Analgesics for Treatment of Fever or Fever with Pain—ENDEVER. The Journal of the Association of Physicians of India. 2025 Jul;73(7):e1-6.
45. Calligaris A, Scaricabarozzi I, Vecchiet L. A multicentre double-blind investigation comparing nimesulide and naproxen in the treatment of minor sport injuries. Drugs. 1993;46 Suppl 1:187–90.
46. Bavbek S, Celik G, Ozer F, Mungan D, Misirligil Z. Safety of selective COX-2 inhibitors in aspirin/nonsteroidal anti-inflammatory drug-intolerant patients: comparison of nimesulide, meloxicam, and rofecoxib. J Asthma. 2004 Feb;41(1):67–75.
47. Monteiro C, Silvestre S, Duarte AP, Alves G. Safety of Non-Steroidal Anti-Inflammatory Drugs in the Elderly: An Analysis of Published Literature and Reports Sent to the Portuguese Pharmacovigilance System. Int J Environ Res Public Health. 2022 Mar 16;19(6):3541.
48. Kim J, Lee J, Shin CM, Lee DH, Park BJ. Risk of gastrointestinal bleeding and cardiovascular events due to NSAIDs in the diabetic elderly population. BMJ Open Diabetes Res Care. 2015 Dec 18;3(1):e000133.
49. Conforti A, Leone R, Moretti U, Mozzo F, Velo G. Adverse drug reactions related to the use of NSAIDs with a focus on nimesulide: results of spontaneous reporting from a Northern Italian area. Drug Saf. 2001;24(14):1081–90.
50. National Institute of Diabetes and Digestive and Kidney Diseases. Nimesulide. In: LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012[cited 2025 Sep 17]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK547948/.
51. Lucas GNC, Leitão ACC, Alencar RL, Xavier RMF, Daher EF, Silva Junior GBD. Pathophysiological aspects of nephropathy caused by non-steroidal anti-inflammatory drugs. J Bras Nefrol. 2019 Jan-Mar;41(1):124–30.
52. Ikdahl E, Kerola A, Sollerud E, Semb AG. Cardiovascular Implications of Non-steroidal Anti-inflammatory Drugs: A Comprehensive Review, with Emphasis on Patients with Rheumatoid Arthritis. Eur Cardiol. 2024 Dec 23;19:e27.
53. Ghlichloo I, Gerriets V. Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). In: StatPearls. Treasure Island (FL): StatPearls Publishing; [cited 2025 Sep 18]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK547742/.
54. Bonadonna P, Olivieri F, Jarkvist J, Nalin F, Zanotti R, Maclachlan L, et al. Non-steroidal anti-inflammatory drug-induced anaphylaxis infrequent in 388 patients with mastocytosis: A two-center retrospective cohort study. Front Allergy. 2022 Dec 5;3:1071807.
55. Dhopeshwarkar N, Sheikh A, Doan R, Topaz M, Bates DW, Blumenthal KG, et al. Drug-Induced Anaphylaxis Documented in Electronic Health Records. J Allergy Clin Immunol Pract. 2019 Jan;7(1):103–11.
56. Sen R, Hurley JA. Osteoarthritis. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025[cited 2025 Dec 12]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK482326/.
57. Casiano VE, Sarwan G, Dydyk AM, Varacallo MA. Back Pain. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025[cited 2025 Dec 12]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK538173/.
58. Kearney PM, Baigent C, Godwin J, Halls H, Emberson JR, Patrono C. Do selective cyclo-oxygenase-2 inhibitors and traditional non-steroidal anti-inflammatory drugs increase the risk of atherothrombosis? Meta-analysis of randomised trials. BMJ. 2006 Jun 3;332(7553):1302–8.
59. Coxib and traditional NSAID Trialists' (CNT) Collaboration; Bhala N, Emberson J, Merhi A, Abramson S, Arber N, et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomised trials. Lancet. 2013 Aug 31;382(9894):769–79.
60. FDA. FDA Drug Safety Communication: FDA strengthens warning that non-aspirin nonsteroidal anti-inflammatory drugs (NSAIDs) can cause heart attacks or strokes. Montgomery : FDA; 2023 Aug 11 [cited 2025 Sep 23]. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-strengthens-warning-non-aspirin-nonsteroidal-anti-inflammatory.
