Abstract
Schizophrenia is a complex, multifactorial neuropsychiatric disorder in which many patients continue to experience disabling negative and cognitive symptoms despite treatment with dopamine-based antipsychotics. This therapeutic limitation has intensified the search for alternative neurobiological pathways that more directly regulate stress, affective processing, and salience. Increasing translational evidence indicates the dynorphin–kappa opioid receptor (DYN–KOR) pathway as a vital molecular regulator within cortico–striatal–limbic circuits that are impaired in schizophrenia. Dynorphins, derived from the prodynorphin precursor, serve as endogenous ligands for kappa opioid receptors (KORs) situated in the mesolimbic and prefrontal networks. The activation of this system affects multiple neurotransmitter pathways, including dopaminergic, glutamatergic, and GABAergic transmission. Kappa opioid receptor stimulation at the intracellular level activates stress-responsive pathways, including c-Jun N-terminal kinase signaling, resulting in changes to synaptic plasticity, motivation, and cognitive integration. These molecular effects parallel the behavioral domains most resistant to current pharmacotherapy. In humans, selective KOR agonists consistently induce perceptual abnormalities, dysphoria, and cognitive impairment, closely mirroring essential characteristics of psychosis. The effects are swiftly counteracted by opioid antagonists, validating a receptor-specific mechanism. Clinical biomarker studies indicate altered dynorphin levels in the cerebrospinal fluid of individuals with schizophrenia, with heightened concentrations associated with greater symptom severity and poor long-term outcomes. In accordance with these findings, animal models demonstrate that KOR activation leads to anhedonia, aversion, and sensory-gating deficits, while pharmaceutical KOR inhibition reinstates reward sensitivity and cognitive function. The combined genetic, behavioral, and clinical data support a revised neuropharmacological framework in which dysregulated DYN–KOR signaling contributes to the pathogenesis of schizophrenia. Focusing on this system may provide a promising therapeutic approach for symptom domains that current medications poorly address.
Keywords
Dynorphin, Kappa opioid receptor, Schizophrenia, Neurotransmitter dysregulation, Translational neuropharmacology
Conflicts of Interest
The author declares no conflicts of interest. The author has no funding sources available for this study.
Introduction
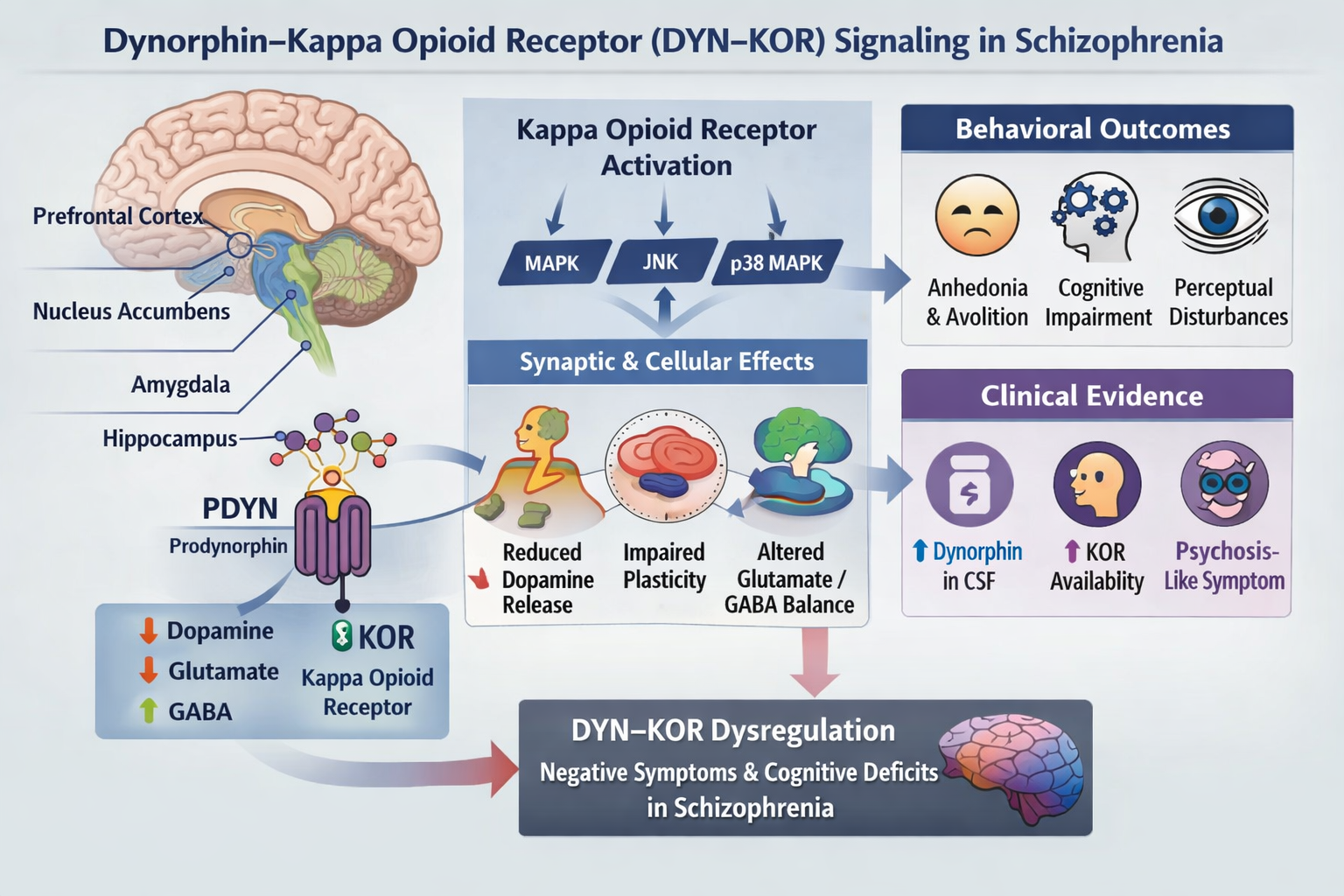
Schizophrenia is a severe and heterogeneous neuropsychiatric disorder characterized by positive symptoms, negative symptoms, and significant cognitive deficits, resulting in profound functional impairment [1]. Although dopamine D2 receptor antagonists remain the foundation of antipsychotic therapy, their therapeutic efficacy is largely confined to the reduction of positive symptoms such as hallucinations and delusions [1]. In contrast, negative symptoms including anhedonia, avolition, and social withdrawal as well as impairments in working memory, executive functioning, and cognitive flexibility frequently persist despite adequate dopaminergic blockade. These treatment-resistant areas are now seen as key factors influencing long-term outcomes, psychosocial functioning, and quality of life in schizophrenia [2–4]. The limited effects of current antipsychotics on motivation and cognition have increased efforts to find alternative neurobiological systems that operate upstream of, or alongside, dopaminergic pathways. These systems more directly impact stress response, salience attribution, and emotional motivation [1–2]. The dynorphin–kappa opioid receptor (DYN–KOR) system has gained attention as a promising candidate because of its strategic position within cortico–striatal–limbic circuits, which are often linked to schizophrenia's pathology [5]. Dynorphins, derived from the prodynorphin precursor, serve as natural ligands for kappa opioid receptors. They influence key brain regions that control reward, emotion, and cognition. KORs are highly expressed in the prefrontal cortex, nucleus accumbens, amygdala, and hippocampus, where they help regulate dopamine levels, modulate glutamate transmission, and maintain GABAergic inhibition [5–7]. Activating the DYN–KOR system affects large-scale brain networks more than individual synapses, altering cortico-striatal interactions and motivation levels. Notably, dynorphin production is sensitive to stress, and repeated psychosocial stressors increase DYN–KOR activity, making this system a link between environmental stress and lasting neurobiological changes [8,9]. Evidence from human studies, molecular imaging, cerebrospinal fluid biomarkers, and animal models supports the idea that abnormal DYN–KOR signaling might lead to psychosis-like symptoms, motivational issues, and cognitive problems seen in schizophrenia [5,10]. For instance, activating kappa opioid receptors in humans reliably causes feelings of dysphoria, depersonalization, perceptual distortions, and cognitive disruptions—symptoms overlapping with psychosis [11,12]. More recent PET scans show that KOR availability correlates with the severity of anhedonia, directly linking DYN–KOR activity to negative symptoms in schizophrenia [13]. Earlier biomarker studies also found altered cerebrospinal fluid dynorphin levels in schizophrenia patients, indicating possible dysregulation of this neuropeptide system [14]. Overall, these findings suggest that abnormal DYN–KOR signaling is not just a consequence of dopaminergic imbalance but a key neurobiological process driving stress sensitivity, misattribution of salience, and persistent motivational deficits in schizophrenia.
Dynorphin–Kappa Opioid Receptor Signaling and Psychotomimetic Mechanisms
The DYN–KOR system is particularly important for modulating neuronal activity associated with stress and for its role in cortico–striatal–limbic circuits. Most kappa opioid receptors are found in the prefrontal cortex, nucleus accumbens, amygdala, hippocampus, and ventral pallidum. Activating KOR in cells initiates stress-related signaling pathways, notably MAPK, which influence synaptic strength, dendritic structure, and long-term plasticity in the prefrontal and limbic regions of the brain [9,15,16]. Recent work further demonstrates that stress-induced dynorphin release drives p38 MAPK–dependent synaptic adaptations that persistently alter motivational circuits [17]. These molecular and cellular alterations underpin enduring motivational and cognitive impairments. Activation of the kappa-opioid receptor (KOR) significantly decreases dopamine release in mesolimbic pathways by reducing the firing of dopaminergic neurons and presynaptic dopamine release in the nucleus accumbens. Contemporary circuit-mapping studies using optogenetic and chemogenetic approaches show that KOR activation suppresses phasic dopamine signaling and shifts encoding from reward to aversion within ventral striatal circuits [6,18]. More recent electrophysiological studies confirm that KOR activation alters excitatory–inhibitory balance within cortico-striatal pathways, thereby impairing behavioral flexibility and adaptive reward learning [8]. This dopaminergic inhibition is associated with parallel alterations in glutamatergic and GABAergic transmission, which disrupt reward responsiveness, degrade signal-to-noise processing, and reduce cognitive flexibility [6,8]. By reshaping cortico-striatal communication and reducing dopaminergic gain, excessive DYN–KOR signaling may contribute to aberrant salience attribution and motivational deficits central to schizophrenia [5,13]. Together, these intracellular and circuit-level effects indicate that excessive DYN–KOR activation integrates stress signaling with reward circuit suppression, providing a mechanistic bridge between molecular signaling events and the behavioral abnormalities characteristic of schizophrenia. Research has shown that activating KOR directly can induce psychosis-like effects. Administering selective KOR agonists consistently induces feelings of dysphoria, perceptual changes, depersonalization, and mental disturbances in healthy individuals. These symptoms are similar to those observed in psychosis and negative emotions. The quick reversal of these effects with opioid antagonists confirms they are specifically caused by KOR activation rather than general neurotoxicity [11]. Overall, the data support a model where abnormal DYN–KOR signaling triggers harmful stress responses, disrupts salience processing, and promotes behaviors linked to psychosis. This underscores the importance of this system in the neurobiology of schizophrenia.
Circuit Mechanisms: Mapping DYN–KOR Signaling onto Schizophrenia Symptom Domains
Within the mesolimbic reward system especially in the ventral striatum and nucleus accumbens dynorphin–kappa opioid receptor (DYN–KOR) signaling plays a key role in regulating motivational salience and how rewards are valued [6,19]. Usually, brief activation of KOR acts as a homeostatic brake, helping to prevent excessive dopamine release during stress or highly salient events [19,20]. But long-term stimulation makes the system more likely to learn things that are bad for it and less likely to respond to rewards [21,22]. This change reduces the drive to work for rewards, weakens the signals that tell you when you could get a reward, and lowers the pleasure you feel when you think about getting one [6,23]. In schizophrenia, these changes may show up in the clinic as avolition, less goal-directed behavior, and trouble making decisions based on effort [24,25]. Recent molecular imaging findings illustrate a correlation between KOR availability and the severity of anhedonia in schizophrenia offer direct in vivo validation for the role of this system in exacerbating negative symptomatology [13]. These results support the hypothesis that mesolimbic dopamine dysfunction in schizophrenia may be modulated by upstream neuropeptide control rather than solely by inherent dopaminergic pathology [6]. In addition to affecting reward pathways, DYN–KOR signaling also modulates cortico-striatal glutamatergic transmission and local inhibitory processes, thereby influencing higher cognitive functions [26,27]. Activation of KOR changes glutamate release in presynaptic neurons, impacting the excitation-inhibition balance in the prefrontal cortex [6,9]. These changes can disturb the cortex's signal-to-noise ratio, impairing working memory and reducing cognitive flexibility [28,29]. Disruptions in cortical prediction signaling may also destabilize striatal gating systems, resulting in incorrect salience attribution, a key feature of psychosis [30,31]. This approach does not invalidate the dopamine theory of schizophrenia; rather, it contextualizes it within a broader neuromodulatory framework, in which stress-sensitive peptide systems actively modulate dopaminergic output and cortical integration [6,9].
The stress-responsive characteristics of dynorphin further associate the DYN–KOR axis with disease progression and susceptibility to relapse [9,21]. Stress exposure significantly increases dynorphin levels in limbic regions, and continuous activation of this pathway might create a feedback loop, leading to higher stress sensitivity and more dynorphin release [32,33]. This cycle can gradually diminish reward processing, enhance unpleasant learning, and hinder adaptive coping [6,22]. In individuals with schizophrenia, such mechanisms may intensify negative symptoms and cognitive deterioration, even when positive symptoms are pharmacologically managed [31,34]. Over time, constant activation of DYN–KOR may lead to functional decline, social withdrawal, and a higher risk of relapse when psychosocial stress is high. All of these circuit-level discoveries point to the DYN–KOR system as a dynamic regulator of reward, cognition, and stress adaptation. Its dysregulation provides a biologically coherent rationale for symptom domains that extends beyond traditional dopaminergic hyperactivity, underscoring the need to incorporate peptide-based neuromodulatory systems into modern schizophrenia models [6,9].
Evidence for DYN–KOR Dysregulation in Schizophrenia
An increasing amount of clinical and translational research suggests that dysregulated dynorphin–kappa opioid receptor (DYN–KOR) signaling plays a significant role in schizophrenia [6,35]. Early neurochemical studies showed changes in dynorphin A levels in the cerebrospinal fluid of individuals with schizophrenia, where higher levels correlated with more severe symptoms and poorer long-term outcomes [14]. These findings imply that elevated dynorphin levels may indicate prolonged activation of stress-related neuropeptide systems, rather than a temporary condition, potentially leading to lasting motivational and emotional issues [8,9]. Additionally, genetic research supports this connection by finding polymorphisms in the prodynorphin promoter region that are linked to a higher risk of schizophrenia in specific populations [36]. Schizophrenia is extremely polygenic, with variations potentially serving as modulatory risk factors that predispose individuals to increased dynorphin production or modified stress responsiveness, thereby interacting with environmental stresses throughout development [37]. This gene–environment convergence is consistent with models proposing exaggerated stress sensitivity as a core feature of the disorder [31]. More recently, molecular imaging studies have provided direct in vivo evidence implicating the KOR system in schizophrenia-related symptom dimensions. Positron emission tomography studies demonstrate a robust correlation between kappa opioid receptor occupancy and clinical phenotypes, particularly adverse sensations [38]. Research shows that increased KOR availability is linked to more severe anhedonia in people with schizophrenia, highlighting the link between this receptor system and challenges in reward processing and motivation [13]. These imaging results underscore a significant connection between biological signaling and behavioral problems.
Therapeutic Consequences and Prospective Pathways
Combining pharmacological, biomarker, genetic, and neuroimaging data highlights the dynorphin–kappa opioid receptor (DYN–KOR) axis as a promising target for treating schizophrenia, particularly for symptoms that do not respond well to dopamine-focused antipsychotics [6,13,35]. Meta-analyses suggest that opioid antagonists, when combined with other therapies, can decrease overall symptom severity, offering early clinical support for focusing on opioid signaling in schizophrenia [39,40]. These effects appear to extend beyond symptom relief, possibly enhancing emotional and motivational challenges [25,34]. Recent clinical trials of selective KOR antagonists for mood disorders have yielded mixed results; however, these outcomes should be evaluated in the context of trial design rather than as evidence against the biological relevance of the target [41,42]. While the potential of kappa-opioid receptor (KOR) antagonists as therapeutic agents is increasingly recognized, the existing clinical evidence presents certain constraints. Initially, the majority of clinical investigations into KOR antagonists have focused on populations with mood or substance-use disorders, rather than schizophrenia; this restricts the direct applicability of these findings to psychotic disorders [7,10]. Furthermore, the psychiatric cohorts involved in these trials frequently exhibit heterogeneity in their symptom profiles, which may mask potential therapeutic advantages for specific subgroups, especially those experiencing significant anhedonia or stress-related symptomatology [25,34]. Finally, early KOR antagonists, such as nor-binaltorphimine (nor-BNI) and analogous compounds, exhibited atypically extended pharmacokinetic characteristics and delayed receptor inactivation, thereby complicating dose optimization and safety evaluations within clinical contexts [7]. In addition, the absence of validated biomarkers capable of identifying individuals with elevated DYN–KOR activity has likely limited the ability of clinical trials to detect therapeutic effects. Emerging approaches such as kappa-opioid receptor molecular imaging and biomarker-guided stratification may therefore be important for improving. The presence of negative symptom burden, anhedonia, and stress sensitivity is not uniform across psychiatric populations, and failing to stratify patients based on these attributes may obscure treatment outcomes [24,43]. In schizophrenia, characterized by stress-reactive neurobiology and impairments in reward processing, KOR antagonism may be particularly pertinent for carefully selected subgroups rather than as a universal therapeutic [2,31]. Future therapeutic strategies may take a precision-focused approach by combining symptom profiles with biological markers of DYN–KOR activity [37]. Techniques such as kappa opioid receptor imaging, assessing dynorphin levels in peripheral or cerebrospinal fluid, and stress-response phenotyping can assist in identifying individuals who are most likely to benefit from therapies targeting KOR [13,41]. Combining KOR antagonists with stable antipsychotics may effectively address negative and cognitive symptoms while preserving dopaminergic control of psychosis [4,34]. Overall, utilizing biomarkers to inform adjustments in the DYN–KOR system could significantly decrease long-term functional impairments and enhance treatment outcomes in schizophrenia.
Conclusion
Dysregulated dynorphin–kappa opioid receptor signaling is a plausible and clinically relevant mechanism involved in the complex pathophysiology of schizophrenia. The DYN–KOR pathway connects neurobiological models of symptoms that are not well treated by dopamine-focused therapies, linking stress responses with changes in reward processing and higher cognitive functions. This system appears particularly relevant for managing unpleasant symptoms, anhedonia, and cognitive deficits—factors that markedly contribute to ongoing functional impairments and reduced quality of life. Evidence from human drug trials, biomarker studies, neuroimaging, and animal research indicates that abnormal DYN–KOR signaling is not merely a consequence of psychosis but may actively affect the disease's development and progression, particularly under chronic stress. These findings emphasize the importance of shifting away from uniform treatments and towards personalized therapies that address specific neurological vulnerabilities. Ongoing translational research—including studies on genetic biomarkers, receptor imaging, and symptom-specific clinical trials—is vital for understanding how KOR regulation could benefit people with schizophrenia. A precision medicine strategy should focus on identifying patient subgroups that are most likely to respond to targeted treatments. DYN–KOR-focused therapies have the potential to develop more effective drugs, particularly for cases where existing antipsychotics are less successful.
Author Contributions
Durairaj Ragu Varman-Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
Abbreviations
DYN–KOR: Dynorphin–Kappa Opioid Receptor; DYN: Dynorphin; KOR: Kappa Opioid Receptor; PDYN: Prodynorphin; MOR: Mu Opioid Receptor; D2R: Dopamine D2 Receptor
PFC: Prefrontal Cortex; NAc: Nucleus Accumbens; MAPK: Mitogen-Activated Protein Kinase; JNK: c-Jun N-terminal Kinase; p38 MAPK: p38 Mitogen-Activated Protein Kinase; PET: Positron Emission Tomography; CSF: Cerebrospinal Fluid; GABA: Gamma-Aminobutyric Acid
References
2. Howes OD, Murray RM. Schizophrenia: an integrated sociodevelopmental-cognitive model. Lancet. 2014 May 10;383(9929):1677–87.
3. Galderisi S, Mucci A, Buchanan RW, Arango C. Negative symptoms of schizophrenia: new developments and unanswered research questions. Lancet Psychiatry. 2018 Aug;5(8):664–77.
4. Correll CU, Schooler NR. Negative Symptoms in Schizophrenia: A Review and Clinical Guide for Recognition, Assessment, and Treatment. Neuropsychiatr Dis Treat. 2020 Feb 21;16:519–34
5. Clark SD, Abi-Dargham A. The Role of Dynorphin and the Kappa Opioid Receptor in the Symptomatology of Schizophrenia: A Review of the Evidence. Biol Psychiatry. 2019 Oct 1;86(7):502-11.
6. Tejeda HA, Bonci A. Dynorphin/kappa-opioid receptor control of dopamine dynamics: Implications for negative affective states and psychiatric disorders. Brain Res. 2019 Jun 15;1713:91-101.
7. Lobe MMM, Verma S, Patil VM, Iyer MR. A review of kappa opioid receptor antagonists and their clinical trial landscape. Eur J Med Chem. 2025 Apr 5;287:117205.
8. Crowley NA, Kash TL. Kappa opioid receptor signaling in the brain: Circuitry and implications for treatment. Prog Neuropsychopharmacol Biol Psychiatry. 2015 Oct 1;62:51–60.
9. Bruchas MR, Land BB, Chavkin C. The dynorphin/kappa opioid system as a modulator of stress-induced and pro-addictive behaviors. Brain Res. 2010 Feb 16;1314:44–55.
10. Jacobson ML, Browne CA, Lucki I. Kappa Opioid Receptor Antagonists as Potential Therapeutics for Stress-Related Disorders. Annu Rev Pharmacol Toxicol. 2020 Jan 6;60:615–36.
11. Pfeiffer A, Brantl V, Herz A, Emrich HM. Psychotomimesis mediated by kappa opiate receptors. Science. 1986 Aug 15;233(4765):774–6.
12. Walsh SL, Strain EC, Abreu ME, Bigelow GE. Enadoline, a selective kappa opioid agonist: comparison with butorphanol and hydromorphone in humans. Psychopharmacology (Berl). 2001 Sep;157(2):151–62.
13. Slifstein M, Qu W, Gil R, Weinstein JJ, Perlman G, Jaworski-Calara T, et al. Kappa opioid receptor availability predicts severity of anhedonia in schizophrenia. Neuropsychopharmacology. 2024 Dec;49(13):2087–93.
14. Heikkilä L, Rimón R, Terenius L. Dynorphin A and substance P in the cerebrospinal fluid of schizophrenic patients. Psychiatry Res. 1990 Dec;34(3):229–36.
15. Chavkin C, Koob GF. Dynorphin, Dysphoria, and Dependence: the Stress of Addiction. Neuropsychopharmacology. 2016 Jan;41(1):373–4.
16. Margolis EB. A switch in kappa opioid receptor signaling from inhibitory to excitatory induced by stress in a subset of cortically-projecting dopamine neurons. BioRxiv [Preprint]. 2025 Oct 3:2025.08.09.669424.
17. Quessy F, Bittar T, Blanchette LJ, Lévesque M, Labonté B. Stress-induced alterations of mesocortical and mesolimbic dopaminergic pathways. Sci Rep. 2021 May 26;11(1):11000.
18. Farahbakhsh ZZ, Siciliano CA. Kappa opioid receptor control of motivated behavior revisited. Neuropsychopharmacology. 2026 Jan;51(2):383–98.
19. Shippenberg TS. The dynorphin/kappa opioid receptor system: a new target for the treatment of addiction and affective disorders? Neuropsychopharmacology. 2009 Jan;34(1):247.
20. Spanagel R, Herz A, Shippenberg TS. Opposing tonically active endogenous opioid systems modulate the mesolimbic dopaminergic pathway. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2046–50.
21. Land BB, Bruchas MR, Lemos JC, Xu M, Melief EJ, Chavkin C. The dysphoric component of stress is encoded by activation of the dynorphin kappa-opioid system. J Neurosci. 2008 Jan 9;28(2):407–14.
22. Wee S, Koob GF. The role of the dynorphin-kappa opioid system in the reinforcing effects of drugs of abuse. Psychopharmacology (Berl). 2010 Jun;210(2):121–35.
23. Berridge KC, Robinson TE. What is the role of dopamine in reward: hedonic impact, reward learning, or incentive salience? Brain Res Brain Res Rev. 1998 Dec;28(3):309–69.
24. Gold JM, Waltz JA, Prentice KJ, Morris SE, Heerey EA. Reward processing in schizophrenia: a deficit in the representation of value. Schizophr Bull. 2008 Sep;34(5):835–47.
25. Strauss GP, Waltz JA, Gold JM. A review of reward processing and motivational impairment in schizophrenia. Schizophr Bull. 2014 Mar;40 Suppl 2(Suppl 2):S107–16.
26. Hjelmstad GO, Fields HL. Kappa opioid receptor activation in the nucleus accumbens inhibits glutamate and GABA release through different mechanisms. J Neurophysiol. 2003 May;89(5):2389–95.
27. Karkhanis A, Holleran KM, Jones SR. Dynorphin/Kappa Opioid Receptor Signaling in Preclinical Models of Alcohol, Drug, and Food Addiction. Int Rev Neurobiol. 2017;136:53–88.
28. Winterer G, Weinberger DR. Genes, dopamine and cortical signal-to-noise ratio in schizophrenia. Trends Neurosci. 2004 Nov;27(11):683–90.
29. Arnsten AF. Catecholamine influences on dorsolateral prefrontal cortical networks. Biol Psychiatry. 2011 Jun 15;69(12):e89–99.
30. Kapur S. Psychosis as a state of aberrant salience: a framework linking biology, phenomenology, and pharmacology in schizophrenia. Am J Psychiatry. 2003 Jan;160(1):13–23.
31. Grace AA. Dysregulation of the dopamine system in the pathophysiology of schizophrenia and depression. Nat Rev Neurosci. 2016 Aug;17(8):524–32.
32. Shirayama Y, Ishida H, Iwata M, Hazama GI, Kawahara R, Duman RS. Stress increases dynorphin immunoreactivity in limbic brain regions and dynorphin antagonism produces antidepressant-like effects. J Neurochem. 2004 Sep;90(5):1258–68.
33. McLaughlin JP, Marton-Popovici M, Chavkin C. Kappa opioid receptor antagonism and prodynorphin gene disruption block stress-induced behavioral responses. J Neurosci. 2003 Jul 2;23(13):5674-83.
34. Millan MJ, Fone K, Steckler T, Horan WP. Negative symptoms of schizophrenia: clinical characteristics, pathophysiological substrates, experimental models and prospects for improved treatment. Eur Neuropsychopharmacol. 2014 May;24(5):645–92.
35. Lutz PE, Kieffer BL. Opioid receptors: distinct roles in mood disorders. Trends Neurosci. 2013 Mar;36(3):195–206.
36. Zhang CS, Tan Z, Lu L, Wu SN, He Y, Gu NF, et al. Polymorphism of Prodynorphin promoter is associated with schizophrenia in Chinese population. Acta Pharmacol Sin. 2004 Aug;25(8):1022–6.
37. Trubetskoy V, Pardiñas AF, Qi T, Panagiotaropoulou G, Awasthi S, Bigdeli TB et al., Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature. 2022 Apr;604(7906):502–8.
38. Placzek MS. Imaging Kappa Opioid Receptors in the Living Brain with Positron Emission Tomography. Handb Exp Pharmacol. 2022;271:547–77.
39. Clark SD, Van Snellenberg JX, Lawson JM, Abi-Dargham A. Opioid antagonists are associated with a reduction in the symptoms of schizophrenia: a meta-analysis of controlled trials. Neuropsychopharmacology. 2020 Oct;45(11):1860-9.
40. Fond G, Mallet J, Urbach M, Benros ME, Berk M, Billeci M, et al., Adjunctive agents to antipsychotics in schizophrenia: a systematic umbrella review and recommendations for amino acids, hormonal therapies and anti-inflammatory drugs. BMJ Ment Health. 2023 Oct;26(1):e300771.
41. Naganawa M, Li S, Nabulsi N, Lin SF, Labaree D, Ropchan J, et al. Kinetic Modeling and Test-Retest Reproducibility of 11C-EKAP and 11C-FEKAP, Novel Agonist Radiotracers for PET Imaging of the κ-Opioid Receptor in Humans. J Nucl Med. 2020 Nov;61(11):1636–42.
42. Reed B, Butelman ER, Kreek MJ. Kappa Opioid Receptor Antagonists as Potential Therapeutics for Mood and Substance Use Disorders. Handb Exp Pharmacol. 2022;271:473–91.
43. Barch DM, Dowd EC. Goal representations and motivational drive in schizophrenia: the role of prefrontal-striatal interactions. Schizophr Bull. 2010 Sep;36(5):919–34.
