Abstract
Music listening and schizophrenia substantially involve emotion, subjective experience, motivation, and cognition in a complex manner. Despite these similarities, existing research appears to offer limited synthesis of their shared neuroscientific foundations. Accordingly, this narrative review integrates findings on the neural mechanisms underlying music listening and schizophrenia, based on analyses of peer-reviewed journal articles retrieved from PubMed, PsycINFO, and Nature Research databases, to specifically address their neurobiological parallels. In conclusion, the ventral tegmental area (VTA), nucleus accumbens (NAc), inferior frontal gyrus, motor areas, amygdala, orbitofrontal cortex, and cingulate cortex, encompassing dopaminergic reward circuitry, have been found to be the shared neural substrates implicated in both domains. Since previous intervention studies have widely discussed its clinical benefits across various disorders, music listening as an easily accessible and effective medium may potentially remain promising; nonetheless, technological and empirical limitations necessitate further investigation alongside methodological refinement, with particular emphasis on enhancing the consistency of findings.
Keywords
Schizophrenia, Music listening, Neuroscience, Musical reward, Neuroimaging, Symptomatology, Dopamine hypothesis
Introduction
Music is intrinsically linked to emotional experience [1]. Despite differences in the types of feelings they convey, some philosophers have argued music to be analogous to language because of its capacity to communicate emotions [2,3]. Proximate evidence could be found in neuroscience, as music listening engages neuroanatomical regions central to emotion, whereby pre-verbal emotion percepts arising from affective and sensory information may later be transferred into symbolic forms, such as language [4,5].
With advances in neuroscientific and clinical research, numerous studies on the relationship between music and emotion have yielded invaluable findings [6]. Rather than acting on isolated brain regions, music has been delineated to enhance functional connectivity across distributed neural networks responsible for affective memory and appraisal, particularly those interconnecting prefrontal regulatory control with limbic reactivity [6]. Applicable to both depression and anxiety, these effects are thought to be mediated through multiple biological pathways, including neurochemical modulation and immune-related inflammatory processes [6].
From a symptomatologic standpoint, mood and emotion have also been reported to be fundamental to schizophrenia [7]. Individuals with this acute psychiatric condition commonly exhibit a reduced capacity to express emotions outwardly, resulting in blunted facial and vocal responses even when exposed to emotionally salient stimuli [8,9]. These disturbances often extend to impairments in emotional identification, understanding, and regulation [10,11], as well as pronounced anhedonia and more negatively valenced responses to real-life experiences [12,13].
Historically, diagnostic concepts, classification criteria, and the clinical emphasis of schizophrenia have evolved over time [14,15]. This long-term shift has led to the view that it should primarily be conceptualized with positive and negative symptoms, considering the cognitive domain as a secondary catalog [16]. In concrete terms, positive symptoms include hallucinations and delusions, whereas negative symptoms entail deficits or loss of expression and motivation [17], cognitive symptoms, by contrast, are commonly described through cognitive deficits [18].
In light of recent debates beyond DSM-5 [19], hallucinations are referred to as internal, perception-like experiences that may be attenuated or phenomenologically distinct from normal perception but are nonetheless held with strong conviction as real and as originating from a non- self-generated source [19], while delusions are regarded as the presence of inaccurate beliefs that contradict available evidence [20,21]. Although they are phenomenologically distinct, music-induced subjective experiences may also elicit comparable alterations in the perception of reality [22,23]. When paired with film scenes, music alters viewers’ perceptions of a character’s emotional state [24,25], increases levels of motivation [26], and modulates cognitive performance, depending on the type of music, arousal level, task demands, and exposure duration [27].
Concurrently, previous research has highlighted the neuroplasticity-related benefits of rhythmic musical perception based on glutamatergic pathways [28], as well as the effectiveness of music-based interventions for schizophrenia [29–30]. Whilst much of the adjacent literature has tended to focus on clinical outcomes with limited neurobiological specificity, qualitative investigations into the shared substrates underlying these two domains appeared to remain largely invisible. To address this gap, this exploratory review synthesized the comparable principles of these phenomena, with the goal of delineating convergent mechanisms and suggesting a conceptual framework to guide future research at the intersection of music, neuroscience, and mental health research.
Methods
This exploratory study was informed by a broad narrative literature review conducted across PubMed, PsycINFO, and the Nature Research database; the search and analysis were performed between December 2025 and January 2026. A range of key terms, including but not limited to “music listening”, “musical reward”, “affective states”, “cognition”, “schizophrenia”, “dopamine hypothesis”, and “neuroimaging”, were employed (Table 1). In parallel, the search strategy was refined iteratively; in turn, keywords were modified as needed, allowing newly identified themes, theoretical frameworks, and influential studies to shape subsequent explorations.
|
Database |
Keywords |
|
PubMed |
Music Listening, Musical Reward, Schizophrenia, Dopamine Hypothesis, Neuroimaging, MRI, fMRI, EEG, EDA, PET scan, and HRV |
|
PsycINFO |
Music Listening, Background Music, Schizophrenia, Cognition, Positive Symptom, Negative Symptom, Cognitive Performance, and Affective States |
|
Nature Research |
Music Listening, Musical Reward, and Schizophrenia |
To limit the inclusion of outdated evidence, priority was given to peer-reviewed articles published in 2016 or later, and only English-language materials were considered. Earlier sources were included selectively when necessary for historical context or diagnostic or conceptual clarification, most notably those addressing the DSM-5, which was finalized in 2013. The APA Dictionary of Psychology was referenced as a supplementary source when reader-friendly definitions were essential for enhancing clarity.
Additionally, only peer-reviewed journal articles were inspected, excluding student papers, university lecture notes, unpublished manuscripts, conference abstracts, and academic dissertations; where appropriate, insights from systematic reviews were incorporated to contextualize findings and mitigate limitations inherent to narrative review methodology. Although studies published in widely cited and high-impact journals were preferentially considered, final inclusion decisions were guided primarily by methodological rigor, clarity of reporting, and relevance to the topic of this review. Articles raising concerns regarding scientific validity, reproducibility, or interpretive overreach, specifically those presenting biased interpretations or restrictive contents (e.g., mini reviews) were excluded irrespective of author affiliation, institutional context, or publisher.
Schizophrenia: An Overview
Schizophrenia is an intricate neuropsychiatric disorder that accompanies substantial functional disabilities ensuing from the interaction of a variety of risk factors [31]. American Psychology Association broadly characterizes it with difficulties in cognition, emotion, and behavior [32]. DSM-5 identifies positive symptoms; delusions, hallucinations, and disorganized speech or behavior (including catatonia), alongside negative symptoms, consisting of diminished emotional expression, alogia, or avolition, as the notable features of this condition [33,34].
When assessed using structural MRI, patients with schizophrenia exhibited lower gray matter volume (GMV) compared with healthy controls [35] (Table 2). Especially, reduced GMV in the medial prefrontal cortex, inferior frontal gyrus, and caudate nucleus positively correlated with performance on a rule-detection task [35] (Table 2). Task-based fMRI studies revealed decreased activation in the dorsomedial prefrontal cortex, supplementary motor area, and right inferior frontal gyrus during neurocognitive tasks, whereas the right angular gyrus reflected attenuated performance during social cognitive tasks [36] (Table 2).
|
Imaging Modality |
Implicated Brain Regions |
Observed Alteration |
Associated Functional Relevance |
|
Structural MRI |
Medial prefrontal cortex, inferior frontal gyrus, and caudate nucleus |
Reduced gray matter volume |
Cognitive stability and rule-detection performance |
|
Task-based fMRI (neurocognitive task) |
Dorsomedial prefrontal cortex, supplementary motor area, and right inferior frontal gyrus |
Decreased activation |
Neurocognitive Task Score |
|
Task-based fMRI (social cognitive task) |
Right angular gyrus |
Decreased activation |
Social Cognitive Task Score |
|
White-matter Integrity Analysis |
Superior longitudinal fasciculus II and fiber tracts (medial orbitofrontal cortex-rostral anterior cingulate cortex) |
Structural dysconnectivity |
Anhedonia-related deficits in reward perception and working memory |
Other than such structural measures, the dopamine hypothesis (DH), one of the most persistently emphasized concepts in psychiatry, has remained influential [37,38], with its historical development and evolution clearly articulated in multiple publications [37,38]. DH refers to an idea that proposes the role of dopaminergic dysregulation on schizophrenia [39]. Consistent with growing evidence of its significance, almost all licensed antipsychotic medications for the condition have developed to bind to dopamine receptors [40,41].
Antipsychotic drugs based on dopamine used in the treatment of schizophrenia act on the mesolimbic and mesocortical dopamine pathways, predominantly via D2 dopamine receptors [42]. These agents inhibit immoderate dopaminergic activity in the mesolimbic pathway; in contrast, enhancing insufficient dopaminergic activity in the mesocortical pathway [42].
Antagonism of dopamine signaling in the mesolimbic pathway induces a reduction in positive symptoms, whereas agonistic activity in the mesocortical pathway contributes to improvements in negative, affective, and cognitive symptoms, together reflecting partial agonist activity that may also function as partial antagonism [42].
Both the mesolimbic and mesocortical systems are defined as dopaminergic neural networks [43–45]; however, the mesolimbic pathway primarily targets the nucleus accumbens and related limbic structures, whereas the mesocortical pathway innervates the medial prefrontal cortex and anterior cingulate cortex [43–45]. Dopaminergic signaling to both systems originates in the ventral tegmental area (VTA), with projections traveling through the NAc, amygdala, and hippocampus within the mesolimbic system and through the cortex within the mesocortical system [46,47]. To a great extent, the mesolimbic pathway modulates reward and motivation, whereas the mesocortical pathway regulates cognition and executive function [46,47] (Figure 1).
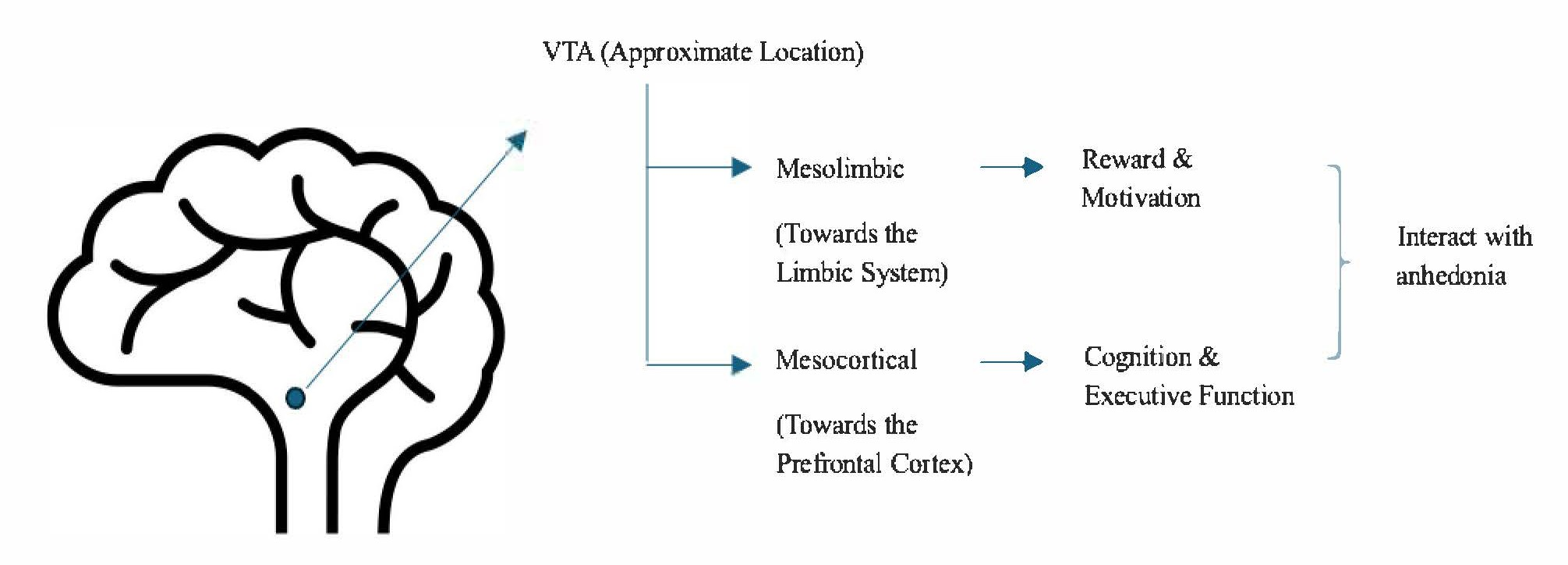
Figure 1. Schematic illustration of dopaminergic pathways implicated in schizophrenia. This figure depicts the principal dopaminergic circuits implicated in schizophrenia, with projections originating from the ventral tegmental area (VTA) and diverging into the mesolimbic and mesocortical pathways. Broadly, the mesolimbic pathway is associated with reward processing and motivation, whereas the mesocortical pathway supports cognition and executive functioning. Dysregulation across both pathways is thought to contribute to anhedonia.
In many instances, dopaminergic issues in schizophrenia do not appear to manifest as an isolated abnormality confined to a single pathway [48]. For instance, anhedonia involves deficits in reward processing, reflecting dysfunction in both the mesocortical and mesolimbic circuits, despite being classified as part of the negative symptoms [48] (Figure 1). Attenuating the subjective perception of reward and working memory, this complex symptom could be driven by various neurobiological abnormalities, such as structural dysconnectivity of the superior longitudinal fasciculus II within the frontoparietal white matter tract and fiber tracts linking the medial orbitofrontal cortex and the rostral anterior cingulate cortex; however, decreased integrity of the limbic system and the reward circuit has been found to exert the greatest impact [48] (Table 2).
Not only pharmacological approaches but also psychological therapies play an important role in reducing the symptoms of schizophrenia [49,50]. Prior fMRI studies involving patients with schizophrenia have found that successful cognitive remediation therapy may increase the activation in multiple neurobiological regions associated with memory; furthermore, cognitive behavioral therapy may improve amygdala connectivity that correlates with improvements in positive symptoms [49]. In addition, memory training, digital motivation support, reasoning training, psychoeducation, and virtual reality interventions have also demonstrated effectiveness in individuals with the disorder [50].
Although existing studies with neuroimaging techniques have provided indispensable insights, significant limitations remain regarding the use of fMRI as either a predictive or an indicative source for schizophrenia [51]. Technologically, the temporal resolution of fMRI is substantially slower than the speed of neural responses, and it cannot directly capture neurotransmitter activity [51]. Moreover, insufficient sample sizes have often raised empirical concerns regarding the generalizability of findings [51].
In essence, schizophrenia incorporates sophisticated symptoms and neurobiological structures. In spite of the fact that positive, negative, and cognitive symptoms are well-recognized in the field [33,34], researchers continue to debate the limitations of the DSM-5 and advocate for the refinement of diagnostic definitions to enhance precision [19]. Alterations in the GMV, along with the theoretical contribution of DH and mesolimbic and mesocortical pathways, predominantly govern contemporary neurobiological understandings of the disorder.
Accordingly, dopaminergic modulation seems crucial in the treatment of schizophrenia [41]. Diverse psychological therapeutic approaches are employed to alleviate symptoms; nowadays, increasingly applying virtual reality-based techniques. While much of the existing literature presents neuroimaging evidence, distinct methodological limitations warrant further investigations.
Music Listening: An Overview
Prior interdisciplinary research integrating music and neuroscience has documented multitudinous benefits of music-based interventions (e.g., music listening, music production, and musical training) for emotional regulation, cognitive function, and motor skills, in parallel with their competence to promote long-term neuroplasticity and enhance quality of life [52]. A range of neuroscientific and physiological measures; not limited to positron emission tomography (PET) [53], heart rate variability (HRV) [54,55], electroencephalogram (EEG) [56,57], electrodermal activity (EDA) [58,59], and functional MRI [60,61], have been applied to explore the psychological effects of music listening in human subjects. Collectively, these extensive approaches have provided converging evidence for the robust and measurable impact of music listening on various neural and physiological processes.
Menon and Levitin pioneered one of the earliest approaches employing functional and effective connectivity analyses with fMRI to examine neural responses to music listening, demonstrating links with major neurobiological regions that are implicated in reward processing [62,63]. Despite a substantial body of subsequent research, the principles of musical pleasure and reward have yet to be fully characterized, in part due to challenges in isolating acoustic features, responses, cultural factors, and interactions [64]. More recent work has highlighted the influence of music listening on neurochemical systems, including prolactin, oxytocin, and dopamine, as well as its capacity to modulate the mesolimbic reward circuit and the mesocorticolimbic pathway, with additional downstream effects on cognitive control, affective state, and motivation [64].
Vuust et al. presented a comprehensive synthesis of the neuroanatomical regions central to music listening, demonstrating that music-induced emotion recruits the ventral tegmental area (VTA), nucleus accumbens (NAc), amygdala, orbitofrontal cortex, insula, cingulate cortex, hypothalamus, periaqueductal gray, and ventral pallidum; in contrast, sensorimotor coupling during music perception engages the motor and premotor cortices, inferior frontal gyrus, basal ganglia, and cerebellum [65] (Figure 2). Neural activity within the motor cortices, dorsal striatum, and cerebellum has been regarded to support rhythm perception regardless of the presence or absence of overt movement, suggesting potential applications for music-based interventions in Parkinson’s disease, particularly in enhancing mood, motivation, and emotional well-being [66] (Figure 2). Similarly, musical reward, operating through neural mechanisms largely overlapping with the traditional reward circuitry, has been recognized to be applicable for mental health rehabilitation across a wide range of reward-related disorders [66] (Figure 2).
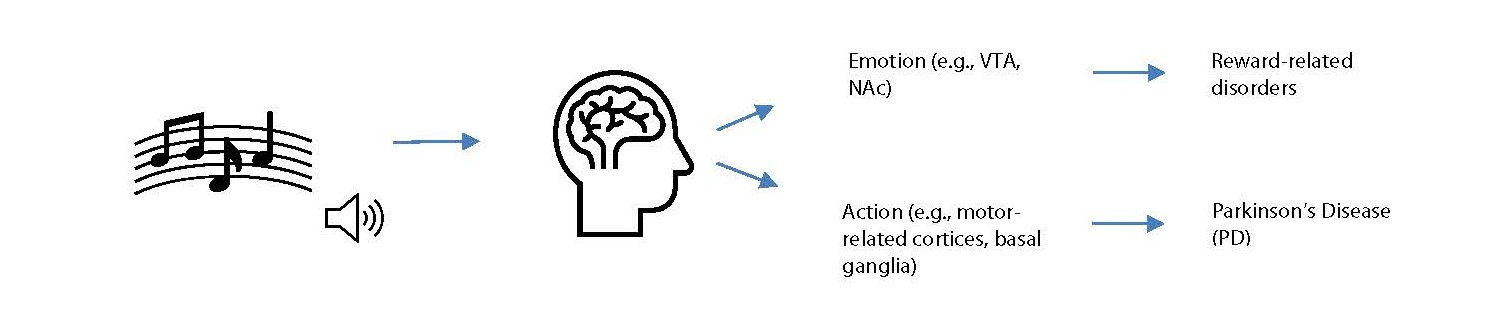
Figure 2. Music listening, brain responses, and clinical implications. This figure illustrates neuroanatomical regions responsive to music listening, with corresponding clinical interventions indicated on the right. Music listening engages distributed brain regions that could be generalized into “emotion” and “action.” Music-induced neural responses within “emotion” are thought to be applicable to reward-related disorders, whereas responses in “action” are considered to have implications for Parkinson’s disease (PD).
As for cognitive benefits and drawbacks, behavioral research has played a significant role in elucidating how background music is used and perceived in relation to the cognitive outcomes [67,68]. Unpleasant background music (BGM) tends to distract cognitive performance, largely due to its excessive control over mood and arousal; conversely, the presence of a BGM that enhances affective state has been shown to facilitate a positive aftereffect [67,68]. These findings have indicated that the relationship between music listening and cognitive performance depends on attention, mood, and arousal [67,68] (Table 3).
|
Music Listening (BGM) |
Effects on Mood and Arousal |
Effects on Cognitive Performance |
|
Unpleasant Background Music |
Distracts mood and arousal |
Attenuates cognitive performance |
|
Beneficial Background Music |
Enhances mood and arousal |
Enhances cognitive performance |
In tandem with subsequent research in cognition, recognition of music-based clinical interventions in the treatment of cognitive disorders has grown rapidly [69]. Music listening has been shown to support cognitive recovery after stroke, with evidence suggesting advantages over other auditory stimuli depending on the type of music and comparator condition [70,71]. However, challenges remain in establishing music as a consistently effective intervention for cognitive deficits, as systematic review has reported little or no effect on cognitive outcomes [72].
In sum, music listening has been investigated via an expensive array of neuroscientific and physiological techniques and has also been adopted to examine its clinical relevance, particularly in reward-related psychopathological conditions and Parkinson’s disease. Brain structures associated with motivation, mental processing, and motor function have shown sensitivity to music. Additionally, prolactin, oxytocin, and dopamine have been linked to listening experiences [64], with dopaminergic responses identified within the mesolimbic reward circuit and mesocorticolimbic pathway.
The outcomes of music listening with respect to the cognitive performance seemed contingent upon its capacity to modulate affective state [67,68]. Sustained exposure has been demonstrated to be effective for clinical recovery [70,71]; nonetheless, variability arising from individual differences; for instance, prior listening history and cultural background, and acoustic features has led to inconsistencies in empirical findings [64,72]. These findings imply that music listening may remain as a promising, but not yet fully delineated contributor to the positive consequences of mental health and well-being, rather than an infallible substitute for established therapeutic approaches.
Discussion
Superficially, music listening and schizophrenia appear to share diverse neuroscientific commonalities, as they both accompany the VTA-NAc-mediated dopaminergic reward circuitry entailing mesolimbic and mesocortical pathways. Other neuroanatomical regions, encompassing the inferior frontal gyrus, motor areas, amygdala, orbitofrontal cortex, and cingulate cortex have also behaved importantly for both contexts to a great extent. These overlapping aspects may consequently support the underlying principles of clinical interventions employing music listening to treat patients with schizophrenia.
Nevertheless, there remains substantial uncertainty and several limitations within research on music listening and schizophrenia that necessitate further investigation. Foremost, the molecular-level causal relationships between music listening and brain function remain underexplored [52]. More sophisticated clarification of these mechanisms could be essential for justifying the clinical application of music-based interventions, as dopamine alone does not account for the neurobiological complexity of schizophrenia, which also involves glutamatergic and GABAergic dysfunction and their interactions [40,73–75].
Secondly, in light of this review, current research on music listening vastly neglect the potential interference of audio resolution. Since there is a small but statistically significant potential for perceptibility [76,77], emerging insights into audio resolution may help clarifying its precise impact on affective states and clinical applications. With refinement of all these limitations, the potential of music listening; an affordable, prevalent, and easily accessible medium, may remain significant for mental health research, particularly considering the current findings on the neuroscientific parallels between music listening and schizophrenia.
However, several limitations temper these scholarly interpretations. Because this work has adopted a narrative and integrative approach, the studies that have been reviewed vary widely in design, measurement, and analytic focus, making it difficult to derive firm mechanistic conclusion. Much of the existing evidence remains associative rather than causal, and a heavy emphasis on dopamine-related explanations may overlook the broader neurochemical complexity that characterizes schizophrenia.
There are also practical and methodological constraints to consider. Neuroimaging tools and sample characteristics often limit the precision and generalizability of findings, and individuals differ substantially in how they perceive, value, and respond to music based on personal, cultural, acoustical, and clinical factors. Moving forward, carefully designed longitudinal and multimodal studies, paired with well-controlled musical stimuli and clinically meaningful outcomes, could particularly be valuable for clarifying when and how music listening may serve as a useful complementary approach; in addition, further qualitative research employing systematic methods and narrower scope may also support the advancement of this line of scholarship.
Conclusion
In summary, this review highlights meaningful overlap between how the brain responds to music and the neural systems disrupted in schizophrenia, particularly those involved in reward, motivation, and emotional experience. The shared involvement of midbrain and cortical networks linked to pleasure, salience, and action supports the idea that music may engage circuits that are often functionally altered in the disorder. These intersections help explain why music listening may be highly relevant to schizophrenia, especially in relation to everyday challenges such as reduced motivation, blunted affect, or difficulties with emotional engagement.
Conflicts of Interest
The author declares no conflict of interest.
Funding Statement
This research received no external funding or financial support.
Acknowledgement
The author would like to acknowledge the supportive academic environment and collegial feedback from the recording arts community during the MSRA 5000 course at the University of Colorado Denver, which were instrumental in shaping the ideas explored in this review. All figures and tables were created by the author and were not reproduced from any external sources; the figures were designed using default icons available in Microsoft Word.
References
2. Clark A. Is music a language? Jour of Aesth and Art Critic. 1982 Dec 1;41(2):195–204.
3. Guter E. Musical Expression: From Language to Music and Back. Philosophies. 2025 Jan 14;10(1):9.
4. Koelsch S. Brain correlates of music-evoked emotions. Nat Rev Neurosci. 2014 Mar;15(3):170–80.
5. Proverbio AM, Russo F. Multimodal recognition of emotions in music and language. Psychology of Music. 2022 Jan;50(1):54–68.
6. Dan Y, Xiong Y, Xu D, Wang Y, Yin M, Sun P, et al. Potential common targets of music therapy intervention in neuropsychiatric disorders: the prefrontal cortex-hippocampus -amygdala circuit (a review). Front Hum Neurosci. 2025 Feb 3; 19:1471433.
7. Wang Y, Xu Y, Wu P, Zhou Y, Zhang H, Li Z, et al. Exploring the interplay between core and mood symptoms in schizophrenia: a network analysis. Schizophr Res. 2024; 269:28–35.
8. Kring AM, Moran EK. Emotional response deficits in schizophrenia: insights from affective science. Schizophr Bull. 2008 Sep;34(5):819–34.
9. Zhao W, Zhang Q, Gao L, Fan N, Yun Y, Song J, et al. A network analysis of facial and vocal emotion recognition deficits in schizophrenia. Front Psychiatry. 2025 May 23; 16:1598026.
10. Kee KS, Horan WP, Salovey P, Kern RS, Sergi MJ, Fiske AP, et al. Emotional intelligence in schizophrenia. Schizophr Res. 2009 Jan;107(1):61–8.
11. Raugh IM, Strauss GP. Deconstructing emotion regulation in schizophrenia: the nature and consequences of abnormalities at the identification stage. Eur Arch Psychiatry Clin Neurosci. 2022 Sep;272(6):1061–1071.
12. Trémeau F. A review of emotion deficits in schizophrenia. Dialogues Clin Neurosci. 2006;8(1):59–70.
13. Krzyzanowski DJ, Wu S, Carnovale M, Agarwal SM, Remington G, Goghari V. Trait Anhedonia in Schizophrenia: A Systematic Review and Comparative Meta-analysis. Schizophr Bull. 2022 Mar 1;48(2):335–346.
14. Andreasen NC, Flaum M. Schizophrenia: the characteristic symptoms. Schizophr Bull. 1991;17(1):27–49.
15. Orsolini L, Pompili S, Volpe U. Schizophrenia: A Narrative Review of Etiopathogenetic, Diagnostic and Treatment Aspects. J Clin Med. 2022 Aug 27;11(17):5040.
16. Leucht S, Yurtseven DS, Priller J, Davis JM, van Os J, Sommer IEC. Towards a name change of schizophrenia: Positive and Negative Symptoms Disorder (PND). Eur Arch Psychiatry Clin Neurosci. 2025 Jun;275(4):963–71.
17. Striebel JM. What is schizophrenia - symptomatology. CNS Spectr. 2024 Dec 2;30(1): e12.
18. Safwi SR, Rizvi A, Usmani MA, Husain K, Brar K, Yadava D. Transcranial direct current stimulation and its effect on cognitive symptoms of schizophrenia: An updated review. Schizophr Res Cogn. 2024 Oct 23; 39:100335.
19. Moritz S, Borgmann L, Heinz A, Fuchs T, Gallinat J. Towards the DSM-6: Results of a Survey of Experts on the Reintroduction of First-Rank Symptoms as Core Criteria of Schizophrenia and on Redefining Hallucinations. Schizophr Bull. 2024 Aug 27;50(5):1050–54.
20. Heilskov SER, Urfer-Parnas A, Nordgaard J. Delusions in the general population: A systematic review with emphasis on methodology. Schizophr Res. 2020 Feb; 216:48–5.
21. Fouladirad S, Chen LV, Roes M, Chinchani A, Percival C, Khangura J, et al. Functional brain networks underlying probabilistic reasoning and delusions in schizophrenia. Psychiatry Res Neuroimaging. 2022 Jul; 323:111472.
22. Jolij J, Meurs M. Music alters visual perception. PLoS One. 2011 Apr 21;6(4): e18861.
23. Kuch M, Wöllner C. Effects of mobile music listening: Patterns of changes in focus of attention, environmental perception, and self-experience. Musicae Scientiae. 2024 Dec;28(4):740–57.
24. Tan SL, Spackman MP, Bezdek MA. Viewers' interpretations of film characters' emotions: Effects of presenting film music before or after a character is shown. Music Perception. 2007 Dec 1;25(2):135–52.
25. Bente G, Kryston K, Jahn NT, Schmälzle R. Building blocks of suspense: subjective and physiological effects of narrative content and film music. Humanities and Social Sciences Communications. 2022 Dec 13;9(1):1–3.
26. Dimitriadis T, Della Porta D, Perschl J, Evers AWM, Magee WL, Schaefer RS. Motivation and music interventions in adults: A systematic review. Neuropsychol Rehabil. 2024 Jun;34(5):649–78.
27. Sun Y. The Impact of Background Music on Flow, Work Engagement and Task Performance: A Randomized Controlled Study. Behav Sci (Basel). 2025 Mar 25;15(4):416.
28. Hegde S, Keshavan MS. The brain on the beat: How music may heal schizophrenia. Schizophr Res. 2023 Nov; 261:113–5.
29. Ivanova E, Panayotova T, Grechenliev I, Peshev B, Kolchakova P, Milanova V. A Complex Combination Therapy for a Complex Disease-Neuroimaging Evidence for the Effect of Music Therapy in Schizophrenia. Front Psychiatry. 2022 Mar 15; 13:795344.
30. Legge AW. On the neural mechanisms of music therapy in mental health care: Literature review and clinical implications. Music Therapy Perspectives. 2015 Jan 1;33(2):128–41.
31. Lv Y, Wen L, Hu WJ, Deng C, Ren HW, Bao YN, et al. Schizophrenia in the genetic era: a review from development history, clinical features and genomic research approaches to insights of susceptibility genes. Metab Brain Dis. 2024 Jan;39(1):147–71.
32. American Psychological Association. Schizophrenia. In: APA dictionary of psychology [Internet]. Washington (DC): American Psychological Association; 2023 [cited 2025 Dec 28]. Available from: https://dictionary.apa.org/schizophrenia.
33. Tandon R, Gaebel W, Barch DM, Bustillo J, Gur RE, Heckers S, et al. Definition and description of schizophrenia in the DSM-5. Schizophr Res. 2013 Oct;150(1):3–10.
34. Demyttenaere K, Leenaerts N, Acsai K, Sebe B, Laszlovszky I, Barabássy Á, et al. Disentangling the symptoms of schizophrenia: Network analysis in acute phase patients and in patients with predominant negative symptoms. Eur Psychiatry. 2021 Oct 13;65(1): e18.
35. Herkströter F, Zahedi A, Standke I, Dannlowski U, Lencer R, Schubotz RI, et al. Gray matter matters: Cognitive stability and flexibility in schizophrenia spectrum disorder. Psychophysiology. 2024 Sep;61(9): e14596.
36. Picó-Pérez M, Vieira R, Fernández-Rodríguez M, De Barros MAP, Radua J, Morgado P. Multimodal meta-analysis of structural gray matter, neurocognitive and social cognitive fMRI findings in schizophrenia patients. Psychol Med. 2022;52(4):614–24.
37. Howes OD, Kapur S. The dopamine hypothesis of schizophrenia: version III-the final common pathway. Schizophr Bull. 2009 May;35(3):549–62.
38. Weinberger DR. It's Dopamine and Schizophrenia All Over Again. Biol Psychiatry. 2022 Nov 15;92(10):757–9.
39. American Psychological Association. Dopamine hypothesis. In: APA dictionary of psychology [Internet]. Washington (DC): American Psychological Association; 2018 [cited 2025 Dec 31]. Available from: https://dictionary.apa.org/dopamine-hypothesis.
40. Leucht S, Siafis S, McGrath JJ, McGorry P, Howes OD, Tamminga C, et al. Schizophrenia. Nat Rev Dis Primers. 2025 Nov 27;11(1):83.
41. McCutcheon RA, Abi-Dargham A, Howes OD. Schizophrenia, Dopamine and the Striatum: From Biology to Symptoms. Trends Neurosci. 2019 Mar;42(3):205–20.
42. Bliźniewska-Kowalska KM, Gałecki P. Dopamine D2 receptor partial agonists in the treatment of schizophrenia-example of brexpiprazole. Psychiatr Pol. 2024 Aug 31;58(4):581–93.
43. American Psychological Association. Mesocortical system. In: APA dictionary of psychology [Internet]. Washington (DC): American Psychological Association; 2018 [cited 2026 Jan 4]. Available from: https://dictionary.apa.org/mesocortical-system.
44. American Psychological Association. Mesolimbic system. In: APA dictionary of psychology [Internet]. Washington (DC): American Psychological Association; 2018 [cited 2026 Jan 4]. Available from: https://dictionary.apa.org/mesolimbic-system.
45. Terauchi A, Johnson-Venkatesh EM, Umemori H. Establishing functionally segregated dopaminergic circuits. Trends Neurosci. 2025 Feb;48(2):156–70.
46. Corkrum M, Araque A. Astrocyte-neuron signaling in the mesolimbic dopamine system: the hidden stars of dopamine signaling. Neuropsychopharmacology. 2021 Oct;46(11):1864–72.
47. Szabadi E. Functional neuroanatomy of dopaminergic arousal systems: implications for the wake-promoting effect of psychostimulants, with particular reference to modafinil. Front Neuroanat. 2025 Dec 9; 19:1670564.
48. Liang S, Wu Y, Hanxiaoran L, Greenshaw AJ, Li T. Anhedonia in Depression and Schizophrenia: Brain Reward and Aversion Circuits. Neuropsychiatr Dis Treat. 2022 Jul 7; 18:1385–96.
49. Dabiri M, Dehghani Firouzabadi F, Yang K, Barker PB, Lee RR, Yousem DM. Neuroimaging in schizophrenia: A review article. Front Neurosci. 2022 Nov 15; 16:1042814.
50. Pike B, Ambrosio L, Ellett L. Brief psychological interventions for schizophrenia: a systematic review and meta-analysis. Psychol Med. 2025 May 13;55:e146.
51. Voineskos AN, Hawco C, Neufeld NH, Turner JA, Ameis SH, Anticevic A, et al. Functional magnetic resonance imaging in schizophrenia: current evidence, methodological advances, limitations and future directions. World Psychiatry. 2024 Feb;23(1):26–51.
52. Kunikullaya UK, Pranjić M, Rigby A, Pallás-Ferrer I, Anand H, Kunnavil R, et al. The molecular basis of music-induced neuroplasticity in humans: A systematic review. Neurosci Biobehav Rev. 2025 Aug; 175:106219.
53. Putkinen V, Seppälä K, Harju H, Hirvonen J, Karlsson HK, Nummenmaa L. Pleasurable music activates cerebral µ-opioid receptors: a combined PET-fMRI study. Eur J Nucl Med Mol Imaging. 2025 Aug;52(10):3540–9.
54. Lee WL, Sung HC, Liu SH, Chang SM. Meditative music listening to reduce state anxiety in patients during the uptake phase before positron emission tomography (PET) scans. Br J Radiol. 2017 Feb;90(1070):20160466.
55. Lin PH, Fu SH, Lee YC, Yang SY, Li YL. Examining the effects of binaural beat music on sleep quality, heart rate variability, and depression in older people with poor sleep quality in a long-term care institution: A randomized controlled trial. Geriatr Gerontol Int. 2024 Mar;24(3):297–304.
56. Tian Y, Ma L, Xu W, Chen S. The Influence of Listening to Music on Adults with Left-behind Experience Revealed by EEG-based Connectivity. Sci Rep. 2020 May 5;10(1):7575.
57. Miyapuram KP, Ahmad N, Pandey P, Lomas JD. Electroencephalography (EEG) dataset during naturalistic music listening comprising different genres with familiarity and enjoyment ratings. Data Brief. 2022 Oct 8; 45:108663.
58. Bartolomé-Tomás A, Sánchez-Reolid R, Fernández-Sotos A, Fernández-Caballero A, Latorre JM. Arousal Detection in Elderly People from Electrodermal Activity Using Musical Stimuli. Sensors (Basel). 2020 Aug 25;20(17):4788.
59. Sterenberg Mahon AJ, Roth EA. What elicits music-evoked nostalgia? An exploratory study among college students. Psych of Musi. 2023 Jan;51(1):159–71.
60. Taruffi L, Pehrs C, Skouras S, Koelsch S. Effects of Sad and Happy Music on Mind-Wandering and the Default Mode Network. Sci Rep. 2017 Oct 31;7(1):14396.
61. Chan MMY, Han YMY. The functional brain networks activated by music listening: A neuroimaging meta-analysis and implications for treatment. Neuropsychology. 2022 Jan;36(1):4–22.
62. Menon V, Levitin DJ. The rewards of music listening: response and physiological connectivity of the mesolimbic system. Neuroimage. 2005 Oct 15;28(1):175–84.
63. Mori K, Zatorre R. State-dependent connectivity in auditory-reward networks predicts peak pleasure experiences to music. PLoS Biol. 2024 Aug 12;22(8): e3002732.
64. Reybrouck M, Eerola T. Musical Enjoyment and Reward: From Hedonic Pleasure to Eudaimonic Listening. Behav Sci (Basel). 2022 May 19;12(5):154.
65. Vuust P, Heggli OA, Friston KJ, Kringelbach ML. Music in the brain. Nat Rev Neurosci. 2022 May;23(5):287–305.
66. Bowling DL. Biological principles for music and mental health. Transl Psychiatry. 2023 Dec 4;13(1):374.
67. Goltz F, Sadakata M. Do you listen to music while studying? A portrait of how people use music to optimize their cognitive performance. Acta Psychol (Amst). 2021 Oct; 220:103417.
68. Hofbauer LM, Lachmann T, Rodriguez FS. Background music varying in tempo and emotional valence differentially affects cognitive task performance: experimental within- participant comparison. J Cult Cogn Sci. 2024;8(2):139–50.
69. Lin TH, Liao YC, Tam KW, Chan L, Hsu TH. Effects of music therapy on cognition, quality of life, and neuropsychiatric symptoms of patients with dementia: A systematic review and meta-analysis of randomized controlled trials. Psychiatry Res. 2023 Nov; 329:115498.
70. Sihvonen AJ, Leo V, Ripollés P, Lehtovaara T, Ylönen A, Rajanaro P, et al. Vocal music enhances memory and language recovery after stroke: pooled results from two RCTs. Ann Clin Transl Neurol. 2020 Nov;7(11):2272–87.
71. Fan LP, Quijano-Ruiz A, Wang C, Zhao HW, Wang DN, Wu HM, et al. Effects of personalized music listening on post-stroke cognitive impairment: A randomized controlled trial. Complement Ther Clin Pract. 2024 Nov; 57:101885.
72. van der Steen JT, Smaling HJ, van der Wouden JC, Bruinsma MS, Scholten RJ, Vink AC. Music-based therapeutic interventions for people with dementia. Cochrane Database Syst Rev. 2018 Jul 23;7(7):CD003477.
73. Carlsson A, Waters N, Holm-Waters S, Tedroff J, Nilsson M, Carlsson ML. Interactions between monoamines, glutamate, and GABA in schizophrenia: new evidence. Annu Rev Pharmocol Toxicology. 2001; 41:237–60.
74. Coyle JT. NMDA receptor and schizophrenia: a brief history. Schizophr Bull. 2012 Sep;38(5):920–6.
75. Wassef A, Baker J, Kochan LD. GABA and schizophrenia: a review of basic science and clinical studies. J Clin Psychopharmacology. 2003 Dec;23(6):601–40.
76. Reiss JD. A meta-analysis of high resolution audio perceptual evaluation. J Audio Eng Soc. 2016 Jun 27;64(6):364–79.
77. Kalarikkal G, Jain C. A systematic review of psychoacoustic abilities in post-lingually hearing-impaired cochlear implant users. The Egyptian Jour of Otol. 2025 Apr 21;41(1):65.
