Abstract
The endometrium is thought to serve as the precursor tissue for diseases of the uterus such as endometrial cancer, adenomyosis, and endometriosis. More specifically, endometrial glands are proposed to be the source of developing endometrial cancer within the uterine body while ectopic endometrial glands and surrounding stroma which develop within the myometrium give rise to adenomyosis, and those that establish outside of the uterus give rise to endometriosis. While adenomyosis and endometriosis are benign diseases, they do share several cellular characteristics with endometrial cancer including enhanced cell survival/proliferation, migration and invasion. Further, the pathophysiology of these diseases is driven by unopposed estrogen and each exhibit various degrees of progesterone resistance and altered downstream expression and/or function of genes relevant to epithelial-to-mesenchymal transition, tissue remodeling and transcriptional signaling. In this review we summarize our current understanding on differences and similarities in major cell signaling pathways among endometrial cancer, adenomyosis and endometriosis. In closing, we emphasize the necessity for additional studies to delve deeper into common attributes in the pathophysiology of these diseases towards the goal of identifying novel, non-hormonal therapies for their treatment.
Keywords
Endometrial cancer, Adenomyosis, Endometriosis, Protease, Protease inhibitor, Tissue remodeling, Epithelial-to-mesenchymal transition, Steroid receptor, Transcription factor
Introduction
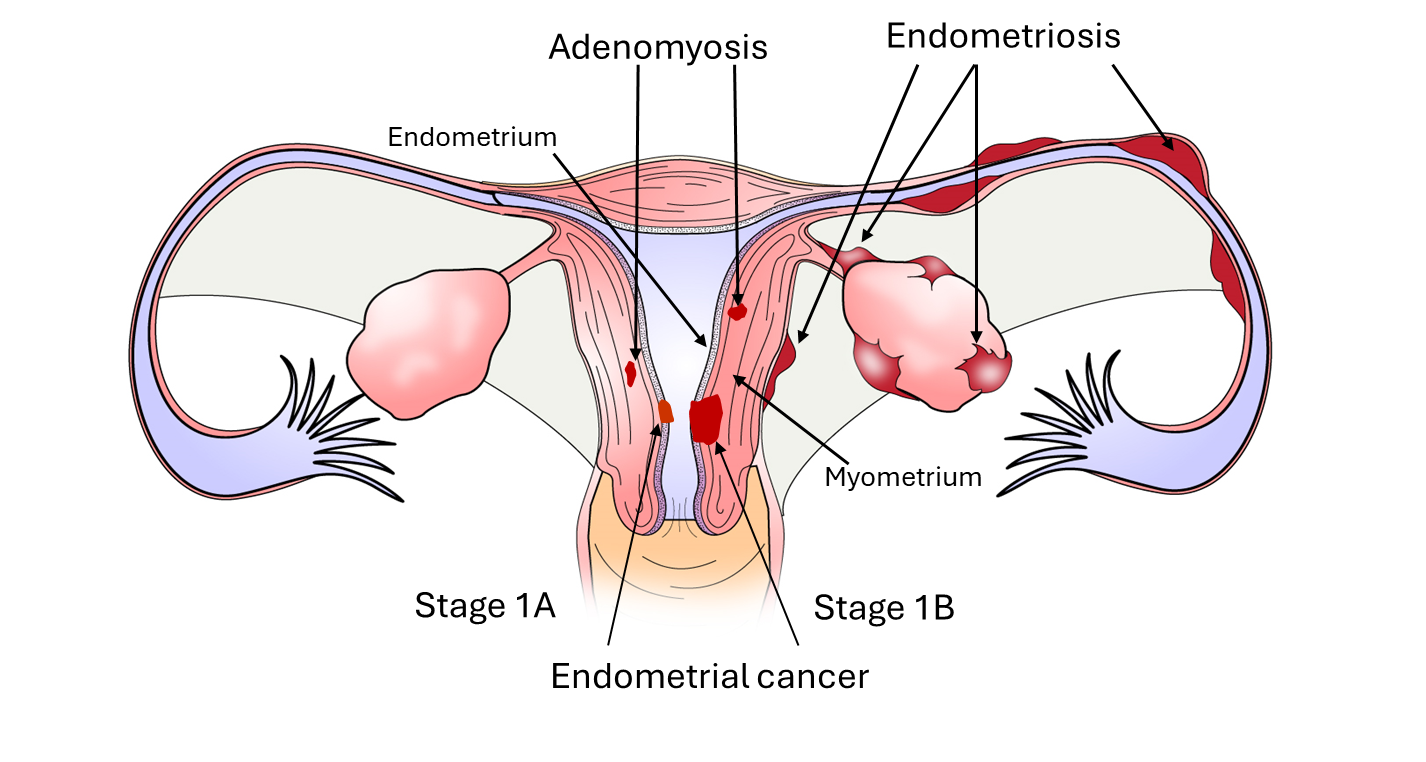
The uterus is an essential female organ required for the propagation of all mammalian species. The uterus is composed of two major tissue layers, the endometrium, which is the inner layer as is further classified as the functionalis or basalis layer whose major functions are to provide the site of attachment of implanting embryos and to give rise to a new functionalis layer with regeneration of the endometrial lining which occurs with each menstrual cycle. The myometrium is the muscular tissue component of the uterus which is critical for contractions during labor and delivery. The endometrial lining is composed of three major cell types, the endometrial luminal epithelial cells, the glandular epithelial cells and the stromal cells. Endometrial cancer [1,2] develops when endometrial glands become hyperplastic and invasive developing first within the endometrium (Stage IA; Figure 1) and then spreading to the underlying myometrium (Stage 1B; Figure 1) with both limited to the uterine body. Stage II tumors typically invade into cervical stromal connective tissue, but without invading beyond the uterus, while stage III tumors may invade local or regional structures beyond the uterus including the lymph nodes. Adenomyosis is defined as growth of endometrial epithelial glands and surrounding stroma which develop within the myometrium (Figure 1) while endometriosis is composed of both glands and stroma which develop ectopically in the pelvic cavity (Figure 1). In the case of endometriosis, these lesions can develop throughout the pelvic cavity with the majority of lesions developing on the ovary, the peritoneal lining and in the cul-de-sac. Endometrial cancer cells, adenomyotic epithelial and stromal cells and endometriotic epithelial and stromal cells all exhibit characteristics of enhanced estrogen signaling and reduced progesterone action including augmented cell proliferation, migration and invasion. Key players in the pathophysiology include the sex steroid receptors, mediators of epithelial-to-mesenchymal transition (which contribute to cell migration and invasion), proteases and protease inhibitors and transcription factors. In the following paragraphs, we examine the expression of steroid receptors, epithelial-mesenchymal transition proteins, proteases, protease inhibitors, and transcription factors among these three endometrial diseases. To do so, we evaluated the literature through a search of the PUBMED/MEDLINE database. Search terms included publications with “endometrial cancer”, “adenomyosis” and “endometriosis” in the title. Each disease was separately assessed in conjunction (and) with “steroid receptor”, or “epithelial-mesenchymal-transition”, or “protease”/”protease inhibitor” or “transcription factor” in the article title.
Steroid Receptors Expression in Malignant and Benign Endometrial Diseases
Endometrial cancer
Both the level of expression and cellular localization of the steroid receptors, estrogen receptor and progesterone receptor are critical for normal responses of the endometrium to the sex hormones estrogen and progesterone, respectively [3]. Estrogen receptor (ER) is known for its role in increasing cell proliferation during the proliferative phase of the female menstrual cycle, while progesterone receptor (PR) is known to block estrogen-induced proliferation of endometrial glands and promote stromal cell proliferation and differentiation. However, these steroid receptors have differing patterns of expression in endometrial cancer and diseases of the uterus [4–6]. A systematic review and meta-analysis by Zhang and colleagues [5] strongly support the notion that in subjects with endometrial cancer there is an inverse relationship between the levels of ER and PR and survival. More recently, a collaborative study by a European Network for Individualized Treatment of Endometrial Cancer [6], identified distinct clinical outcomes based upon level of ER and PR expression level. Cutoff values of 0–10% ER/PR positive was associated with unfavorable outcomes, 20–80% was associated with intermediate outcomes (defined as) while 90–100% was associated with favorable outcomes. Di Nezza and colleagues further explore how reduced PR expression may impact the pathophysiology of EC. In this study they discovered that progesterone inhibits the expression of matrix metalloproteinases (MMPs), thus suppressing mechanisms such as extracellular matrix degradation and the processing of growth factors and cytokines that aid and promote cancer cells to invade [7]. Fujimoto and colleagues observed a decrease in the mRNA levels of ER-α and ER-β, and an upregulation in ERR α in relation to clinical stage, myometrial invasion, and dedifferentiation (Table 1) [8]. On the other hand, some studies postulate that in cancer patients who are obese, there is an upregulation of steroid receptors. This signifies a possible link between the two. For example, patients with positive PR expression were most often young and obese [9], and additionally, those who were obese had a higher probability of having an ER or PR-positive tumor [10]. From these studies it is clear that low ER and PR expression is associated with advanced endometrial cancer and poor outcomes.
|
Steroid Receptor |
Expression |
References |
|
ER- β |
Downregulated in EC |
[8] |
|
ER- α |
Downregulated in EC |
[8] |
|
ERR- α |
Upregulated in EC |
[8] |
|
ER |
Inverse relationship between ER and survival Higher in obese patients |
[5] [9,10] |
|
PR |
Inverse relationship between PR and survival Inhibits MMPs, suppresses growth factors and extracellular matrix degradation Higher in obese patients |
[5] [7] [9,10] |
Adenomyosis
Compared to EC, there is limited information on ER and PR expression in adenomyosis as well as inconsistencies between findings regarding steroid receptor expression in adenomyosis. For example, Tamaya and colleagues evaluated steroid receptor levels in ten cases of adenomyosis utilizing Scatchard plot analysis [11]. It was found that the expression of both ER and androgen receptor (AR) was detected in each case, while PR was not detected in the majority of cases. Similarly, using tissue microarray and dendrogram cluster analysis, Jian-Jun Wei and colleagues found that the larger the tumor size, the more downregulated ER and PR expression was [12]. A morphometric investigation and evaluation of cytokeratin, vimentin, and steroid receptor content was conducted by Donnez and colleagues to compare what they initially thought to be endometriotic nodules of the rectovaginal septum with peritoneal endometriosis [13]. By the end of the study, a new type of adenomyoma was categorized, specifically rectovaginal adenomyoma, and their results demonstrated that the expression of ER and PR content in this type of lesion was significantly lower compared to eutopic endometrium tissue [13].
In contrast to other studies, Maria Sztachelska and colleagues, in a study in which the function and expression of ER and PR were evaluated confirmed the presence of ER and PR at both gene and protein levels and an upregulation of membrane progesterone receptors α and β [14]. Another study found that ERβ expression was significantly high in adenomyotic lesions but declined with increasing lesional fibrosis, while there was no significant difference in ERα expression in comparison to control tissues. They determined that with increased fibrosis in ectopic endometrium, estrogen signaling decreases [15]. Mehasseb and colleagues detected reduced levels of ER-α in eutopic and ectopic (adenomyosis) endometrium in subjects with adenomyosis compared to those without, while ER-β expression was increased in adenomyotic functionalis glands during the proliferative phase. Similar to ER-α expression, PR-A and PR-B expression in the basalis stroma, and inner and outer myometrium were reduced compared to controls [16] (Table 2).
|
Steroid Receptor |
Expression |
References |
|
ER |
While its expression has been detected, it is decreased in adenomyoma comparison to eutopic tissue. |
[11–13,15] |
|
ER- α |
Reduced levels in eutopic and ectopic endometrium |
[16] |
|
ERβ |
Decreased expression as lesional fibrosis increases Elevated expression during proliferative phase |
[15] [16] |
|
PR |
While its expression has been detected, it is decreased in adenomyoma comparison to eutopic tissue. Downregulation detected |
[12,13] [16] |
|
mPR α |
Upregulated in adenomyosis |
[14] |
Endometriosis
A Scatchard plot analysis was utilized by Tamaya and colleagues to determine the expression levels of steroid receptors in endometriosis [11]. In comparison to normal endometrium tissue, the ER and PR levels were lower in endometriosis tissues. An immunohistochemical analysis carried out by Nisolle and colleagues assessed the differences in proliferative activity throughout the menstrual cycle between eutopic and ectopic endometrium [17]. It was observed that while the eutopic endometrium was not proliferatively active during the late secretory phase, the ectopic endometrium was, and instead of decreasing, PR expression in the glandular epithelium and ER expression in the glandular epithelium and stroma stayed consistent (Table 3). A study by Pluchino and colleagues using tissue microarray and immunohistochemistry demonstrated that in endometriosis, ER-α, PR, AR, and aromatase were expressed [18]. Specifically, as the severity of the symptoms experienced by patients was evaluated, it was determined that ER- α was linked to the differences in severity, with higher levels being more severe. Fazleabas and colleagues discovered through RT-PCR and immunocytochemistry that while PR and ER-α were expressed in both eutopic and ectopic endometrium, ER-β was only found in ectopic endometriotic lesions, thus signifying its upregulation in the disease [19]. Aromatase was additionally detected, but specifically ten months after the baboons were inoculated with menstrual endometrium to induce experimental endometriosis. In a study that examined both the expression of ER-α and ER- β, low expression levels of ER-α, and higher levels of expression of ER- β were observed [20]. The expression of steroid receptor coactivators has been found to differ between eutopic and ectopic tissue samples as well.
|
Steroid Receptor |
Expression |
References |
|
ER |
Lower expression in endometriotic tissue. Instead of decreasing during the late secretory phase, levels remained constant |
[11] [17] |
|
PR |
Lower expression in endometriotic tissue Instead of decreasing during the late secretory phase, levels remained constant |
[11] [17] |
|
ER-α |
The higher its expression, the more severe the disease Lower expression |
[18] [20] |
|
ER-β |
Upregulated in endometriosis |
[19,20] |
Epithelial-to-Mesenchymal Transition (EMT) Proteins in Malignant and Benign Endometrial Diseases
Endometrial cancer
Epithelial-mesenchymal transition (EMT) induces the spread of many diseases and cancers, including endometrial cancer (EC). EMT occurs when the epithelial cells transform into mesenchymal cells as they gain migratory and invasive characteristics [21]. The cells lose their cell polarity and cell-to-cell adhesion. E-cadherin (CDH1), a protein that regulates cellular signaling and tumor suppression, is a biomarker of EMT. In general, it is downregulated in EMT. Zhou and colleagues observed that a high expression of E-cadherin in EC, which was caused by an upregulation of SOX17, is linked to a better prognosis (Table 4) [22]. As mentioned earlier, ZEB1, or Zinc finger E-box binding homeobox 1 (ZEB1) is a transcription factor linked to EMT. Xiao and colleagues silenced ZEB1 and observed that this decreased cell migration and invasion [23]. They also found that the β -catenin/TCF4 signaling feedback stimulated ZEB1 transcription. ZEB2, along with SLUG, are overexpressed in EC, and act as predictors of higher FIGO stages [24]. In one study, it was found that bone morphogenetic protein 2 (BMP2) induced EMT by activating factors such as SLUG or SNAIL [25]. Snail has been linked to a worse prognosis for EC [26]. Piwil1 is an oncogene-like stem cell protein that is upregulated in EC. Chen and colleagues were able to determine that Piwil1 has a role in increasing mesenchymal markers and suppressing E-cadherin, which results in an EMT-like phenotype [27].
|
EMT Proteins |
Expression |
References |
|
E-cadherin |
Higher expression is linked to a better prognosis When migration was inhibited, its expression increased |
[22] |
|
ZEB1 |
Induces EMT |
[23] |
|
ZEB2 |
Higher expression linked to higher FIGO stages |
[24] |
|
Snail |
Linked to a worse prognosis |
[26] |
|
SLUG |
Higher expression linked to higher FIGO stages |
[24] |
|
BMP2 |
Triggers EMT by SLUG or SNAIL |
[25] |
|
Piwil1 |
Causes an increase in mesenchymal markers and suppresses E-cadherin |
[27] |
Adenomyosis
In a study by Zheng and colleagues in which the role of focal adhesion kinase (FAK) was evaluated, they determined that it is upregulated in adenomyosis and may regulate the EMT through the FAK/phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) pathway (Table 5) [28]. Many studies have discovered a link between E-cadherin and EMT in adenomyosis. When decreased, migration and invasion are induced [29]. N-cadherin, on the other hand, is found to be upregulated in adenomyosis, specifically when EMT is induced, which is why it is considered a characterization of a mesenchymal phenotype [29,30]. More specifically, Yoo and colleagues proposed that β -catenin activates TGF- β induced EMT, as there was a strong correlation between β -catenin and TGF- β2 in women with this disease [31]. Overall, β-catenin activity promotes EMT in adenomyosis. High activity levels of Notch1, a transmembrane receptor in which its signaling pathway regulates cellular signaling, were observed by Bourdon and associates in adenomyosis-induced mice. As Notch1 activation correlated with elevated levels of immune and EMT markers, including E-cadherin, Vimentin, Tgfβ, Snail1, and Slug, Snail3, it was concluded that Notch1 activation corresponds with the atypical expression of these markers in the progression of adenomyosis [32]. SPARC-related modular calcium binding 2 (SMOC2) is an extracellular matrix-associated protein. Its overexpression results in the upregulation of N-cadherin and alpha-SMA – another mesenchymal marker – as well as the downregulation of E-cadherin. This suggests that SMOC2 promotes EMT [33]. Hu and colleagues aimed to determine the role of transmembrane glycoprotein neuropilin 1 (NRP1) in EMT by infecting endometrial cells with NRP1 retroviruses so as to upregulate NRP1. A mesenchymal phenotype, downregulation of E-cadherin, upregulation of alpha-SMA and N-cadherin, and enhanced migration were observed. In contrast, NRP1 shRNA inhibited EMT. This signifies its possible role in the EMT and the progression of adenomyosis [34]. Similar to endometrial cancer, several EMT proteins are misexpressed in adenomyosis. ZEB1 is elevated in endometrial adenomyosis tissue, and its upregulation is modulated by SKP1 and SKP2 [35] in eutopic adenomyosis endometrial stromal cells. Qi and colleagues [29] reported that Notch1, N-cadherin, Snail and Slug were upregulated in eutopic endometrial tissue from adenomyosis patients compared to normal endometrium while E-cadherin expression was reduced.
|
EMT Proteins |
Expression |
References |
|
Focal Adhesion Kinase (FAK) |
Upregulated in adenomyosis; regulates EMT through FAK/PI3K/AKT pathway |
[28] |
|
E-cadherin |
Inhibits EMT and migration/invasion |
[29] |
|
N-cadherin |
Induces EMT and migration/invasion. Upregulated in eutopic endometrial tissue of adenomyosis patients |
[29,30] |
|
β -catenin |
Induces EMT |
[31] |
|
TGF β-2 |
Induces EMT |
[31] |
|
SMOC2 |
Promotes EMT by upregulating mesenchymal markers and downregulating E-cadherin |
[33]
|
|
Notch1 |
Upregulated |
[29,32] |
|
NRP1 |
Overexpression caused a mesenchymal phenotype, downregulated E-cadherin, and upregulated mesenchymal markers |
[34] |
|
ZEB1 |
Elevated in adenomyosis tissue; modulated by SKP1 and SKP2 |
[35] |
|
Slug |
Upregulated in eutopic endometrial tissue of adenomyosis patients |
[29]
|
|
Snail |
Upregulated in eutopic endometrial tissue of adenomyosis patients |
[29] |
Endometriosis
Like endometrial cancer and adenomyosis, E-cadherin expression is reduced in endometriosis (Table 6). Gaetje and associates first reported that endometriotic epithelial cells lack E-cadherin expression which was associated with cell invasion in vitro and that E-cadherin negative epithelial cells were increased in endometriotic tissue compared to controls [36]. Sirtuin 1(SIRT1) is a protein that deacetylates transcription factors to impact gene expression. When overexpressed in endometriotic epithelial cells, it inhibits oncogene induced senescence and is therefore able to trigger EMT [37]. Enhancer of Zeste homolog 2 (EZH2) has been identified as an enzyme that promotes EMT in endometriosis. Its overexpression upregulated Snail, Slug, and vimentin expression, while it downregulated E-cadherin. When inhibited, the opposite occurred, resulting in the restoration of the epithelial phenotype of the EECs and a reduction in migration and invasion [38]. Huang and colleagues evaluated the role of matrix metalloproteinase-9 (MMP-9) through a statistical analysis and discovered that there is a higher concentration of MMP-9 in EMT patients, and these concentrations are proportional to the stage of the disease [39]. Regarding the mesenchymal markers, TWIST1, SNAIL, and SLUG were overexpressed, while CDH1, or E-cadherin, expression was downregulated. MYC is another protein associated with endometriosis. It was concluded that both TWIST1 and MYC induce EMT, resulting in the development of endometriotic lesions [40]. Snail was additionally found to be activated by β-catenin/TCF-3 in endometriosis. β -catenin knockdown resulted in the inhibition of oestrogen-induced Snail mediated EMT [41]. Wu and colleagues determined that ZEB1 has a vital role in inducing endometriosis, as it was overexpressed, along with EMT markers Vimentin and N-cadherin. When ZEB1 was downregulated, migration, invasion, and EMT of the Ishikawa cells were inhibited [42].
In summary, among the three diseases, E-cadherin expression appears to be reduced, while ZEB1, Snail and Slug expression are elevated in each case and associated with cell migration/invasion.
|
EMT Proteins |
Expression |
References |
|
E-cadherin |
Reduced expression |
[36] |
|
SIRT1 |
Triggers EMT by inhibiting apoptosis |
[37] |
|
EZH2 |
Overexpression induces EMT by upregulating mesenchymal markers |
[38] |
|
MMP9 |
Higher concentration in EMT patients than healthy controls |
[39] |
|
TWIST1 |
Induces EMT |
[40] |
|
SLUG |
Overexpressed |
[40] |
|
SNAIL |
Overexpressed |
[40] |
|
β -catenin |
Activates Snail; knockdown inhibits estrogen-induced Snail mediated EMT |
[41] |
|
ZEB1 |
Overexpression induced migration and EMT |
[42] |
Protease/Protease Inhibitor Expression in Malignant and Benign Endometrial Diseases
Endometrial cancer
Proteases play a crucial role in regulating endometrial diseases, which can be used to the advantage during treatment. For example, Huang and colleagues determined that ubiquitin-specific protease 31 (USP31) can be used to foretell a prognosis. Those with lower levels of USP31 will most likely have a worse prognosis, and vice versa [43] (Table 7). Another protease serves a similar function. The expression of HtrA serine peptidase 1 (HtrA1) is inversely related to tumor grade. Its downregulation increased the invasiveness of papillary serous EC cell lines, and its upregulation decreased their invasive ability [44]. Another important group of proteases is cathepsins, which are generally found within lysosomes. In comparison to normal endometrium, an upregulation of Cathepsin B has been observed in EC tissues. When suppressed, it resulted in a decrease in proliferation [45]. It has also been found to be correlated with the FIGO stage of EC [46]. Cathepsin D additionally could aid in the prognosis through immunohistochemical detections [47]. Lower levels of expression are correlated with a worse prognosis [48]. Skrzypczak and associates collected data supporting the possibility that cysteine protease cathepsin L (CTSL2) may have a role in the progression of EC. It was highly expressed, and it had a positive correlation with the expression of growth regulatory genes [49]. One study discovered that the caseinolytic mitochondrial matrix peptidase proteolytic subunit (CLPP) plays a vital role in the proliferation of type I EC cancer tissue. Its upregulation in EC suggested that it may also play a role in removing damaged mitochondrial proteins [50]. Another protease that may affect EC and its prognosis is hepsin, a type II transmembrane serine protease. There is an inconsistency of findings regarding its role in endometrial cancer. Matsuo and colleagues concluded that it is overexpressed in EC, and the higher its level of protein expression, the more advanced the cancer is [51]. However, Nakamura and colleagues determined that Hepsin, the gene that encodes for the protein hepsin, instead inhibits cell growth [52].
|
Proteases |
Expression |
References |
|
USP31 |
Downregulated in EC; inverse relationship with prognosis |
[43] |
|
HtrA1 |
Downregulated in EC; inverse relationship with tumor grade |
[44] |
|
Cathepsin B |
Upregulated in EC |
[45,46] |
|
Cathepsin D |
Downregulated in EC; can be used to assess prognosis |
[47,8] |
|
CTSL2 |
Upregulated in EC; may cause the progression of EC |
[49] |
|
CLPP |
Increased expression; may play a role in removing damaged mitochondrial proteins |
[50] |
|
Hepsin |
Inhibits cell growth Upregulated in EC |
[51] [52] |
|
MMP-2 |
Upregulated in EC Inhibition reduces cell proliferation, migration, invasion |
[54] [55,56] |
|
MMP-9 |
Upregulated in EC Inhibition reduces cell proliferation, migration, enhances apoptosis |
[54] [57,58] |
|
Protease Inhibitors |
Expression |
References |
|
PAI-1 |
High expression; associated with grade of cancer |
[60] |
|
SERPINA3 |
Upregulated: regulates G2/M checkpoint and inhibits apoptosis |
[61] |
|
HE4 |
Overexpression associated with worse prognosis |
[62,63] |
|
ZPI |
Upregulated |
[64] |
|
HAI-1 & HAI-2 |
Downregulated; their expression inhibits proliferation |
[65] |
|
Bortezomib |
Increased mRNA expression of caspases-3 and caspases-9; decreased mRNA expression of bcl-2 |
[66] |
|
TIMP-2 |
Elevated in blood serum |
[67] |
|
TIMP-1 |
Decreased expression Increased expression |
[67] [68] |
Matrix metalloproteinases (MMPs) are key proteases which degrade the extracellular matrix and contribute to cell migration and invasion [53]. Among the numerous MMPs, MMP-2 and MMP-9 have been the most studied in endometrial cancer. Aglund and colleagues [54] first reported that overexpression of both MMP-2 and MMP-9 were associated with poor patient survival. More recent studies have demonstrated in vitro functionality of MMP2 and MMP9 using cell assays. SPOCK2, which was reported to be lower in endometrial cancer, was demonstrated in vitro to mediate cell proliferation, invasion and migration by mediating MMP2 (and MT1-MMP) expression and action [55], while Wang and colleagues [56] demonstrated that MMP2 could be inhibited by over-expression of the mature form of IL-37 (IL137bΔ1-45) decreased cell migration and invasion by inhibiting MMP2 expression. Targeting of MMP-9 to reduce cell proliferation, migration and/or invasion was demonstrated in three independent studies. Ruan and associates [57] demonstrated that miR-183 up-regulation in endometrial cancer cells targets MMP-9 expression to augment cell proliferation and invasion in vitro, while Zhao and colleagues [58] reported that the long non-coding RNA, GATA6-AS, is reduced in endometrial cancer tissue and cell lines and that its overexpression in vitro augments cell proliferation, reduced cell apoptosis by mediating MMP-9 expression.
The net result of protease action is a combination of expression of the protease as well as that of the respective protease inhibitors [59]. Thus, several of the inhibitors of the aforementioned proteases have been examined in the context of EC. The expression of protease inhibitors has been found to significantly impact the progression of endometrial cancer. Plasminogen activator inhibitor type 1 (PAI-1) inhibits proteases important in fibrinolysis. A study found high levels of PAI-1, as well as PAI-1 4G/5G polymorphism, in the blood of endometrial cancer patients, indicating a vital role in EC. The higher the PAI-1 level, the higher the grade [60]. Another protease inhibitor that is upregulated in EC is SERPINA3. Yang and colleagues discovered that it modulates the G2/M checkpoint of the cell cycle and additionally inhibits cell death [61]. Human epididymis protein 4 (HE4) is a protease inhibitor that can signify a worse prognosis for EC patients [62]. Its expression increases with the clinical stage. When overexpressed, proliferation is promoted and malignant phenotypes are enhanced [63]. The upregulation of protein Z-dependent protease inhibitor (ZPI), as well as its cofactor protein Z (PZ), in EC cells also indicate that they play a role in endometrial cancer [64]. They were not expressed in normal tissues. On the other hand, hepatocyte growth factor activator inhibitors HAI-1 and HAI-2 are downregulated in EC. Their decrease is associated with a worse prognosis. Nakamura and colleagues observed that they have an inhibitory effect on EC cell migration and invasion, which is achieved through decreasing matriptase and hepsin levels. These protease inhibitors reduced ER and PR signal transduction, reduced the expression of mesenchymal markers, and increased that of epithelial markers [65]. Lastly, bortezomib, a treatment for some cancers, may also be able to affect endometrial cancer cells. In an Ishikawa cell line, bortezomib decreased the mRNA expression of bcl-2, a protein that inhibits apoptosis, while it increased the mRNA expression of caspases-3 and caspases-9, both of which are activated during apoptosis. Thus, bortezomib was able to increase the susceptibility of Ishikawa cells to chemotherapy [66]. Contrary to bortezomib, the appearance of carcinoma and benign neoplasms are correlated with TIMP-2 elevation in the blood serum. On the other hand, there were lower TIMP-1 levels in endometrial cancer patients in comparison to controls and patients with polyps and hyperplasia. The researchers concluded that they cannot be considered diagnostic markers but can be used in the prognosis process [67]. Additionally, TIMP-1 was upregulated in a high risk patient group [68].
Adenomyosis
In a study that evaluated how adenomyosis impacted the coagulation and fibrinolysis system during menstruation, 50% of the patients had an increase in thrombin-antithrombin complex (TAT) and plasmin-alpha 2-plasmin inhibitor complex (PIC). According to Yamanaka and colleagues, this data indicates that adenomyosis patients are at risk of developing thrombosis, infarction, and exacerbated menorrhagia [69]. Menorrhagia could be caused by activated fibrinolysis during menstruation. Mori and colleagues investigated the effects of orally active-matrix metalloproteinase (MMP) inhibitor (ONO-4817) on mice with uterine adenomyosis. There was a lower probability that adenomyosis would progress in mice treated with ONO-4817. Mice with 1% ONO-4817 had less progression in the uteri, specifically 2.71 ± 0.93, with the degree of pathological progression graded from 1 to 5, while the uteri that was not exposed had 4.33 ± 0.75. Additionally, ONO-4817 inhibited the invasion of stromal cells into the gel, indicating that ONO-4817 could be potent in suppressing the development of adenomyosis [70]. Many studies have discovered that matrix metalloproteinases are upregulated in adenomyosis lesions. MMP-2 and MMP-9 specifically are most often elevated (Table 8) [71]. As a result, some have concluded that they may play a role in the invasion of endometrial tissues into the myometrium [72]. In the endometrial stromal cells of patients with adenomyosis, there was a higher secretion of TIMP-1 in comparison to those without the disease [73]. On the other hand, in active adenomyosis, there was a lesser expression of TIMP-1, TIMP-3, and TIMP-4, a decrease in the occurrence of apoptosis, and more proliferation by glandular and stromal cells [74].
|
Proteases |
Expression |
References |
|
MMP-2 |
Elevated expression |
[71] |
|
MMP-9 |
Elevated expression |
[71] |
|
Protease Inhibitors |
Expression |
References |
|
TAT |
Increases risk of developing thrombosis, infarction, and exacerbated menorrhagia |
[69] |
|
ONO-4817 |
Suppressed development of adenomyosis |
[70] |
|
TIMP-1 |
Higher secretion in those with adenomyosis than those without. Decrease in expression |
[73] [74] |
|
TIMP-3 & TIMP-4 |
Decrease in expression |
[74] |
Endometriosis
There are a variety of proteases that regulate endometriosis. For one, Porter and associates supported their hypotheses that cysteine cathepsins promote tissue invasion and lesion establishment, and when inhibited it could block the attachment of endometriosis lesions with the data collected from their study. Cathepsins L and K were significantly upregulated in the endometriotic lesions of both mice and humans in comparison to other tissues, and when inhibited by E-64, there were much less attached endometriosis lesions [75]. Cathepsin G concentration was additionally significantly higher in endometriosis tissues (Table 9) [76]. Calcium-activated neutral protease 7 (CAPN7) is a protease that upregulates MMP-2, thus promoting human endometrial stromal cell (hESC) migration and invasion [77]. Thrombin also activated MMP-2 in endometriotic stromal cells (ESCs). This was suppressed by thrombin inhibitor d-phenylalanyl-l-prolyl-l arginine chloromethyl ketone (PPACK). The researchers concluded that the thrombin system stimulates the inflammatory response of endometriotic cells and mitogenic activity [78]. Kusama and associates discovered that thrombin induces transformations similar to epithelial-mesenchymal transition (EMT) and fibroblast to myofibroblast transdifferentiation [79].
Tissue remodeling is an important process regulated by MMPs and TIMPs. There are conflicting observations regarding the concentration of MMP-2 in endometriotic tissues. Tang and associates discovered that the promoter regions of MMP2, as well as MMP3 and MMP7, underwent notable changes in endometriotic lesions in comparison to eutopic tissue during the proliferative phase [80]. Chen and colleagues found that MMP2 expression decreased with disease [81], while Malvezzi and colleagues observed that in infertile women, its levels increased in stage III/IV in comparison to women with stage I/II endometriosis [82]. MMP-9 is overexpressed in endometriotic tissues [83]. Additionally, there is elevated expression of membrane-type 5 metalloproteinase (MT5-MMP) in eutopic endometrium and a greater amount of transcript levels in most peritoneal lesions [84]. An immunohistochemistry demonstrated strong staining in luminal epithelial cells. Put together with the results of an rt-PCR, it was deduced that MT5-MMP expression leads to an increase in tissue remodeling and cell migration. In red peritoneal and ovarian endometriosis, MMP1 mRNA expression was detected, but not in black peritoneal and rectovaginal lesions [85]. Its expression was also linked to tissue remodeling and bleeding, with a possible role in the secondary shedding and reimplantation of endometriotic lesions.
The development of endometriosis may be regulated by Ubiquitin Specific Protease 7 (USP7). It is upregulated in ectopic endometrial stromal cells (EECs), promoting irregular behavior and regulating the expression of DNA methyltransferase 1 (DNMT1), a protein that when silenced, suppresses the oncogenic characteristics caused by USP7 [86]. Furthermore, the overexpression of ubiquitin-specific protease 10 (USP10) is also linked to migration, proliferation, and suppression of apoptosis. Chen and colleagues believe it accomplishes this by activating the Raf-1/ERK kinase (MEK)/extracellular signal-regulated kinase (ERK) signaling pathway [87]. Finally, one protease that is instead downregulated in endometriosis is high temperature requirement A1 (HtrA1). The researchers determined that it had a direct correlation with TGFβ1 [88].
A multitude of studies have also discovered many protease inhibitors that may influence or cause the onset and progression of endometriosis. A comparative study conducted by Fazleabas evaluated the general concentration of protease inhibitors in women with endometriosis. It was observed that during the luteal phase, there was a decrease in protease inhibitor concentration in the uterine cavity, but an increase in the peritoneal fluid [89]. Next, both the secretory leukocyte protease inhibitor (SLPI), which inhibits human leukocyte elastase, and its gene were significantly upregulated in the peritoneal fluid of women [90,91]. In the endometriosis-like grafts of mice that underwent a unilateral ovariectomy (uOVX), the expression level of alpha1-antitrypsin (alpha1-AT) decreased, which Tamura and colleagues suggested may induce inflammation. It also blocked interleukin-8 expression (IL-8; [92]). Mao and associates assessed the role kallistatin (KS) plays in endometriosis. It had low levels of expression, which decreased with the severity of the disease. It was found that it has the ability to increase intracellular reactive oxygen species (ROS) levels, and it additionally, modulates caspase 3 signaling and manganese superoxide dismutase (MnSOD), which together inhibit the progression of the disease [93]. Recently, the production of alpha-2 macroglobulin (A2M), a broad-spectrum plasma protease inhibitor, in human endometrium was reported. Sayegh and colleagues studied its expression and effects on mouse embryo development. Strong signaling expression was detected in the secretory endothelium compared to proliferative endothelium. The blastocyte development of mouse embryos in vitro was suppressed at A2M concentrations of 400-400 mmol/L. It also inhibited mouse embryo development [94]. TIMP-1, the tissue inhibitor of metalloproteinases-1, was found in a multitude of studies to have reduced expression in endometriosis [95,96]. Madjid and colleagues discovered that in the endometrial cells of subjects with decreased TIMP-1 expression had a higher risk of endometriosis [97]. However, one study did not determine a statistical significance between the difference in expression of TIMP-1 in the eutopic endometrium of subjects with early endometriosis and subjects with a healthy endometrium [98]. In late endometriosis, TIMP-2 is downregulated, which could signify an increase in MMP-2 as endometriosis advances [99]. Additionally, the promoter regions of TIMP3 and TIMP4 underwent notable changes in endometriotic lesions in comparison to eutopic tissue during the proliferative phase [80].
In summary, each of the three diseases exhibit signs of tissue remodeling and invasion. Not surprisingly, increases in protease expression has been reported in each of these diseases while the expression of protease inhibitor expression is less consistent, some report reduction while others have been reported to be elevated
|
Protease |
Expression |
References |
|
Cathepsins L & K |
Upregulated; when inhibited, less endometriotic lesions can attach |
[75] |
|
Cathepsin G |
High concentrations |
[76] |
|
CAPN7 |
Upregulates MMP-2 and promotes migration and invasion of hESC |
[77] |
|
MMP-2 |
Decreased Overexpressed, promotes migration and invasion, related to decrease in TIMP-2 |
[80] [81,82] |
|
MMP-9 |
upregulated |
[83] |
|
MT5-MMP |
High transcript levels in peritoneal endometriosis; leads to increase in tissue remodeling |
[84] |
|
MMP-1 |
Expression detected in red peritoneal and ovarian endometriosis; linked to tissue remodeling and bleeding |
[85] |
|
USP7 |
Upregulated, regulates DNMT1, promotes migration and invasion |
[86] |
|
USP10 |
Activates Raf-1/MEK/ERK signaling pathway and promotes proliferation and suppresses apoptosis |
[87] |
|
HtrA1 |
Downregulated |
[78] |
|
Thrombin |
Activates MMP-2; promotes changes resembling EMT and FMT |
[78] |
|
Protease Inhibitors |
Expression |
References |
|
Protease inhibitor |
During the luteal phase, downregulated in uterine cavity, upregulated in PT |
[89] |
|
SLPI |
Upregulated in peritoneal fluid |
[90,91] |
|
Alpha1-AT |
Decreased in mice that underwent ovariectomy; could induce inflammation |
[92] |
|
Kallistatin |
Downregulation correlated with decrease in severity; increases ROS levels, modulates caspase 3 signaling and MnSOD |
[93] |
|
A2M |
Suppressed blastocyte and mouse embryo development |
[94] |
|
TIMP-1 |
Downregulated in endometriosis. In early endometriosis, no significant difference in expression |
[95,96] |
|
TIMP-2 |
Downregulated in advanced endometriosis |
[99] |
|
TIMP-3 |
Promoter regions changed significantly during proliferative phase |
[80] |
|
TIMP-4 |
Promoter regions changed significantly during proliferative phase |
[80] |
Transcription Factor Expression in Malignant and Benign Endometrial Diseases
Endometrial cancer
A variety of transcription factors have been found to significantly impact endometrial cancer through their regulation of gene expression. Firstly, Yang and colleagues evaluated the effect the knockdown of E2F1 has on EC, and it was discovered that it inhibited invasion and metastasis, thus allowing one to conclude that it regularly promotes a malignant phenotype (Table 10) [100]. Another study supports this conclusion, as it identified a strong correlation of E2F1 with EC [101]. This study additionally found that HMGA1 and PGR are closely correlated with EC. A different form of E2F1, E2F7, was highly expressed in EC, and its silencing may suppress the effect RAD51AP1 overexpression has on cell growth [102]. It is important to note that many endometrial-mesenchymal transition (EMT) proteins mentioned in an earlier section, such as Notch1 [103], SNAIL [104] and SLUG [105], are also transcription factors, and their expression, whether its elevated or lowered, plays a significant role in the development of endometrial cancer, adenomyosis, and endometriosis.
|
Transcription Factor |
Expression |
References |
|
E2F1 |
Knockdown inhibited invasion and migration Closely correlated with EC |
[100] [101] |
|
E2F7 |
Highly expressed. Silencing could suppress proliferative effects of RAD51AP1 |
[102] |
|
HMGA2 |
Upregulation associated with worse prognosis. It promoted proliferation, migration, invasion, and drug resistance |
[106] |
|
TBX2 |
Upregulated in EC; enhances cell resistance by regulation of NRF2; upregulates FSP1 to suppress ferroptosis |
[107,108] |
|
TCF19 |
Linked to worse prognosis |
[109] |
|
REST |
Downregulation linked to proliferation |
[110] |
|
FOXP1 |
Lowered expression in early secretory phase, and in nucleus and cytoplasm of grade 3 EC |
[111] |
|
FOXO1 |
Inhibits proliferation and migration; may target SREBP1 (it induces tumorigenesis) which was downregulated |
[112] |
|
FOXA1 |
Could be tumor suppressor because its downregulation promoted proliferation |
[113] |
|
HOXA10 |
Downregulation induced development of EC hyperplasia in animals |
[115] |
|
HOXB9 |
Upregulated in EC; linked to worse prognosis |
[116] |
|
KLF2 |
Expression inhibits EC proliferation by suppressing NPM1 |
[117] |
|
KLF12 |
Overexpressed in EC; increases proliferation and decreases apoptosis |
[118] |
|
KLF17 |
Induces EMT and cell invasion. Transactivates TWIST1 expression |
[119] |
|
ETV4 |
Controls EC development and ER signaling |
[120] |
|
ETV5 |
Overexpression leads to EMT; regulates NID1 and NUPR1 |
[121] |
|
SIRT1 |
Overexpression of SIRT1 leads to proliferation, migration, invasion, and an inhibition of apoptosis; enhances deacetylation of FOXO3 |
[114] |
|
ATF3 |
Downregulated; expression can inhibit proliferation, JunB expression, and promote apoptosis |
[122] |
|
ATF4 |
Overexpressed |
[123] |
|
NFAT5 |
Expression increases with grade |
[124] |
|
NF1C |
Inhibited proliferation, motility, and invasion of HECs |
[64] |
HMGA2 is a transcription factor known to promote cancer. One study observed its upregulation in endometrial cancer, which was linked to a worse prognosis, as its overexpression promoted proliferation, migration, invasion, and drug resistance. HMGA2 knockdown had the opposite effects [106]. Another transcription factor in which its upregulation was observed in EC is T-Box transcription factor (TBX2). Ding and colleagues discovered it enhanced cell resistance to chemotherapy by regulating the nuclear factor erythroid 2-related factor (NRF2) pathway [107]. It was additionally found to suppress ferroptosis by upregulating ferroptosis suppressor protein 1 (FSP1; [108]. Transcription factor 19 (TCF19), known to be differentially expressed in microsatellite stable and instable, was linked to a worse prognosis and immune exhaustion signature in endometrial cancer patients. It promoted tripartite motif-containing 14 (TRIM14) transcription, which induced tumorigenicity [109]. Repressor Element 1 (RE1) silencing transcription factor (REST), on the other hand, was downregulated in human EC samples. While it was highly expressed in the cell lines, the decrease in expression is linked to an increase in proliferation [110].
There are many significant transcription factors in endometrial cancer that are a part of the forkhead box family. To start, FOXP1, a P subfamily, was differently expressed in human samples based on the phase of the menstrual cycle and the grade of malignancy. There was a reduction in its immunostaining during the early secretory stage in the basalis [111]. Additionally, its expression was reduced in the nucleus and cytoplasm of grade 3 EC in comparison to grades 1 and 2. This study concluded that FOXP1 is specifically involved in estrogen dependent ECs as well. Next, the overexpression of forkhead transcription factor 1 (FOXO1) was observed to inhibit cell proliferation, migration, invasion, and tumorigenesis. In these cells, sterol regulatory element-binding protein 1 (SREBP1), another transcription factor, was downregulated, which may signify that FOXO1 targets SREBP1 [112]. SREBP1 is usually upregulated in EC and promotes tumorigenesis. Another transcription factor of the forkhead box family is forkhead box A1(FOXA1). When downregulated, Abe and colleagues observed that proliferation was promoted. Thus, FOXA1 could potentially be a tumor suppressor [113]. FOXO3 was found to be associated with SIRT1 [114]. SIRT1 induces FOXO3 deacetylation and activity. Overexpression of SIRT1 leads to proliferation, migration, invasion, and an inhibition of apoptosis. Two transcription factors of the homeobox gene family were found to play a role in EC. When HOXA10 was downregulated, endometrial hyperplasia began to develop in animals [115]. On the other hand, elevated levels of HOXB9 indicated a worse prognosis, as it was upregulated in EC and associated with histological grade and lymph node metastasis [116]. Two transcription factors of the Kruppel-like transcription family were detected. The expression of Kruppel-like factor 2 (KLF2) resulted in the in inhibition of EC proliferation through the suppression of nuclephosmin 1 (NPM1) transcription [117]. On the other hand, Krüppel-like factor 12 (KLF12) was found to be overexpressed in EC, which was associated with an increase in proliferation and tumor size, a decrease in apoptosis [118]. Additionally, Dong and colleagues demonstrated that when Krüppel-like factor 17 (KLF17), a transcription factor, induced EMT and cell invasion, the expression of TWIST1 increased [119]. This is because KLF17 transactivates TWIST1 expression by binding to its promoter. Interestingly, ETV4 has been found to control ER activity, a key oncogene of endometrial cancer, as well as the development of EC [120]. ETV5 is overexpressed in Hec1A EC cells and has been found to induce the EMT [121]. The study additionally discovered that ETV5 regulates the protein expression of Nidogen 1 (NID1) and Nuclear Protein 1 (NUPR1). Together, an invasive phenotype is achieved. Similarly, from the activating transcription factor family, ATF3 and ATF4 were detected. ATF3 was observed to be downregulated in endometrial cancer cells. When overexpressed, it inhibits proliferation, JunB expression, a transcription factor upregulated in EC and promotes apoptosis [122]. Liu and associates discovered that ATF4 is normally upregulated in EC cell lines, and its knockdown inhibits tumor growth [123]. Two transcription factors of the nuclear factor family were found to have different effects on endometrial cancer. Nuclear factor of activated T cells 5 (NFAT5) expression increased with grade [124], while Nuclear Factor 1-C (NF1C) inhibited the proliferation, motility, and invasion of human uterine endometrial cancer cells [63].
Adenomyosis
Compared to endometrial cancer, there is a paucity of work on transcription factors and adenomyosis. Expression of two zinc-finger transcription factors, GATA binding proteins, 2 and 6, were determined by immunohistochemistry. GATA2 had a two-fold lower expression while GATA6 a four-fold higher expression in ESCs of adenomyosis patients (Table 11) [125]. These levels impaired ESC decidualization. Another transcription factor that impairs decidualization is transcription factor 21 (TCF21). It achieves this by inhibiting decidual markers and cytoskeleton alterations [126]. STAT3 is a transcription factor known to play a significant role in endometrial regeneration. According to Hiraoka and colleagues, its activation promotes adenomyosis. In Stat3-deficient mice, a decrease in lesions and expression of genes associated with tissue regeneration were observed in c comparison to control mice (STAT3; [127]. In one study enacted by Li and colleagues, notably higher expressions of NF-κB DNA-binding activity, which was linked to dysmenorrhea severity, and p50 and p65 were detected [128]. It was concluded that NF-κB could be a transcription factor of great significance regarding the development of adenomyosis. Nie and colleagues additionally collected data signaling an elevation in expression of p50, p65, and p52, and a downregulation in progesterone receptor isoform B (PR-B) and a decrease in its immunoreactivity [129]. Lupicka and associates hypothesized that disturbances in stem cell differentiation of uterine tissue could be a main promoter of the development of adenomyosis. This is backed up by their data. There was a decrease in protein level of NANOG and SOX2 in stromal cells, while an elevation of OCT4 and SOX2 in the myometrial cells of dysfunctional uteri [130]. Two studies observed a decrease in mRNA and protein expression of HOXA10 [131]. Only the study conducted by Guo and colleagues observed a decrease in mRNA and protein expression of HOXA11 as well [132].
|
Transcription Factor |
Expression |
References |
|
GATA2 |
Low expression (H-SCORE) |
[125] |
|
GATA6 |
High expression (H-SCORE) |
[125] |
|
TCF21 |
Impairs decidualization |
[126] |
|
STAT3 |
Activation promotes adenomyosis; deficiency leads to decrease in lesions |
[127] |
|
NF-kB |
High DNA-binding activity, which was linked to dysmenorrhea severity |
[128] |
|
p50 |
High protein expression and immunoreactivity |
[128,129] |
|
p52 |
High immunoreactivity |
[129] |
|
p65 |
High protein expression and immunoreactivity |
[128,129] |
|
PR-B |
Downregulated and decrease in immunoreactivity |
[129] |
|
NANOG |
Protein level decreased in stromal cells |
[130] |
|
SOX2 |
Protein level decreased in stromal cells, but increase in myometrial cells |
[130] |
|
OCT4 |
Increased protein level in myometrial cells |
[130] |
|
HOXA10 |
Decrease in expression |
[130,131] |
|
HOXA11 |
Decrease in expression |
[132] |
Endometriosis
In deep infiltrating endometriosis (DIE), Ganieva and associates detected a significant amount of TCF21 expression in comparison to normal tissues. Its expression increased with severity of the disease (Table 12), and it was also determined to play a role in the regulation of fibrosis in endometriosis [133]. Additionally, Wu and colleagues determined that TCF12 plays a role in the pathogenesis of endometriosis, as it has a higher expression in endometriotic than endometrial stromal cells, a change in its expression significantly impacted the proliferation and invasion of ESCs, and also affected the binding activities of other transcription factors like USF2, MMP9, and SF-1 [134]. Ntzeros and associates observed a difference in ZEB1 expression in the endometriotic cysts of endometrioma cases with and without DIE, which could reflect differences in their pathogenetic mechanisms [135]. Another study determined that ZEB1 could be linked to an invasive type of endometriosis. Its expression was only detected in the epithelial of endometriotic lesions, not of normal endometria, and it was most often found in the epithelial cells of invasive endometriosis [136]. AP-1 is another transcription factor that may aid in the development of invasive endometriosis, as its expression has been shown in multiple models, and it promotes invasive phenotypes [137]. There was a high activation of transcription factor nuclear factor-kappa B (NK-kappaB) in red, or active, endometriotic lesions [138]. p50 is also a part of the NF-kappaB family. In p50 knockdown mice, the size of the endometrial implants greatly decreased while NF-kappaB activation was interfered with. Additionally, NF-YA, NF-YB, and NF-YC are additional nuclear factors with possible significant roles in this disease. NF-YA expression was found to be upregulated during the proliferative phase in eutopic endometrium. In ectopic endometrium, NF-YB and NF-YC were also upregulated [139]. Amirteimouri and colleagues suggested that because of NF-Y’s regulation of ID genes, it may be behind the epigenetic changes that occur in endometrial tissues. In severe endometriosis, in which patients experienced intense pain, Badary and colleagues found significant expression through immunohistochemistry of HIF-1a [140]. Early growth response 1 (EGR) is another transcription factor detected in endometriosis tissues. It was determined to induce proliferation, migration, and invasion [141]. Transcription factors of the GATA family have been found to regulate key genes that impact endometriosis. GATA2 regulates genes important for the differentiation of stromal cells [142]. It is repressed in endometriotic cells. GATA6 on the other hand, is upregulated in endometriotic cells, inhibits GATA2 and promotes endometriotic markers. Likewise, GATA-3 promotes proliferation. In a study by Chen and colleagues, cells where GATA-3 was introduced had higher proliferation rates than those without GATA-3 [143]. The cells in which it was suppressed had lower proliferation rates than others. In Ishikawa cells, SOX18 overexpression was found to induce EMT, increase proliferation, as well as progress endometriosis overtime through the growth and bleeding of tissue outside of the uterus [144]. KLF11, a gene known to be associated with disease in humans, was found to be under expressed in endometriotic tissues of humans. When Klf11 was knocked out in mice, their lesions were larger and resembled the lesions of humans with late-stage diseases. In comparison, Klf9 knockout had no significant result on the lesions of the animals [145]. According to Chen and colleagues, transcription factor EB (TFEB) is highly expressed in the endometrium. It improves the proliferative abilities of human endometrial stromal cells (HESCs). In rat models, TFEB knockdown suppresses the growth of lesions [146]. OCT4 is a transcription factor known to be an essential to upholding a tissue’s undifferentiated state [147]. Chang and colleagues discovered that in endometriotic tissues, the expression of OCT4 mRNA, which was upregulated in ectopic endometriotic tissues, was associated with genes that regulate migration in the cell [148]. In women who ultimately develop endometriosis, an increase in inflammation in eutopic and ectopic endometrium is observed, which can now be linked to a disruption in the homeostasis of regulatory T cells (Treg). BACH2, a transcription factor that regulates Treg genes, is upregulated in these tissues, indicating an iron-heme overload response [149]. Additionally, the overexpression of ATF4 has been found to allow cells to resist ferroptosis that is caused by an iron overload [150]. In a comparison between chicken ovalbumin upstream promoter transcription factor (COUP-TF) and steroidogenic factor-1 (SF-1), where antibodies were employed against both, it was found that in eutopic endometrial cells, only COUP-TF bound to the nuclear receptor half-site (NRHS), while both COUP-TF and SF-1 transcripts were detected in endometriotic tissues [151]. The researchers concluded that as SF-1 expression is undetected in eutopic endometrial stromal cells, one of the transcription factors that causes the inhibition of aromatase P450 is COUP-TF. However, in endometriotic stromal cells, SF-1 can compete for the same DNA-binding site, thus stopping the inhibition caused by COUP-TF. Lastly, among the transcription factors investigated in endometriosis pathophysiology, perhaps the most-well studied is HOXA10. Lazim and colleagues recently published a systematic review on the topic [152]. Briefly, the majority of data on this topic would support the notion that sufficient data are provided to support the notion that women with endometriosis express low levels of HOXA10 compared to women without endometriosis. More specifically, low levels of eutopic endometrial HOXA10 expression was associated with infertility which may be due to altered endometrial receptivity, decidualization and/or embryo implantation failure. With respect to ectopic, endometriotic lesion tissue, HOXA10 expression is also reduced compared to eutopic endometrial tissue.
In summary, there is a vast amount of literature on transcription factor expression in each of the three diseases but little consistency among the diseases. Among those transcription factors, HOXA10 has been reported to be reduced in all three conditions, with most reports on endometriosis. The bulk of the literature reports GATA2 and GATA6 expression in endometriosis and to a lesser extent adenomyosis.
|
Transcription Factor |
Expression |
References |
|
TCF21 |
Highly expression in DIE; increases with severity of disease |
[133,134] |
|
ZEB1 |
Indicator of invasive endometriosis |
[135,136] |
|
EGR1 |
Induces proliferation, migration, and invasion |
[141] |
|
GATA2 |
Repressed in endometriosis; vital to regulation of genes that control differentiation of stromal cells |
[142] |
|
GATA-3 |
Promotes proliferation |
[142] |
|
GATA6 |
Upregulated in endometriosis; inhibits GATA2 |
[142] |
|
NK-kappaB |
High activation in red (active) lesions |
[138] |
|
NF-YA, B, & C |
Upregulated |
[139] |
|
HIF-1alpha |
Significant expression in severe endometriosis |
[140] |
|
KLF11 |
Under expressed in human lesions, knockout can cause proliferative lesions in mice |
[145] |
|
TFEB |
Highly expressed – knockdown suppressions lesion growth |
[146] |
|
OCT4 |
Maintains undifferentiated state in tissues; upregulated in endometriosis; expression correlated with genes that control migration |
[147] [148] |
|
BACH2 |
Regulates Treg genes; upregulated |
[149] |
|
ATF4 |
Expression allows cells to resist ferroptosis |
[150] |
|
AP-1 |
Promotes invasive phenotype |
[137] |
|
p50 |
Part of NF-kappaB family – p50 knockdown decreased size of implants; there was disruption in NF-kappaB activation |
[138] |
|
SOX18 |
Upregulated – induces EMT, increase invasion and migration, deteriorates endometriosis |
[144] |
|
COUP-TF |
Detected in both eutopic and endometriotic cells; might inhibit aromatase P450 in eutopic endometrial cells |
[151] |
|
SF-1 |
Transcription undetected in eutopic endometrial cells; can compete for same DNA-binding site to prevent inhibition caused by COUP-TF in endometriotic stromal cells |
[151] |
|
HOXA10 |
Reduced expression in both eutopic and ectopic tissue; reduced eutopic endometrial expression associated with infertility |
[152] |
Summary
Endometrial cancer, adenomyosis and endometriosis are diseases which arise from the endometrial lining of the uterus. Each disease is characterized by augmented cell proliferation/survival, migration and invasion. Augmented estrogen action and reduced progesterone signaling is postulated to contribute these common cellular attributes among these diseases. Expression of estrogen and progesterone receptors appear to display similar patterns among the three conditions but there is less consistency in assessment of epithelial-to-mesenchymal transition proteins (such as ZEB2, EZH2) among the three diseases. With respect to EMT protein assessment, there is a larger body of work and consistency in patterns of expression for key proteins such as CDH1, SLUG, SNAIL and ZEB1 between endometrial cancer and endometriosis. As all three of these diseases exhibit differing degrees of invasiveness, which is linked to tissue remodeling, there is some consistency with respect to patterns of expression for proteases such as MMP-2 and MMP-9 which is elevated in all three diseases. With respect to protease inhibitors, TIMP-1 expression is reduced in all three diseases, while TIMP-2 expression is reduced in endometrial cancer and endometriosis. While many studies have evaluated expression of different transcription factors in endometrial cancer, adenomyosis, and endometriosis, few studies have assessed the same transcription factors. Thus, there are several factors associated with endometrial cancer, adenomyosis and endometriosis, most notably altered steroid receptor expression and imbalances in the MMP/TIMP systems.
Limitations
While the studies cited in this review provide important information on pathways associated with endometrial cancer, adenomyosis and endometrial cancer pathophysiology, there are limitations in the approaches and study designs. For example, the majority of the studies which have examined EMT markers, proteases/protease inhibitors and/or transcription factors, many have been descriptive with few studies concurrently assessing expression/localization and function using in vitro assays. This is extremely important especially when studying the protease/protease inhibitors. Immunohistochemical localization and Western blot analysis of protease/protease inhibitors do not provide information on function of the proteases/proteases inhibitors. Further, few studies concurrently evaluate protease and respective protease inhibitor expression and/or ratio. Often both protease and protease inhibitor may be expressed, and it is an alteration that favors net protease activity which leads to tissue invasion/remodeling. Additionally, greater emphasis on patient demographics, inclusion, exclusion criteria, stage of disease (for endometrial cancer and endometriosis) and type of lesion (for endometriosis) must be taken into account. This in turn should minimize variability and heterogeneity of experimental outcomes allowing for more robust data.
Future Studies
Future studies may focus on further dissecting the intermediates (transcription factors) associated with impaired steroid signaling and altered MMP/TIMP expression. Clearly, compared to endometrial cancer and endometriosis, adenomyosis is the least explored of the three diseases. As adenomyosis diagnosis is increasing, more detailed examination of this disease is warranted. When embarking upon future studies, it will be essential to take into account the aforementioned limitations to increase experimental rigor and reproducibility. In turn, we anticipate this will more clearly align the similarities and differences among these three common diseases in women.
Authors Contribution Statement
YA and WBN wrote and edited the review article.
Conflict of Interest
The authors declare no conflict of interest.
Data Availability Statement
All data generated and included in this manuscript are available upon the request of the corresponding author.
Funding
There was no funding associated with the work described in this review.
References
2. Felix AS, Weissfeld JL, Stone RA, Bowser R, Chivukula M, Edwards RP, et al. Factors associated with Type I and Type II endometrial cancer. Cancer Causes Control. 2010;21(11):1851–6.
3. Dias Da Silva I, Wuidar V, Zielonka M, Pequeux C. Unraveling the Dynamics of Estrogen and Progesterone Signaling in the Endometrium: An Overview. Cells. 2024;13(15):1236.
4. Yu K, Huang ZY, Xu XL, Li J, Fu XW, Deng SL. Estrogen Receptor Function: Impact on the Human Endometrium. Front Endocrinol (Lausanne). 2022;13:827724.
5. Zhang Y, Zhao D, Gong C, Zhang F, He J, Zhang W, et al. Prognostic role of hormone receptors in endometrial cancer: a systematic review and meta-analysis. World J Surg Oncol. 2015;13:208.
6. van Weelden WJ, Reijnen C, Küsters-Vandevelde HVN, Bulten J, Bult P, Leung S, et al. The cutoff for estrogen and progesterone receptor expression in endometrial cancer revisited: a European Network for Individualized Treatment of Endometrial Cancer collaboration study. Hum Pathol. 2021;109:80–91.
7. Di Nezza LA, Jobling T, Salamonsen LA. Progestin suppresses matrix metalloproteinase production in endometrial cancer. Gynecol Oncol. 2003;89(2):325–33.
8. Fujimoto J, Sato E. Clinical implication of estrogen-related receptor (ERR) expression in uterine endometrial cancers. J Steroid Biochem Mol Biol. 2009;116(1-2):71–5.
9. Jeon YT, Park IA, Kim YB, Kim JW, Park NH, Kang SB, et al. Steroid receptor expressions in endometrial cancer: clinical significance and epidemiological implication. Cancer Lett. 2006;239(2):198–204.
10. Busch EL, Crous-Bou M, Prescott J, Chen MM, Downing MJ, Rosner BA, et al. Endometrial Cancer Risk Factors, Hormone Receptors, and Mortality Prediction. Cancer Epidemiol Biomarkers Prev. 2017;26(5):727–35.
11. Tamaya T, Motoyama T, Ohono Y, Ide N, Tsurusaki T, Okada H. Steroid receptor levels and histology of endometriosis and adenomyosis. Fertil Steril. 1979;31(4):396–400.
12. Wei JJ, Chiriboga L, Mittal K. Expression profile of the tumorigenic factors associated with tumor size and sex steroid hormone status in uterine leiomyomata. Fertil Steril. 2005;84(2):474–84.
13. Donnez J, Nisolle M, Smoes P, Gillet N, Beguin S, Casanas-Roux F. Peritoneal endometriosis and "endometriotic" nodules of the rectovaginal septum are two different entities. Fertil Steril. 1996;66(3):362-8.
14. Sztachelska M, Ponikwicka-Tyszko D, Martínez-Rodrigo L, Bernaczyk P, Palak E, Półchłopek W, et al. Functional Implications of Estrogen and Progesterone Receptors Expression in Adenomyosis, Potential Targets for Endocrinological Therapy. J Clin Med. 2022;11(15):4407.
15. Nie J, Yi Y, Liu X, Guo SW. Progressively diminished estrogen signaling concordant with increased fibrosis in ectopic endometrium. Hum Reprod Open. 2025;2025(3):hoaf028.
16. Mehasseb MK, Panchal R, Taylor AH, Brown L, Bell SC, Habiba M. Estrogen and progesterone receptor isoform distribution through the menstrual cycle in uteri with and without adenomyosis. Fertil Steril. 2011;95(7): 2228-35, 2235.e1.
17. Nisolle M, Casanas-Roux F, Donnez J. Immunohistochemical analysis of proliferative activity and steroid receptor expression in peritoneal and ovarian endometriosis. Fertil Steril. 1997;68(5):912–9.
18. Pluchino N, Mamillapalli R, Wenger JM, Ramyead L, Drakopoulos P, Tille JC, et al. Estrogen receptor-α immunoreactivity predicts symptom severity and pain recurrence in deep endometriosis. Fertil Steril. 2020;113(6):1224–31.e1.
19. Fazleabas AT, Brudney A, Chai D, Langoi D, Bulun SE. Steroid receptor and aromatase expression in baboon endometriotic lesions. Fertil Steril. 2003;80 Suppl 2:820–7.
20. Lin K, Ma J, Wu R, Zhou C, Lin J. Influence of ovarian endometrioma on expression of steroid receptor RNA activator, estrogen receptors, vascular endothelial growth factor, and thrombospondin 1 in the surrounding ovarian tissues. Reprod Sci. 2014;21(2):183–9.
21. Tanaka Y, Terai Y, Kawaguchi H, Fujiwara S, Yoo S, Tsunetoh S, et al. Prognostic impact of EMT (epithelial-mesenchymal-transition)-related protein expression in endometrial cancer. Cancer Biol Ther. 2013;14(1):13–9.
22. Zhou W, Wang K, Wang J, Qu J, Du G, Zhang Y. SOX17 Inhibits Tumor Metastasis Via Wnt Signaling In Endometrial Cancer. Onco Targets Ther. 2019;12:8275–86.
23. Xiao YY, Lin L, Li YH, Jiang HP, Zhu LT, Deng YR, et al. ZEB1 promotes invasion and metastasis of endometrial cancer by interacting with HDGF and inducing its transcription. Am J Cancer Res. 2019;9(11):2314–30.
24. Sadłecki P, Jóźwicki J, Antosik P, Walentowicz-Sadłecka M. Expression of Selected Epithelial-Mesenchymal Transition Transcription Factors in Endometrial Cancer. Biomed Res Int. 2020;2020:4584250.
25. Liu G, Zhang L, Zhou X, Xue J, Xia R, Gan X, et al. Inducing the "re-development state" of periodontal ligament cells via tuning macrophage mediated immune microenvironment. J Adv Res. 2024;60:233–48.
26. Abouhashem NS, Ibrahim DA, Mohamed AM. Prognostic implications of epithelial to mesenchymal transition related proteins (E-cadherin, Snail) and hypoxia inducible factor 1α in endometrioid endometrial carcinoma. Ann Diagn Pathol. 2016;22:1–11.
27. Chen Z, Che Q, He X, Wang F, Wang H, Zhu M, et al. Stem cell protein Piwil1 endowed endometrial cancer cells with stem-like properties via inducing epithelial-mesenchymal transition. BMC Cancer. 2015;15:811.
28. Zheng D, Duan H, Wang S, Xu Q, Gan L, Li J, et al. FAK regulates epithelial‑mesenchymal transition in adenomyosis. Mol Med Rep. 2018;18(6):5461–72.
29. Qi S, Zhao X, Li M, Zhang X, Lu Z, Yang C, et al. Aberrant expression of Notch1/numb/snail signaling, an epithelial mesenchymal transition related pathway, in adenomyosis. Reprod Biol Endocrinol. 2015;13:96.
30. Nguyen HPT, Xiao L, Deane JA, Tan KS, Cousins FL, Masuda H, et al. N-cadherin identifies human endometrial epithelial progenitor cells by in vitro stem cell assays. Hum Reprod. 2017;32(11):2254–68.
31. Yoo JY, Ku BJ, Kim TH, Il Ahn J, Ahn JY, Yang WS, et al. β-catenin activates TGF-β-induced epithelial-mesenchymal transition in adenomyosis. Exp Mol Med. 2020;52(10):1754–65.
32. Yoo JY, Ku BJ, Kim TH, Il Ahn J, Ahn JY, Yang WS, et al. β-catenin activates TGF-β-induced epithelial-mesenchymal transition in adenomyosis. Exp Mol Med. 2020;52(10):1754–65.
33. Wang LN, Ren L, Li L, Liu SL, Lu HJ, Guo ML, et al. Role of SMOC2 in adenomyosis: implications for ECM remodeling and EMT pathogenesis. BMC Womens Health. 2025;25(1):155.
34. Hu R, Peng GQ, Ban DY, Zhang C, Zhang XQ, Li YP. High-Expression of Neuropilin 1 Correlates to Estrogen-Induced Epithelial-Mesenchymal Transition of Endometrial Cells in Adenomyosis. Reprod Sci. 2020;27(1):395–403.
35. Guo Y, Wang J, Jia C, Liao Y. SKP2 regulates ZEB1 expression and stimulates eutopic endometrial stromal cell invasion and proliferation of adenomyosis. Reprod Biol. 2022;22(1):100578.
36. Gaetje R, Kotzian S, Herrmann G, Baumann R, Starzinski-Powitz A. Nonmalignant epithelial cells, potentially invasive in human endometriosis, lack the tumor suppressor molecule E-cadherin. Am J Pathol. 1997;150(2):461–7.
37. Wang M, Wu Y, He Y, Liu J, Chen Y, Huang J, et al. SIRT1 upregulation promotes epithelial-mesenchymal transition by inducing senescence escape in endometriosis. Sci Rep. 2022;12(1):12302.
38. Zhang Q, Dong P, Liu X, Sakuragi N, Guo SW. Enhancer of Zeste homolog 2 (EZH2) induces epithelial-mesenchymal transition in endometriosis. Sci Rep. 2017;7(1):6804.
39. Huang Q, Song Y, Lei X, Huang H, Nong W. MMP-9 as a clinical marker for endometriosis: a meta-analysis and bioinformatics analysis. Front Endocrinol (Lausanne). 2024;15:1475531.
40. Proestling K, Birner P, Gamperl S, Nirtl N, Marton E, Yerlikaya G, et al. Enhanced epithelial to mesenchymal transition (EMT) and upregulated MYC in ectopic lesions contribute independently to endometriosis. Reprod Biol Endocrinol. 2015;13:75.
41. Xiong W, Zhang L, Liu H, Li N, Du Y, He H, et al. E(2) -mediated EMT by activation of β-catenin/Snail signalling during the development of ovarian endometriosis. J Cell Mol Med. 2019;23(12):8035–45.
42. Wu RF, Chen ZX, Zhou WD, Li YZ, Huang ZX, Lin DC, et al. High expression of ZEB1 in endometriosis and its role in 17β-estradiol-induced epithelial-mesenchymal transition. Int J Clin Exp Pathol. 2018;11(10):4744–58.
43. Huang Y, Jiang P, Chen Y, Wang J, Yuan R. Systemic analysis of the expression and prognostic significance of USP31 in endometrial cancer. Biomol Biomed. 2023;23(3):426–36.
44. Mullany SA, Moslemi-Kebria M, Rattan R, Khurana A, Clayton A, Ota T, et al. Expression and functional significance of HtrA1 loss in endometrial cancer. Clin Cancer Res. 2011;17(3):427–36.
45. Bao W, Fan Q, Luo X, Cheng WW, Wang YD, Li ZN, et al. Silencing of Cathepsin B suppresses the proliferation and invasion of endometrial cancer. Oncol Rep. 2013;30(2):723–30.
46. Devetzi M, Scorilas A, Tsiambas E, Sameni M, Fotiou S, Sloane BF, et al. Cathepsin B protein levels in endometrial cancer: Potential value as a tumour biomarker. Gynecol Oncol. 2009;112(3):531–6.
47. Lösch A, Kohlberger P, Gitsch G, Kaider A, Breitenecker G, Kainz C. Lysosomal protease cathepsin D is a prognostic marker in endometrial cancer. Br J Cancer. 1996;73(12):1525–8.
48. Falcón O, Chirino R, León L, López-Bonilla A, Torres S, Fernández L, et al. Low levels of cathepsin D are associated with a poor prognosis in endometrial cancer. Br J Cancer. 1999;79(3-4):570–6.
49. Skrzypczak M, Springwald A, Lattrich C, Häring J, Schüler S, Ortmann O, et al. Expression of cysteine protease cathepsin L is increased in endometrial cancer and correlates with expression of growth regulatory genes. Cancer Invest. 2012;30(5):398–403.
50. Cormio A, Musicco C, Gasparre G, Cormio G, Pesce V, Sardanelli AM, et al. Increase in proteins involved in mitochondrial fission, mitophagy, proteolysis and antioxidant response in type I endometrial cancer as an adaptive response to respiratory complex I deficiency. Biochem Biophys Res Commun. 2017;491(1):85–90.
51. Matsuo T, Nakamura K, Takamoto N, Kodama J, Hongo A, Abrzua F, et al. Expression of the serine protease hepsin and clinical outcome of human endometrial cancer. Anticancer Res. 2008;28(1a):159–64.
52. Nakamura K, Takamoto N, Abarzua F, Hongo A, Kodama J, Nasu Y, et al. Hepsin inhibits the cell growth of endometrial cancer. Int J Mol Med. 2008;22(3):389–97.
53. Wang Y, Zheng L, Zhang L, Tai Y, Lin X, Cai Z. Roles of MMP-2 and MMP-9 and their associated molecules in the pathogenesis of keloids: a comprehensive review. Front Pharmacol. 2024;15:1444653.
54. Aglund K, Rauvala M, Puistola U, Angström T, Turpeenniemi-Hujanen T, Zackrisson B, et al. Gelatinases A and B (MMP-2 and MMP-9) in endometrial cancer-MMP-9 correlates to the grade and the stage. Gynecol Oncol. 2004;94(3):699–704.
55. Ren F, Wang D, Wang Y, Chen P, Guo C. SPOCK2 Affects the Biological Behavior of Endometrial Cancer Cells by Regulation of MT1-MMP and MMP2. Reprod Sci. 2020;27(7):1391–9.
56. Wang X, Wei Z, Tang Z, Xue C, Yu H, Zhang D, et al. IL-37bΔ1-45 suppresses the migration and invasion of endometrial cancer cells by targeting the Rac1/NF-κB/MMP2 signal pathway. Lab Invest. 2021;101(6):760–74.
57. Ruan H, Liang X, Zhao W, Ma L, Zhao Y. The effects of microRNA-183 promots cell proliferation and invasion by targeting MMP-9 in endometrial cancer. Biomed Pharmacother. 2017;89:812–8.
58. Zhao Y, Zou X, Wang G, Liu Y, Zhang C, Lu W, et al. Effects of GATA6-AS/MMP9 on malignant progression of endometrial carcinoma. J buon. 2021;26(5):1789–95.
59. Bode W, Fernandez-Catalan C, Grams F, Gomis-Rüth FX, Nagase H, Tschesche H, et al. Insights into MMP-TIMP interactions. Ann N Y Acad Sci. 1999;878:73–91.
60. Yıldırım ME, Karakuş S, Kurtulgan HK, Kılıçgün H, Erşan S, Bakır S. The Association of Plasminogen Activator Inhibitor Type 1 (PAI-1) Level and PAI-1 4G/5G Gene Polymorphism with the Formation and the Grade of Endometrial Cancer. Biochem Genet. 2017;55(4):314–21.
61. Yang GD, Yang XM, Lu H, Ren Y, Ma MZ, Zhu LY, et al. SERPINA3 promotes endometrial cancer cells growth by regulating G2/M cell cycle checkpoint and apoptosis. Int J Clin Exp Pathol. 2014;7(4):1348–58.
62. Li X, Gao Y, Tan M, Zhuang H, Gao J, Hu Z, et al. Expression of HE4 in Endometrial Cancer and Its Clinical Significance. Biomed Res Int. 2015;2015:437468.
63. Li J, Chen H, Mariani A, Chen D, Klatt E, Podratz K, et al. HE4 (WFDC2) Promotes Tumor Growth in Endometrial Cancer Cell Lines. Int J Mol Sci. 2013;14(3):6026–43.
64. Sierko E, Zabrocka E, Ostrowska-Cichocka K, Tokajuk P, Zimnoch L, Wojtukiewicz MZ. Co-localization of Coagulation Factor X and its Inhibitory System, PZ/ZPI, in Human Endometrial Cancer Tissue. In Vivo. 2019;33(3):771–6.
65. Nakamura K, Hongo A, Kodama J, Hiramatsu Y. The role of hepatocyte growth factor activator inhibitor (HAI)-1 and HAI-2 in endometrial cancer. Int J Cancer. 2011;128(11):2613–24.
66. Shen Y, Hu HY, Lu L. [Mechanism of bortezomib in inducing apoptosis and improving chemosensitivity of Ishikawa cells]. Nan Fang Yi Ke Da Xue Xue Bao. 2010 Jun;30(6):1301-3, 1309.
67. Gershtein ES, Mushtenko SV, Ermilova VD, Levchenko NE, Kushlinskii NE. Matrix Metalloproteinases and Their Tissue Inhibitors in Blood Serum of Patients with Endometrial Cancer: Clinical and Morphological Correlations. Bull Exp Biol Med. 2018;165(1):75–9.
68. Honkavuori M, Talvensaari-Mattila A, Puistola U, Turpeenniemi-Hujanen T, Santala M. High serum TIMP-1 is associated with adverse prognosis in endometrial carcinoma. Anticancer Res. 2008;28(5a):2715–9.
69. Yamanaka A, Kimura F, Yoshida T, Kita N, Takahashi K, Kushima R, et al. Dysfunctional coagulation and fibrinolysis systems due to adenomyosis is a possible cause of thrombosis and menorrhagia. Eur J Obstet Gynecol Reprod Biol. 2016;204:99–103.
70. Mori T, Yamasaki S, Masui F, Matsuda M, Sasabe H, Zhou YF. Suppression of the development of experimentally induced uterine adenomyosis by a novel matrix metalloproteinase inhibitor, ONO-4817, in mice. Exp Biol Med (Maywood). 2001;226(5):429–33.
71. Tokyol C, Aktepe F, Dilek FH, Sahin O, Arioz DT. Expression of cyclooxygenase-2 and matrix metalloproteinase-2 in adenomyosis and endometrial polyps and its correlation with angiogenesis. Int J Gynecol Pathol. 2009;28(2):148–56.
72. Li T, Li YG, Pu DM. Matrix metalloproteinase-2 and -9 expression correlated with angiogenesis in human adenomyosis. Gynecol Obstet Invest. 2006;62(4):229–35.
73. Yang JH, Wu MY, Chen MJ, Chen SU, Yang YS, Ho HN. Increased matrix metalloproteinase-2 and tissue inhibitor of metalloproteinase-1 secretion but unaffected invasiveness of endometrial stromal cells in adenomyosis. Fertil Steril. 2009;91(5 Suppl):2193–8.
74. Kogan EA, Unanian AL, Demura TA, Grechukhina OM, Sidorova IS, Kiselev VI. Clinical and morphological parallels and molecular aspects of the morphogenesis of adenomyosis. Arkh Patol. 2008;70(5):8–12.
75. Porter KM, Wieser FA, Wilder CL, Sidell N, Platt MO. Cathepsin Protease Inhibition Reduces Endometriosis Lesion Establishment. Reprod Sci. 2016;23(5):623–9.
76. Grzywa R, Gorodkiewicz E, Burchacka E, Lesner A, Laudański P, Łukaszewski Z, et al. Determination of cathepsin G in endometrial tissue using a surface plasmon resonance imaging biosensor with tailored phosphonic inhibitor. Eur J Obstet Gynecol Reprod Biol. 2014;182:38–42.
77. Liu H, Jiang Y, Jin X, Zhu L, Shen X, Zhang Q, et al. CAPN 7 promotes the migration and invasion of human endometrial stromal cell by regulating matrix metalloproteinase 2 activity. Reprod Biol Endocrinol. 2013;11:64.
78. Hirota Y, Osuga Y, Hirata T, Yoshino O, Koga K, Harada M, et al. Possible involvement of thrombin/protease-activated receptor 1 system in the pathogenesis of endometriosis. J Clin Endocrinol Metab. 2005;90(6):3673–9.
79. Kusama K, Fukushima Y, Yoshida K, Azumi M, Yoshie M, Mizuno Y, et al. PGE2 and Thrombin Induce Myofibroblast Transdifferentiation via Activin A and CTGF in Endometrial Stromal Cells. Endocrinology. 2021;162(12):bqab207.
80. Tang L, Xiang Y, Zhou Y, Mu J, Zai M, Xing Q, et al. The DNA methylation status of genes encoding Matrix metalloproteinases and tissue inhibitors of Matrix metalloproteinases in endometriosis. Mol Reprod Dev. 2018;85(1):17–25.
81. Chen CH, Miller MA, Sarkar A, Beste MT, Isaacson KB, Lauffenburger DA, et al. Multiplexed protease activity assay for low-volume clinical samples using droplet-based microfluidics and its application to endometriosis. J Am Chem Soc. 2013;135(5):1645–8.
82. Malvezzi H, Aguiar VG, Paz CC, Tanus-Santos JE, Penna IA, Navarro PA. Increased circulating MMP-2 levels in infertile patients with moderate and severe pelvic endometriosis. Reprod Sci. 2013;20(5):557–62.
83. Collette T, Bellehumeur C, Kats R, Maheux R, Mailloux J, Villeneuve M, et al. Evidence for an increased release of proteolytic activity by the eutopic endometrial tissue in women with endometriosis and for involvement of matrix metalloproteinase-9. Hum Reprod. 2004;19(6):1257–64.
84. Gaetje R, Holtrich U, Engels K, Kourtis K, Cikrit E, Kissler S, et al. Expression of membrane-type 5 matrix metalloproteinase in human endometrium and endometriosis. Gynecol Endocrinol. 2007;23(10):567–73.
85. Kokorine I, Nisolle M, Donnez J, Eeckhout Y, Courtoy PJ, Marbaix E. Expression of interstitial collagenase (matrix metalloproteinase-1) is related to the activity of human endometriotic lesions. Fertil Steril. 1997;68(2):246–51.
86. Gao X, Liu L, Liu X, Shen Y, Fan Y. Study on the mechanism of USP7 promoting endometriosis by regulating DNMT1 deubiquitination level. Biochem Cell Biol. 2025;103:1–14.
87. Chen Q, Hang Y, Zhang T, Tan L, Li S, Jin Y. USP10 promotes proliferation and migration and inhibits apoptosis of endometrial stromal cells in endometriosis through activating the Raf-1/MEK/ERK pathway. Am J Physiol Cell Physiol. 2018;315(6):C863–72.
88. Goteri G, Altobelli E, Tossetta G, Zizzi A, Avellini C, Licini C, et al. High temperature requirement A1, transforming growth factor beta1, phosphoSmad2 and Ki67 in eutopic and ectopic endometrium of women with endometriosis. Eur J Histochem. 2015;59(4):2570.
89. Fazleabas AT, Khan-Dawood FS, Dawood MY. Protein, progesterone, and protease inhibitors in uterine and peritoneal fluids of women with endometriosis. Fertil Steril. 1987;47(2):218–24.
90. Suzumori N, Sato M, Yoneda T, Ozaki Y, Takagi H, Suzumori K. Expression of secretory leukocyte protease inhibitor in women with endometriosis. Fertil Steril. 1999;72(5):857–67.
91. Shimoya K, Moriyama A, Ogata I, Nobunaga T, Koyama M, Azuma C, et al. Increased concentrations of secretory leukocyte protease inhibitor in peritoneal fluid of women with endometriosis. Mol Hum Reprod. 2000;6(9):829–34.
92. Tamura K, Takashima H, Fumoto K, Kajihara T, Uchino S, Ishihara O, et al. Possible Role of α1-Antitrypsin in Endometriosis-Like Grafts From a Mouse Model of Endometriosis. Reprod Sci. 2015;22(9):1088–97.
93. Mao Y, Qiao JD, Chen S, Zhou X, Wang Z, Cai S, et al. Kallistatin Inhibits Anoikis Resistance and Metastasis of Ectopic Endometrium Cells by Modulating MnSOD and Caspase 3 Signaling. Reprod Sci. 2021;28(4):1012–9.
94. Sayegh RA, Tao XJ, Leykin L, Isaacson KB. Endometrial alpha-2 macroglobulin; localization by in situ hybridization and effect on mouse embryo development in vitro. J Clin Endocrinol Metab. 1997;82(12):4189–95.
95. Muharam R, Bowolaksono A, Maidarti M, Febri RR, Mutia K, Iffanolida PA, et al. Elevated MMP-9, Survivin, TGB1 and Downregulated Tissue Inhibitor of TIMP-1, Caspase-3 Activities are Independent of the Low Levels miR-183 in Endometriosis. Int J Womens Health. 2024;16:1733–42.
96. Nanda A, K T, Banerjee P, Dutta M, Wangdi T, Sharma P, et al. Cytokines, Angiogenesis, and Extracellular Matrix Degradation are Augmented by Oxidative Stress in Endometriosis. Ann Lab Med. 2020 Sep;40(5):390–7.
97. Madjid TH, Ardiansyah DF, Permadi W, Hernowo B. Expression of Matrix Metalloproteinase-9 and Tissue Inhibitor of Metalloproteinase-1 in Endometriosis Menstrual Blood. Diagnostics (Basel). 2020;10(6):364.
98. Szymanowski K, Mikołajczyk M, Wirstlein P, Dera-Szymanowska A. Matrix metalloproteinase-2 (MMP-2), MMP-9, tissue inhibitor of matrix metalloproteinases (TIMP-1) and transforming growth factor-β2 (TGF-β2) expression in eutopic endometrium of women with peritoneal endometriosis. Ann Agric Environ Med. 2016;23(4):649–53.
99. Jana S, Chatterjee K, Ray AK, DasMahapatra P, Swarnakar S. Regulation of Matrix Metalloproteinase-2 Activity by COX-2-PGE2-pAKT Axis Promotes Angiogenesis in Endometriosis. PLoS One. 2016;11(10):e0163540.
100. Yang X, Cheng Y, Li X, Zhou J, Dong Y, Shen B, et al. A Novel Transcription Factor-Based Prognostic Signature in Endometrial Cancer: Establishment and Validation. Onco Targets Ther. 2021;14:2579–98.
101. Song Y, Chen QT, He QQ. Identification of key transcription factors in endometrial cancer by systems bioinformatics analysis. J Cell Biochem. 2019;120(9):15443–54.
102. Huang X, Wu Z, Xiao C, Chen XI. E2F7/RAD51AP1 Axis Inhibits Endometrial Cancer Sensitivity to 5-FU via the Fatty Acid Metabolic Pathway. Anticancer Res. 2023;43(11):4905–14.
103. Wilkens AB, Fulton EC, Pont MJ, Cole GO, Leung I, Stull SM, et al. NOTCH1 signaling during CD4+ T-cell activation alters transcription factor networks and enhances antigen responsiveness. Blood. 2022;140(21):2261–75.
104. Yi Y, Wang Z, Sun Y, Chen J, Zhang B, Wu M, et al. The EMT-related transcription factor snail up-regulates FAPα in malignant melanoma cells. Exp Cell Res. 2018;364(2):160–7.
105. Riechelmann H, Steinbichler TB, Sprung S, Santer M, Runge A, Ganswindt U, et al. The Epithelial-Mesenchymal Transcription Factor Slug Predicts Survival Benefit of Up-Front Surgery in Head and Neck Cancer. Cancers (Basel). 2021;13(4):772.
106. Jiang P, Yu J, Zheng Y, Tian C, Tu Y, Gong C, et al. Prognostic significance and immune infiltration analysis of HMGA2 in endometrial cancer. Front Immunol. 2025;16:1559278.
107. Ding N, Zhang T, Yu X, Zhuang S. T-Box Transcription Factor 2 Enhances Chemoresistance of Endometrial Cancer by Mediating NRF2 Expression. Curr Protein Pept Sci. 2022;23(8):563–70.
108. Yu X, Yao X, Song F, Zhu X. T-Box Transcription Factor 2 Mediates Chemoresistance of Endometrial Cancer via Regulating FSP1-involved Ferroptosis. Cell Biochem Biophys. 2025;83(1):1313–20.
109. Ma X, Wang Q, Sun C, Agarwal I, Wu H, Chen J, et al. Targeting TCF19 sensitizes MSI endometrial cancer to anti-PD-1 therapy by alleviating CD8(+) T cell exhaustion via TRIM14-IFN-β axis. Cell Rep. 2023;42(8):112944.
110. Abedin Y, Minchella P, Peterson R, Gonnella F, Graham A, Cook I, et al. Functional Analysis of RE1 Silencing Transcription Factor as a Putative Tumor Suppressor in Human Endometrial Cancer. Int J Mol Sci. 2024;25(17):9693.
111. Fu L, Girling JE, Rogers PA. Expression of Fox head protein 1 in human eutopic endometrium and endometriosis. Reprod Sci. 2008;15(3):243–52.
112. Zhang Y, Zhang L, Sun H, Lv Q, Qiu C, Che X, et al. Forkhead transcription factor 1 inhibits endometrial cancer cell proliferation via sterol regulatory element-binding protein 1. Oncol Lett. 2017;13(2):731–7.
113. Abe Y, Ijichi N, Ikeda K, Kayano H, Horie-Inoue K, Takeda S, et al. Forkhead box transcription factor, forkhead box A1, shows negative association with lymph node status in endometrial cancer, and represses cell proliferation and migration of endometrial cancer cells. Cancer Sci. 2012;103(4):806–12.
114. Wei X, Xiong X, Wang P, Zhang S, Peng D. SIRT1-mediated deacetylation of FOXO3 enhances mitophagy and drives hormone resistance in endometrial cancer. Mol Med. 2024;30(1):147.
115. Mishra A, Ganguli N, Majumdar SS, Modi D. Loss of HOXA10 causes endometrial hyperplasia progressing to endometrial cancer. J Mol Endocrinol. 2022;69(3):431–44.
116. Wan J, Liu H, Feng Q, Liu J, Ming L. HOXB9 promotes endometrial cancer progression by targeting E2F3. Cell Death Dis. 2018;9(5):509.
117. Cheng X, Shen C, Liao Z. KLF2 transcription suppresses endometrial cancer cell proliferation, invasion, and migration through the inhibition of NPM1. J Obstet Gynaecol. 2023;43(2):2238827.
118. Ding L, Ding Y, Kong X, Wu J, Fu J, Yan G, et al. Dysregulation of Krüppel-like factor 12 in the development of endometrial cancer. Gynecol Oncol. 2019;152(1):177–84.
119. Dong P, Kaneuchi M, Xiong Y, Cao L, Cai M, Liu X, et al. Identification of KLF17 as a novel epithelial to mesenchymal transition inducer via direct activation of TWIST1 in endometrioid endometrial cancer. Carcinogenesis. 2014;35(4):760–8.
120. Rodriguez AC, Vahrenkamp JM, Berrett KC, Clark KA, Guillen KP, Scherer SD, et al. ETV4 Is Necessary for Estrogen Signaling and Growth in Endometrial Cancer Cells. Cancer Res. 2020;80(6):1234–45.
121. Pedrola N, Devis L, Llauradó M, Campoy I, Martinez-Garcia E, Garcia M, et al. Nidogen 1 and Nuclear Protein 1: novel targets of ETV5 transcription factor involved in endometrial cancer invasion. Clin Exp Metastasis. 2015;32(5):467–78.
122. Wang F, Li J, Wang H, Zhang F, Gao J. Activating transcription factor 3 inhibits endometrial carcinoma aggressiveness via JunB suppression. Int J Oncol. 2020;57(3):707–20.
123. Liu B, Chen P, Xi D, Zhu H, Gao Y. ATF4 regulates CCL2 expression to promote endometrial cancer growth by controlling macrophage infiltration. Exp Cell Res. 2017;360(2):105–12.
124. Okumura T, Raja Xavier JP, Pasternak J, Yang Z, Hang C, Nosirov B, et al. Rel Family Transcription Factor NFAT5 Upregulates COX2 via HIF-1α Activity in Ishikawa and HEC1a Cells. Int J Mol Sci. 2024;25(7):3666.
125. Pavlovic ZJ, Hsin-Yu Pai A, Hsiao TT, Yen CF, Alhasan H, Ozmen A, et al. Dysregulated expression of GATA2 and GATA6 transcription factors in adenomyosis: implications for impaired endometrial receptivity. F S Sci. 2024;5(1):92–103.
126. Yu R, Wei C, Li G, Ouyang J, Liu N, Gu N, et al. Aberrant TCF21 upregulation in adenomyosis impairs endometrial decidualization by increasing PDE4C expression. Biochim Biophys Acta Mol Basis Dis. 2025;1871(1):167526.
127. Hiraoka T, Hirota Y, Aikawa S, Iida R, Ishizawa C, Kaku T, et al. Constant Activation of STAT3 Contributes to the Development of Adenomyosis in Females. Endocrinology. 2022;163(5):bqac044.
128. Li B, Chen M, Liu X, Guo SW. Constitutive and tumor necrosis factor-α-induced activation of nuclear factor-κB in adenomyosis and its inhibition by andrographolide. Fertil Steril. 2013;100(2):568–77.
129. Nie J, Lu Y, Liu X, Guo SW. Immunoreactivity of progesterone receptor isoform B, nuclear factor kappaB, and IkappaBalpha in adenomyosis. Fertil Steril. 2009;92(3):886–9.
130. Łupicka M, Socha B, Szczepańska A, Korzekwa A. Expression of pluripotency markers in the bovine uterus with adenomyosis. Reprod Biol Endocrinol. 2015;13:110.
131. Stratopoulou CA, El Grari I, Camboni A, Donnez J, Dolmans MM. Expression of Endometrial Receptivity Markers throughout the Menstrual Cycle in Women with and without Uterine Adenomyosis. J Clin Med. 2024;13(17):5016.
132. Guo S, Zhang D, Lu X, Zhang Q, Gu R, Sun B, et al. Hypoxia and its possible relationship with endometrial receptivity in adenomyosis: a preliminary study. Reprod Biol Endocrinol. 2021;19(1):7.
133. Ganieva U, Nakamura T, Osuka S, Bayasula, Nakanishi N, Kasahara Y, et al. Involvement of Transcription Factor 21 in the Pathogenesis of Fibrosis in Endometriosis. Am J Pathol. 2020;190(1):145–57.
134. Wu PL, Zhou Y, Zeng C, Li X, Dong ZT, Zhou YF, et al. Transcription factor 21 regulates expression of ERβ and SF-1 via upstream stimulatory factor-2 in endometriotic tissues. Biochim Biophys Acta Gene Regul Mech. 2018;1861(8):706–17.
135. Ntzeros K, Mavrogianni D, Blontzos N, Soyhan N, Kathopoulis N, Papamentzelopoulou MS, et al. Expression of ZEB1 in different forms of endometriosis: A pilot study. Eur J Obstet Gynecol Reprod Biol. 2023;286:121–5.
136. Furuya M, Masuda H, Hara K, Uchida H, Sato K, Sato S, et al. ZEB1 expression is a potential indicator of invasive endometriosis. Acta Obstet Gynecol Scand. 2017;96(9):1128–35.
137. Wilson MR, Reske JJ, Chandler RL. AP-1 Subunit JUNB Promotes Invasive Phenotypes in Endometriosis. Reprod Sci. 2022;29(11):3266–77.
138. González-Ramos R, Donnez J, Defrère S, Leclercq I, Squifflet J, Lousse JC, et al. Nuclear factor-kappa B is constitutively activated in peritoneal endometriosis. Mol Hum Reprod. 2007;13(7):503–9.
139. Amirteimouri S, Ashini M, Ramazanali F, Aflatoonian R, Afsharian P, Shahhoseini M. Epigenetic role of the nuclear factor NF-Y on ID gene family in endometrial tissues of women with endometriosis: a case control study. Reprod Biol Endocrinol. 2019;17(1):32.
140. Badary DM, Abou-Taleb HA, Ibrahim M. Hypoxia-inducible Factor-1α and mTOR as a Potential Therapeutic Target in Endometriosis: An Immunohistochemical Study. Appl Immunohistochem Mol Morphol. 2023;31(9):629–34.
141. Maurya VK, Szwarc MM, Fernandez-Valdivia R, Lonard DM, Yong S, Joshi N, et al. Early growth response 1 transcription factor is essential for the pathogenic properties of human endometriotic epithelial cells. Reproduction. 2022;164(2):41–54.
142. Dyson MT, Roqueiro D, Monsivais D, Ercan CM, Pavone ME, Brooks DC, et al. Genome-wide DNA methylation analysis predicts an epigenetic switch for GATA factor expression in endometriosis. PLoS Genet. 2014;10(3):e1004158.
143. Chen P, Ren F, Wang DB. GATA-3 Stimulates Proliferation of Endometriotic Cells. Ginekol Pol. 2019;90(6):308–13.
144. Feng Y, Yue J, Fan S, Wu J. The SOX18-OTUB1-YAP1 axis: a new endometriosis target. J Transl Med. 2025;23(1):647.
145. Daftary GS, Zheng Y, Tabbaa ZM, Schoolmeester JK, Gada RP, Grzenda AL, et al. A novel role of the Sp/KLF transcription factor KLF11 in arresting progression of endometriosis. PLoS One. 2013;8(3):e60165.
146. Chen Q, Zhou Y, Yu M, Zhu S, Sun J, Du W, et al. Transcription factor EB-mediated autophagy affects cell migration and inhibits apoptosis to promote endometriosis. Apoptosis. 2024;29(5-6):757–67.
147. Kellner S, Kikyo N. Transcriptional regulation of the Oct4 gene, a master gene for pluripotency. Histol Histopathol. 2010;25(3):405–12.
148. Chang JH, Au HK, Lee WC, Chi CC, Ling TY, Wang LM, et al. Expression of the pluripotent transcription factor OCT4 promotes cell migration in endometriosis. Fertil Steril. 2013;99(5):1332–9.e5.
149. Knez J, Kovačič B, Goropevšek A. The role of regulatory T-cells in the development of endometriosis. Hum Reprod. 2024 May 16:deae103.
150. Dong X, Xu L, Wang S, Jiao X, Yan S, Huang Y, et al. Endometrial stromal cell autophagy-dependent ferroptosis caused by iron overload in ovarian endometriosis is inhibited by the ATF4-xCT pathway. Mol Hum Reprod. 2023;30(1):gaad046.
151. Zeitoun K, Takayama K, Michael MD, Bulun SE. Stimulation of aromatase P450 promoter (II) activity in endometriosis and its inhibition in endometrium are regulated by competitive binding of steroidogenic factor-1 and chicken ovalbumin upstream promoter transcription factor to the same cis-acting element. Mol Endocrinol. 1999;13(2):239–53.
152. Lazim N, Elias MH, Sutaji Z, Abdul Karim AK, Abu MA, Ugusman A, et al. Expression of HOXA10 Gene in Women with Endometriosis: A Systematic Review. Int J Mol Sci. 2023;24(16):12869.
