Abstract
Tuberculosis (TB) remains a considerable global health threat and this is the second leading cause of death among all infectious diseases, caused by the bacterium Mycobacterium tuberculosis (Mtb). TB commonly influences the lungs, but it can also affect other parts of the body such as kidney, brain or spine. When drug can no longer kill the TB bacteria, disease is called as drug resistant TB. Besides drug resistant issues long term and multiple drugs regimes are allied with severe side effects such as organ toxicity. Scientific world believe that nanotechnology proposed new ways to address residual scientific concerns for TB. Nanotechnology based ideology have important prospective for diagnosis, treatment and prevention of tuberculosis (TB). Some nanotechnologybased targeting systems offer a promising solution as their nanostructure systems are highly efficient in encapsulating important amount of TB drugs, highly stable, feasible for distinct administration routes such as oral, intravenous and inhalation and could be designed to inhibit systemic clearance and pre-mature degradation. The development of nanoparticle-based aerosol vaccine is undergoing which could provide a great potential in prevention of tuberculosis infection. Considering the role of nanoparticles (NPs) in these three aspects like diagnosis, treatment and prevention of tuberculosis, this article attempts to compile these aspects in hope for better upgrade.
Keywords
Nanotechnology, Mycobacterium tuberculosis, Drugs, NPs, Granuloma, Isoniazid
Abbreviations
TB: Tuberculosis; WHO: World Health Organization; Mtb: Mycobacterium tuberculosis; MDR TB: Multi Drug Resistant Tuberculosis; XDR TB: Extensively Drug-Resistant Tuberculosis; HIV: Human Immunodeficiency Virus; AIDS: Acquired Immunodeficiency Syndrome; ATD’s: Anti-TB Drugs; DNA: De-oxy Ribonucleic Acid; RNA: Ribonucleic Acid; NPs: Nanoparticles; NMR: Nuclear Magnetic Resonance; PEG: Polyethylene Glycol; GI: Gastrointestinal; BUN: Blood Urea Nitrogen; R&D: Research & Development; TDR: Total Drug-Resistant
Introduction
Despite the curious advancement in medical science and therapeutics, tuberculosis (TB) persist the primary factor of mortality than any other infectious disease and socioeconomic disaster for millions of people around the world [1]. According to the World Health Organization (WHO), World’s One-third of the population is infected with this disease and of these, 8 to 10 million people develop active disease and 2 million people die each year and the rest of the infected people remain asymptomatic. After HIV/AIDS, it is serving as world’s second cause of death [2]. Presence of immunosuppressive condition like, alcoholism, malnutrition, chronic lung disease, diabetes & HIV/AIDS may increase the chances of TB infection. The situation is exacerbated by the emergence of multidrug-resistant TB and the catastrophic nexus between AIDS and TB [3]. Mtb which possesses inherent ability to remain dormant for several years or cause active diseases upon infections trigged the pathogenesis of TB. Macrophages in the lungs are the most important cell type supporting replication of Mtb in humans [4]. Mtb is heralded by its ease of transmission; prolong diagnosis, communicability, therapeutic adherence, and resistance [5]. The evolution of active TB differs with factors such as gender, age, geographical locations, immunity level and period of infection. For example, men are more prone to the infection of TB than women. Apart from gender, young children are also at risk to TB infection, as their lifetime risk of TB was approximately 10%. Principally, this bacterium attacks the lungs, but it can attack any part of the body, like the kidney, lymphatic system, circulatory system (military tuberculosis), central nervous system (meningitis), genitourinary system, bones, and joints. The mortality rate of untreated active tuberculosis is approximately 50%. The success and failure rate to treat TB depends on various factors like patient compliance to the treatment taken, malnutrition, smoking, coexisting diseases like HIV, and skimpy supervision by health care staffs [2]. The serious problem with the current tuberculosis chemotherapy is that when the drug is taken either intravenously or administered orally, it is assigned throughout the body via dissemination of blood in which majority of molecules, stay in the body causing adverse side effects, because they do not influence their targets. Drugs have rapid clearance and a short plasma-life, which limits their effectiveness [1]. The clinical management of TB still remains a problematic task. Patients who are infected with TB strains, resistant to some drugs such as isoniazid, pyrazinamide and rifampicin is called as Multi drug resistant (MDR) TB, which is practically incurable by first-line treatment. MDR-TB strains that are resistant to the drugs like fluoroquinolones such as ciprofloxacin, levofloxacin, and moxifloxacin, and thioamides such as ethionamide, prothionamide, and cycloserine, and second-line injectable drugs are called as extensively drug-resistant (XDR) TB (Table 1) [2,6]. MDR-TB and XDR-TB are increasing in developing countries that cause a challenge to the medical sciences and became matter of great concern. The alarm of spread of the MDR and XDR strains and the inadequacy of successful treatment options strengthen the need to develop new and effective anti-TB drugs to affect the problem of drug resistance, shorten the treatment course, and better compliance [7]. Although current anti-TB drugs are adequate, urgent strategies must be developed in order to accomplish delivery of these drugs.
| Drugs | Administration Routes | Mycobacterium species | Mechanism of Action | Side effects | References | |
|---|---|---|---|---|---|---|
| First Line Drugs | Isoniazid | Oral | Mtb | It inhibits the synthesis of mycolic acids, an essential component of the bacterial cell wall, bactericidal, involved in DNA, lipids, and carbohydrates metabolism. | Hepatitis (symptoms include loss of appetite, nausea, vomiting, fatigue, malaise, and weakness), nausea, vomiting, upset stomach, fever, or rash | [2] |
| Rifampicin | Oral | Mtb | It acts via the inhibition of DNA- dependent RNA polymerase, leading to a suppression of RNA synthesis and cell death, bactericidal effect | Bleeding, loss of appetite, low urine excretion, cramps, drowsiness, fatigue, stomach upset. | [2] | |
| Pyrazinamide | Oral | Mtb | Bactericidal effect, prevent the process of managing damaged proteins and rescue non-functioning ribosome’s in Mtb and dispersed into active Mtb that explicit pyrazinamidase enzyme that converts pyrazinamide to the active form pyrazinoic acid |
Nasal problem, upset stomach, vomiting, loss of appetite, mild muscle or joint pain, or fatigue. | [2] | |
| Ethambutanol | Oral | Mtb | It inhibits arabinosyl transferase which is involved in cell wall biosynthesis. By inhibiting this enzyme, the bacterial cell wall complex production is prohibited. | Etching or rash, joint pain, headache, dizziness or nausea, stomach pain, indigestion, loss of appetite. |
[2,50] | |
| Second Line Drugs | Streptomycin | Intravenous or Intramuscular | Mycobacteirium avium | Bacteriostatic or bactericidal effect. Acts as an irreversible inhibitor of protein (polypeptide) synthesis by binding to 30S ribosomal subunits, thus resulting in translational disruption. | Hypokalemia, hypomagnesemia, nephrotoxicity, and vestibular toxicity | [2] |
| Kanamycin | Intravenous or Intramuscular | Mtb | This bactericidal has strong anti- TB activity. Cross-resistance with amikacin and some data suggesting cross-resistance with capreomycin; inhibits protein synthesis. | Nephrotoxicity, ototoxicity (hearing loss) and vestibular toxicity | [2,51] | |
| Capreomycin | Intravenous or Intramuscular |
Mtb | Inhibit protein synthesis by binding to components in the bacterial cell which result in the synthesis necessary for bacterial survival. | Nephrotoxicity, hypersensitivity, neuromuscular blockade, auditory and vestibular ototoxicity. | [2,52] | |
| Other Drugs | Clofazimine | Oral | Mtb | Antimycobacterial (Bind to mycobacterial DNA leading to disruption of the cell cycle and eventually kills the bacterium) and immunosuppressive properties. | Dry and flaky skin, nausea, abdominal pain as well as discoloration of the skin, retina, cornea and urine. It may also cause irritated eyes, sensitivity to light, headache, fever, increased blood sugar and liver damage. | [2] |
| Clarithromycin | Oral | Mycobacterium ulcerans | Prevents protein synthesis via binding to 50S ribosomal RNA. It also inhibits the hepatic microsomal CYP3A4 isoenzyme and P-glycoprotein, an energy-dependent drug efflux pump | Gastrointestinal (GI) effects, general, abnormal taste, diarrhea, nausea, vomiting, elevated blood urea nitrogen (BUN), abdominal pain, rashes. | [2,53] | |
| Amoxicilin | Oral | Mycobacteriumavium complex | Inhibit the synthesis of third and last stage of bacterial cell wall | Pain in the lower abdomen, itchiness, nausea, vomiting, or no appetite. Yellowish skin or eyes, Dark or brown urine. | [2] |
Some drugs (rifampicin, isoniazid, pyrazinamide, and ethambutol), taken during the first two months of the treatment following a period of four months where only rifampicin and isoniazid are taken. Although the success rate of these regimes has been estimated to be over 85% [8], the full length of treatment is crucial for the effective and complete eradication of the pathogen.
In this context, nanotechnology is one of the most encouraging passages for diagnosis, treatment and prevention of TB and the development of more decisive and more effective drug delivery systems for treatment of TB.
Drug delivery system based on nanotechnology has ability to increase the tolerability of noxious chemotherapies sustained and controlled drug release, and ultimately enhanced bioavailability [9]. The desired particle size of drug required for the localization upon administration by inhalation is between 50 and 200 nm [1]. This technology has provided advancement in the field of pharmacology through the designing of drug delivery systems which is able to target mycobacterial (intracellular pathogen) infected phagocytic cells [10]. NPs can help to improve the diagnosis of mycobacterial strains as well as the delivery of anti-TB drugs (ATD’s) at their targeted tissues. In this paper, we are describing various nanotechnology-based approaches used for the diagnosis of tuberculosis bacilli and several ATDs have been suggested, to arrest the growth of tuberculin bacteria [11].
Nanotechnology in the Diagnosis of Tuberculosis
Tuberculosis case detection remains difficult, partly because of inaccurate diagnostic methods [12]. Diagnosis of TB infection (active or latent) is essential not only for treatment of the infected individual, but also for controlling its spread among various populations [13]. Over the past half-century, many improvements in the diagnosis of infectious diseases have been accomplished [14].
Polymerase chain reaction which is a nucleic acid diagnostic test plays an important role in the detection of TB bacilli at an early stage of TB disease progression [13]. TB epidemic is at its highest peak in our country and this epidemic is a result of many social and economic factors. One of the Nanotechnology-based TB diagnostic kit designed by the Central Scientific Instruments Organization of India is currently in the clinical trials phase. By functionalizing the surface of the particles with targeting ligand, higher bioavailability could be generated at the site of infection and therefore the dose and side effects of the drug are reduced [15,16].
Nanoparticles
Many NPs like metallic NPs (gold and silver NPs), and fluorescent NPs can be utilized for the diagnosis of many infectious diseases including tuberculosis. The gold NPs are the first nonmaterial’s used as nano-diagnostics for the diagnosis of TB in 1996. Many different molecules, such as antibodies, antigens and enzymes, could be conjugated with gold NPs as electrochemical labels, optical probes, and signal transfer amplifiers in the diagnosis of many infectious diseases [11]. Gold NPs attached with small segment of Mtb genome designs the basis of easy-toread test for the presence of TB-bacilli genetic sequences. The NPs have a matrix type structure in which the active ingredient is dispersed throughout, whereas the nanocapsules have an active ingredient and core polymeric membrane [7].
Quantum Dots
Nanotechnology used semiconductor-based fluorescent nanostructures size ranges around 1–10 nm, have many novel properties, such as photo stability, high quantum yield, and bright. These nanostructures can be made to fluoresce in different colors depending on their size to overcome the low specificity of fluorescence or electronic microscope to detect TB bacilli that can be significant more light emissions and more cycles of excitations in comparison to typical organic molecules [11].
Imaging Nanotechnology
The molecule of targeted TB bacilli labeled with quantum dots or synthetic chromophores such as fluorescent proteins that will expedite direct examination of intracellular signaling complex by optical techniques, for example: confocal fluorescence microscopy or correlation imaging [7].
Protein Chips
Proteins play significant role in establishing the biological phenotype and pathogenesis of organisms in healthy and diseased states. Proteomics play a significant role in the diagnosis of tuberculosis and its respective drug development through protein chips. Protein chips can be preserved with specific protein components and with different chemical groups that can particularly bind to the protein of TB bacilli containing a certain structural or sequential motif [14].
Nuclear Magnetic Resonance (NMR) with Micro Fluidic System
Magnetic properties of NPs depend on their molecular structure and composition. Super paramagnetic NPs made of different magnetic materials such as nickel, iron, cobalt etc. are made of magnetic materials (e.g. iron, nickel, cobalt, or alloys of magnetic metals) are promoted for biomedical applications, because they behave non-magnetically when they are not in influence of an external magnetic field, thus avoid undesired selfmagnetic agglomeration [17]. Magnetic NPs have highly sensitive detection of bacillus, for example: Iron based NPs attached with specific antibodies are used for binding the Mtb bacilli although micro fluidic system transports the Mtb and buffer solutions. Assimilation with quantum dots can diagnose not only Mtb but also the subspecies of Mycobacterium avium [18,19].
Sparse Cell Detection
Sparse cells are significant in the diagnosis of various genetic and infectious diseases. These cells are different from their surrounding cells in normal physiological conditions. It is a challenging task to identify and subsequently isolate these sparse cells. With the help of nanotechnology, scientists developed nano-systems capable of effectively sorting sparse cells from blood and other tissues [20].
Nanotechnology in the Treatment of Tuberculosis
Despite the availability of effective antibiotics, serious toxic side effects to the sites other than the infected site limit the doses that can be used clinically [21]. In the field of therapeutics, nanotechnology is very fast emerging technology as drug delivery systems [22]. Over the past years, rising use of therapy, which is based on nanotechnology, has been researched for replacing the administration of antibiotics or other drugs in the free form with an access using drugs that are encapsulated with NPs. Nanotechnology based NPs are very much versatile and distinct in their properties and structural configuration which facilitate them to be used for clinical diagnosis and delivery of drugs in a unique and more decisive manner [11]. NPs can cross the intestinal permeability barrier through the transcellular or paracellular pathways into the circulation [23]. Once NPs enter inside the living system, various proteins adsorb to the surface giving rise to ‘corona’ and even if they are not small enough to enter inside living cell, they still could form bonding with the cell membrane due to surface defects [24]. The nanostructures engaged as drug delivery systems have multiple advantages which make them superior to conventional delivery systems. Some nanosized carriers such as liposome’s, polymeric NPs, nano suspensions, nano emulsions and many other excellent multifunctional nanosystems, are available for drug delivery purpose leading to better pharmacokinetics, bioavailability and biodistribution [25]. These nanocarriers exhibit the important technological advantages such as higher stabilization (long shelf life) and drug-carrier capacity, feasibility of incorporation of both hydrophobic and hydrophilic substances and feasibility of administration routes not only intravenous but also oral and inhalation routes [26]. These carriers can also be designed to facilitate controlled drug release from the matrix [10] and are able to target cellular reservoirs of Mtb [27]. Early success of NPs -based drug delivery has led to the investigation and development of many different compositions of polymeric NPs, including polymeric micelles, dendrimers and drug conjugates etc. (Figure 1) [28].
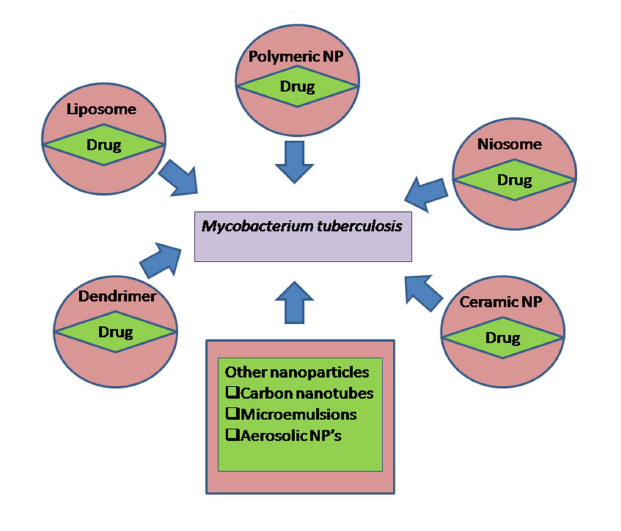
Figure 1. Different nano - technological based approaches to target Mtb.
Nanocarriers
Liposomes
Liposomes are tiny spherical vesicles composed of amphiphilic lipid bilayer membranes with an aqueous core and can also help to achieve targeted therapy [26]. Liposomes are commonly prepared by hydration of a dry phospholipids ?lm above the main phase transition temperature of the lipid [29]. These serve as carrier systems for some hydrophilic drugs such as gentamicin, streptomycin and many other drugs, depending on their biological compatibility. The drug encapsulated liposomes significantly decrease the bacterial count in liver and spleen in comparison to free drug [20]. Liposomes are made up of the mixture of phosphatidylcholine, cholesterol, dicetylphosphate, o-stearoyl amylopectin, distearoyl phosphatidylethanolamine and polyethylene glycol (PEG). Phosphatidylcholine is the most commonly used lipid in liposomal preparation besides using cholesterol (for maintaining rigidity and stability) [11]. The circulatory lifespan of PEG encapsulated liposome enhances in the blood stream [30]. Drug-loaded nanocarriers showed a significantly reduced toxic effects when assessed for cytotoxicity in peritoneal macrophages compared to free drugs [31].
Polymeric NPs
Polymeric NPs are used as carriers for drug delivery that possess very good biocompatibility and biodegradable features in which the drug is attached, entrapped or encapsulated in polymeric core and depending upon the method of preparation, they are classified as NPs, nanospheres or nanocapsules [32]. The smaller size of these particles facilitates them to permeate capillaries and to be taken up by cells, thereby increasing the accumulation of drugs at their target sites [33]. Polymeric NPs serve as an attractive alternative to liposome [34]. These NPs are physically much more stable and can be synthesized with various properties such as drug release profile and zeta potential etc. by selecting different polymer lengths, monomer dimensions, surfactants and choice of organic solvents. Polymeric NPs possess typical functional groups that can be transformed according to either structural moiety of drugs or targeted ligand [35]. NPs are exciting systems for brain drug delivery because they can be modulated in terms of shape, size, hydrophobicity, coating, chemistry and surface charge [36]. NPs which release drugs in response to an internal or external stimulus such as pH, temperature, redox, magnetic and light have been actively pursued [37].
These NPs can increase the solubility of constituents, reduce the therapeutic dose and improve the absorption of the active components. Furthermore, they are beneficial when used in blood because they are non-toxic, non thrombogenic, non-immunogenic, non-inflammatory, and avoid the reticulo-endothelial system. NPs increase immunization by avoidance of degradation of the vaccine and increased uptake by immune cells. These all properties of NPs make them very useful to target TB bacilli [38].
Dendrimers
Dendrimers are very large chained, repeated three dimensional arrangements of group of atoms. The diameter of this synthetic nanomaterial is 5-10 nm [39,40]. These are very accomplished molecules with respect to their multifunctional and geometrical nature. A range of different molecules can be coupled to these three regions of dendrimers, which is the reason for their high functionality. Each of these regions possesses different functionalities and therefore the properties such as solubility, thermal stability can be easily manipulated for a range of applications [41].
Dendrimers are very useful in the drug delivery of tuberculosis because these have hydrophilic materials on the outer core and a hydrophobic inside core. The cell wall composition of Mycobacterium (a rich layer of mycolic acid) renders the potential anti-TB medicinal preparations difficult to enter the infected cells [41,42]. With the help of dendrimers, the arrangement of the carried medicinal formulation is biochemically altered in a way which favors its entry into the target cells [43]. Moreover, dendrimers prevent the disease in its latent period itself. After entering into the cell, the polymeric drug complex is readily cleaved by the cell’s lysosomal compartments, which results in the release of drug into the cell at a high concentration [44].
Niosomes
Niosomes are nanosized and nonionic surfactant vesicles and examined as novel drug delivery systems, can improve the solubility and stability of natural pharmaceutical molecules [45]. Their particle size ranges from 10 nm to 100 nm. These are the structural analogue of liposome, but they have some additional advantages over liposome. These molecules are amphipathic molecules that can be stabilized using surfactants [32]. Niosomes are immensely useful in ensuring some critical parameters for optimum drug biodistribution in the living systems [41].
Till date TB treatment using niosomes has been performed using the conventional ATDs isoniazid, rifampicin, and pyrazinamide. Niosomes are particularly used for the sitespecific delivery of the drug and significantly reduced the systemic toxicity of the drug delivered, TB, by reducing the side-effects to almost nil, also making the treatment more accessible [45].
Carbon nanotubes
They are the tubes having the length of several micrometers and cross-sectional diameter in the ranges of 1–100 nm. These can be functionalized with several different chemical moieties attached to their surface and possess wide applications. They are mainly of two types: single-walled and multi-walled. These nanotubes can behave in both metallic and nonmetallic manner, thereby increasing their biochemical utility manifold. Many groups such as bioactive peptides, nucleic acids and proteins can be coupled to their surface to make their carriers more easily available to cells and organs [46].
Micro emulsions
They serve as a stable thermodynamic mixture of two immiscible liquids such as oil and water, which are combined with the help of surfactant molecules to behave as one phase. The dispersed phase typically constitutes small droplets, with a range of 5 nm-200 nm size, has very low oil/water interfacial tension and hence these systems are also termed as nano-emulsions. They have droplet size, viscosity, density, refractive index, phase separation and pH measurements shall be executed to characterize these micro emulsions. They aid in the improvement of drug bioavailability, resistance against enzymatic hydrolysis, and reduced toxicity. It has been fruitfully used for killing the germs of TB at low dosage [46].
Aerosolic NPs
These NPs are a suspension of NPs in the form of aerosol dry powder. They are used to develop the NPs as potential drug delivery molecules that can be inhaled and treat the subsequent pulmonary infections. Tuberculosis is being one of the respiratory infections having multidrug resistance characteristic that can be therefore immensely powerful when dealt with the use of NPs. With the help of these NPs efficient diagnosis and treatment of the TB is almost sure to be achieved with much less systemic toxicity [18].
Ceramic NPs
These NPs are the particles fabricated from some inorganic compounds with porous characteristics such as alumina, silica, and titania [47,48]. These particles can be prepared according to the desired size, shape and porosity. The size of these NPs is less than 100 nm and can avoid uptake by the reticule-endothelial system as foreign bodies. At physiological pH and temperature entrapped molecules such as drugs, proteins and enzymes are protected from denaturation. These particles are not biodegradable and so there is concern that they may acquire in the body and cause harmful effects, so these are not much useful in the treatment of tuberculosis and other infectious diseases. They have a wide range of applications because of their favorable properties, such as chemical inertness and high heat resistance (Figure 2) [49].
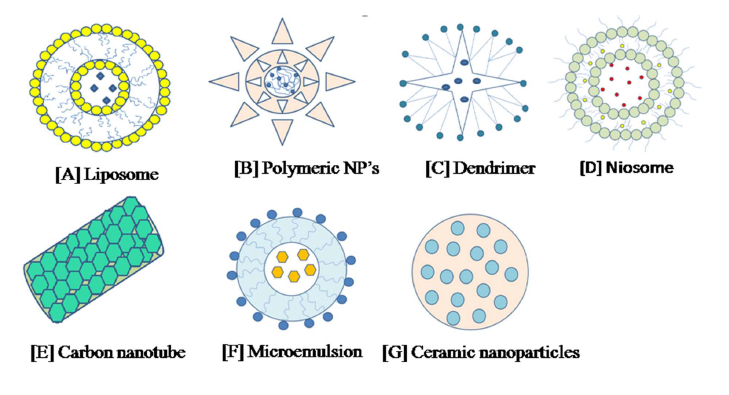
Figure 2. Advancement in pulmonary drug delivery systems for treatment of tuberculosis by using different NPs: [A] Liposome [B] Polymeric NPs [C] Dendrimer [D] Niosome [E] Carbon nano-tube [F] Micro emulsion [G] Ceramic NPs.
Nanotechnology in the Prevention of Tuberculosis
The aerosol vaccine made by nanotechnology could administer a needle-free and low-cost, TB treatment that is highly stable at room temperature. Although, most new TB vaccines continue to call for needle injection, but this new vaccine could provide safer, more consistent protection by excluding these injections and the need for refrigerated storage. Several drugs including MDR, XDR and TDR drugs (ethambutanol, kanamycin, capreomycin etc.) merged with nanotechnological strategies to cure and prevent a person from this deadly disease (Table 1) [50-53].
A successful result of these nanotechnology based aerosol delivery offers a potential new platform for prevention. In the aerosol vaccine, particles form at micrometer and nanometer scales and in spherical and elongated shapes, a combination that appears to enhance dispersal in the mouth. While commonly used with cosmetics, food and pharmaceuticals, this spray drying of small and large molecules is seldom used for drying cellular material.
Conclusion and Future Outlook
In this review, we have provided an overview regarding tuberculosis diagnosis, treatment and prevention with the help of some nanotechnology-based approaches. Because tuberculosis turned to be a major issue for population, and therefore we require a suitable way to cure people from this infectious disease as soon as possible. Nanotechnology is globally developing technology for targeted drug delivery and disease diagnosis. The obvious advantages of nanotechnology based diagnostic schemes are their ability to provide results within hours, with increased sensitivities and specificities at a fraction of a cost in comparison to conventional microbiological and molecular biology methodologies. The increasing awareness and R&D in the area of nanotechnology-based drug delivery would continue to change the whole concept of medicines including aspects such as characteristics of product, pharmacokinetics, bioavailability, stability, drug use, and toxicity in many infectious diseases including TB. In an area of many infectious diseases such as tuberculosis, nanotechnology has the potential to entitle a local response to many challenges for example the diagnosis, treatment and prevention of this deadly disease and we can see a better approach to sort out the problems. Nano diagnostics future trends will provide capability of non-specialized health personnel to use them through miniaturization of biochip technology to the nano-scale range for point-ofcare diagnostics with a specimen-in answer-out approach.
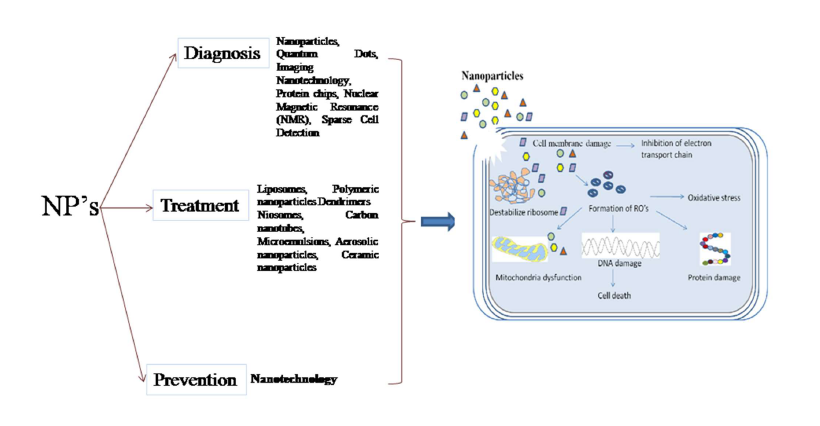
Graphical Abstract. Mechanism of action of NPs against Mtb: Different types of NPs (NPs) can be used in diagnosis, treatment and prevention of TB by employing different strategically mechanism of action against the bacilli.
Acknowledgements
The authors acknowledge financial support from the Department of Science and Technology-SERB, Council of Scientific and Industrial Research-Institute of Genomics and Integrative Biology under the research project GAP0145 (SERB-DST Grant no: EEQ/2016/000514).
Conflict of Interest
There is no conflict of interest.
References
2. Lee WH, Loo CY, Young PM, Traini D. The Potential of Nanotechnology for tuberculosis treatment. Tuberculosis. 2016 May.
3. Meena LS, Rajni. Survival mechanisms of pathogenic Mycobacterium tuberculosis H37Rv. The FEBS Journal. 2010 Jun 1;277(11):2416-27.
4. Kisich KO, Gelperina S, Higgins MP, Wilson S, Shipulo E, Oganesyan E, et al. Encapsulation of moxifloxacin within poly (butyl cyanoacrylate) nanoparticles enhances efficacy against intracellular Mycobacterium tuberculosis. International Journal of Pharmaceutics. 2007 Dec 10;345(1-2):154-62.
5. Edagwa BJ, Guo D, Puligujja P, Chen H, McMillan J, Liu X, et al. Long-acting antituberculous therapeutic nanoparticles target macrophage endosomes. The FASEB Journal. 2014 Dec;28(12):5071-82.
6. Seung KJ, Keshavjee S, Rich ML. Multidrug-resistant tuberculosis and extensively drug-resistant tuberculosis. Cold Spring Harbor Perspectives in Medicine. 2015 Sep 1;5(9):a017863.
7. Griffiths G, Nyström B, Sable SB, Khuller GK. Nanobead-based interventions for the treatment and prevention of tuberculosis. Nature Reviews Microbiology. 2010 Nov;8(11):827-34.
8. Miranda MS, Rodrigues MT, Domingues RM, Torrado E, Reis RL, Pedrosa J, et al. Exploring inhalable polymeric dry powders for anti-tuberculosis drug delivery. Materials Science and Engineering: C. 2018 Dec 1;93:1090-103.
9. Kumari A, Yadav SK, Yadav SC. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids and surfaces B: Biointerfaces. 2010 Jan 1;75(1):1- 8.
10. Mathuria JP. Nanoparticles in tuberculosis diagnosis, treatment and prevention: a hope for future. Digest Journal of Nanomaterials & Biostructures (DJNB). 2009 Jun 1;4(2).
11. Mishra V, Bansal KK, Verma A, Yadav N, Thakur S, Sudhakar K, et al. Solid lipid nanoparticles: Emerging colloidal nano drug delivery systems. Pharmaceutics. 2018 Dec;10(4):191.
12. Wallis RS, Pai M, Menzies D, Doherty TM, Walzl G, Perkins MD, et al. Biomarkers and diagnostics for tuberculosis: progress, needs, and translation into practice. The Lancet. 2010 May 29;375(9729):1920-37.
13. Wang S, Inci F, De Libero G, Singhal A, Demirci U. Point-of-care assays for tuberculosis: role of nanotechnology/microfluidics. Biotechnology Advances. 2013 Jul 1;31(4):438-49.
14. Wang Y, Yu L, Kong X, Sun L. Application of nanodiagnostics in point-of-care tests for infectious diseases. International Journal of Nanomedicine. 2017;12: 4789.
15. Xu K, Liang ZC, Ding X, Hu H, Liu S, Nurmik M, et al. Nanomaterials in the prevention, diagnosis, and treatment of Mycobacterium tuberculosis infections. Advanced Healthcare Materials. 2018 Jan;7(1):1700509.
16. Kingsley JD, Dou H, Morehead J, Rabinow B, Gendelman HE, Destache CJ. Nanotechnology: a focus on nanoparticles as a drug delivery system. Journal of Neuroimmune Pharmacology. 2006 Sep 1;1(3):340-50.
17. Saraf S. Applications of novel drug delivery system for herbal formulations. Fitoterapia. 2010 Oct 1;81(7):680-9.
18. Fakruddin M, Hossain Z, Afroz H. Prospects and applications of nanobiotechnology: a medical perspective. Journal of Nanobiotechnology. 2012 Dec;10(1):1-8.
19. Veigas B, Doria G, Baptista PV. Nanodiagnostics for tuberculosis. Understanding Tuberculosis-Global Experiences and Innovative Approaches to the Diagnosis. 2012 Feb 15:20.
20. Chun AL. Nanoparticles offer hope for TB detection. Nature Nanotechnology. 2009 Nov;4(11):698-9.
21. Shivangi, Meena LS. A novel approach in treatment of tuberculosis by targeting drugs to infected macrophages using biodegradable nanoparticles. Applied Biochemistry and Biotechnology. 2018 Jul 1;185(3):815-21.
22. Tanwar M, Meena J, Meena LS. Nanoparticles: scope in drug delivery. Advanced Biomaterials and Biodevices. 2014 Jul 25:487.
23. Meena J, Singh M, Sahare PD, Meena LS. Interaction of nanoparticles in biological systems and their role in therapeutical treatment of tuberculosis and cancer. Journal Luminescene Applications. 2014;1:7-22.
24. Cheepsattayakorn A, Cheepsattayakorn R. Roles of nanotechnology in diagnosis and treatment of tuberculosis. Journal of Nanotechnology in Diagnosis and Treatment. 2013 May;1(1):19-25.
25. Yih TC, Al-Fandi M. Engineered nanoparticles as precise drug delivery systems. Journal of Cellular Biochemistry. 2006 Apr 15;97(6):1184-90.
26. Shegokar R, Al Shaal L, Mitri K. Present status of nanoparticle research for treatment of tuberculosis. Journal of Pharmacy & Pharmaceutical Sciences. 2011 Apr 8;14(1):100-16.
27. Alexis F, Pridgen E, Molnar LK, Farokhzad OC. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Molecular Pharmaceutics. 2008 Aug 4;5(4):505-15.
28. Pinheiro M, Lúcio M, Lima JL, Reis S. Liposomes as drug delivery systems for the treatment of TB. Nanomedicine. 2011 Oct;6(8):1413-28.
29. Huh AJ, Kwon YJ. “Nanoantibiotics”: a new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. Journal of Controlled Release. 2011 Dec 10;156(2):128-45.
30. Soppimath KS, Aminabhavi TM, Kulkarni AR, Rudzinski WE. Biodegradable polymeric nanoparticles as drug delivery devices. Journal of Controlled Rselease. 2001 Jan 29;70(1-2):1-20.
31. Ochekpe NA, Olorunfemi PO, Ngwuluka NC. Nanotechnology and drug delivery part 2: nanostructures for drug delivery. Tropical Journal of Pharmaceutical Research. 2009;8(3).
32. Bala I, Hariharan S, Kumar MR. PLGA nanoparticles in drug delivery: the state of the art. Critical Reviews™ in Therapeutic Drug Carrier Systems. 2004;21(5).
33. Sharma A, Sharma S, Khuller GK. Lectinfunctionalized poly (lactide-co-glycolide) nanoparticles as oral/aerosolized antitubercular drug carriers for treatment of tuberculosis. Journal of Antimicrobial Chemotherapy. 2004 Oct 1;54(4):761-6.
34. Saraiva C, Praça C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-mediated brain drug delivery: overcoming blood–brain barrier to treat neurodegenerative diseases. Journal of Controlled Release. 2016 Aug 10;235:34-47.
35. Cheng R, Meng F, Deng C, Klok HA, Zhong Z. Dual and multi-stimuli responsive polymeric nanoparticles for programmed site-specific drug delivery. Biomaterials. 2013 May 1;34(14):3647-57.
36. Singh J, Pandit S, Bramwell VW, Alpar HO. Diphtheria toxoid loaded poly-(e-caprolactone) nanoparticles as mucosal vaccine delivery systems. Methods. 2006 Feb 1;38(2):96-105.
37. da Silva PB, de Freitas ES, Bernegossi J, Gonçalez ML, Sato MR, Leite CQ, et al. Nanotechnology-based drug delivery systems for treatment of tuberculosis-A review. Journal of Biomedical Nanotechnology. 2016 Feb 1;12(2):241-60.
38. Banyal S, Malik P, Tuli HS, Mukherjee TK. Advances in nanotechnology for diagnosis and treatment of tuberculosis. Current Opinion in Pulmonary Medicine. 2013;19(3):289-297.
39. Klajnert B, Bryszewska M. Dendrimers: properties and applications. Acta Biochimica Polonica. 2001 Mar 31;48(1):199-208.
40. Cheng Y, Xu Z, Ma M, Xu T. Dendrimers as drug carriers: applications in different routes of drug administration. Journal of Pharmaceutical Sciences. 2008 Jan 1;97(1):123-43.
41. Pandey R, Zahoor A, Sharma S, Khuller GK. Nanoparticle encapsulated antitubercular drugs as a potential oral drug delivery system against murine tuberculosis. Tuberculosis. 2003 Jan 1;83(6):373-8.
42. Gharbavi M, Amani J, Kheiri-Manjili H, Danafar H, Sharafi A. Niosome: a promising nanocarrier for natural drug delivery through blood-brain barrier. Advances in Pharmacological and Pharmaceutical Sciences. 2018 Jan 1;2018.
43. Uchegbu IF, Florence AT. Non-ionic surfactant vesicle (niosomes): physical and pharmaceutical chemistry. Advances in Colloid and Interface Science. 1995;58(1):1- 55.
44. Mujoriya R, Bodla RB, Dhamande K, Singh D, Patle L. Niosomal drug delivery system: The magic bullet. Journal of Applied Pharmaceutical Science. 2011;1(09):20-3.
45. Hu C, Rhodes DG. Proniosomes: a novel drug carrier preparation. International Journal of Pharmaceutics. 1999 Aug 5;185(1):23-35.
46. Ajayan PM, Zhou OZ. Applications of carbon nanotubes. InCarbon nanotubes 2001 (pp. 391-425). Springer, Berlin, Heidelberg.
47. Bianco A, Kostarelos K, Prato M. Applications of carbon nanotubes in drug delivery. Current Opinion in Chemical Biology. 2005 Dec 1;9(6):674-9.
48. Devarajan V, Ravichandran V. Nanoemulsions: as modified drug delivery tool. International Journal of Comprehensive Pharmacy. 2011;2(4):1-6.
49. C Thomas S, Kumar Mishra P, Talegaonkar S. Ceramic nanoparticles: fabrication methods and applications in drug delivery. Current Pharmaceutical Design. 2015 Dec 1;21(42):6165-88.
50. Peloquin CA. Therapeutic drug monitoring in the treatment of tuberculosis. Drugs. 2002 Oct 1;62(15):2169- 83.
51. Bastian I, Rigouts L, Palomino JC, Portaels F. Kanamycin susceptibility testing of Mycobacterium tuberculosis using Mycobacterium Growth Indicator Tube and a colorimetric method. Antimicrobial Agents and Chemotherapy. 2001 Jun 1;45(6):1934-6.
52. Heifets L, Simon J, Pham V. Capreomycin is active against non-replicating M. tuberculosis. Annals of Clinical Microbiology and Antimicrobials. 2005 Dec 1;4(1):6.
53. Alffenaar JW, Nienhuis WA, De Velde F, Zuur AT, Wessels AM, Almeida D, et al. Pharmacokinetics of rifampin and clarithromycin in patients treated for Mycobacterium ulcerans infection. Antimicrobial Agents and Chemotherapy. 2010 Sep 1;54(9):3878-83..
