Abstract
Calcific aortic valve disease (CAVD) is a prevalent and progressive disorder marked by fibrocalcific remodeling of the aortic valve, for which no effective pharmacological therapies currently exist. While CAVD is recognized as an active, cell-driven process, the intercellular communication networks that govern pathological valve remodeling are not fully understood. Extracellular vesicles (EVs) have emerged as key mediators of such communication in cardiovascular disease, transferring bioactive molecules like microRNAs (miRNAs) between cells to influence disease progression. This review synthesizes the current understanding of EV-mediated miRNA signaling in cardiovascular calcification, with a specific focus on its emerging role in CAVD. We discuss the established functions of EV-miRNAs in vascular disease and calcification and then examine recent paradigm-shifting evidence regarding miR-145-5p in the valvular context. Contrary to its well-established protective role in vascular smooth muscle cell homeostasis, recent work demonstrates that endothelial-derived EVs enriched in miR-145-5p are taken up by valvular interstitial cells (VICs), where they promote osteogenic reprogramming under calcifying conditions. This finding challenges the generalized view of miR-145-5p as a universally anti-calcific agent and highlights a critical dependence on cellular context and mode of intercellular transfer. By positioning EV-associated miR-145-5p as an active signaling molecule in valve calcification, this review underscores the complexity of miRNA function in different cardiovascular compartments and emphasizes the need to consider the specific cellular origin and delivery mechanisms when investigating the pathophysiology of CAVD.
Keywords
Calcific aortic valve disease, Cardiovascular therapeutics, Extracellular vesicles, microRNAs, MiR-145-5p, RNA therapeutics, Valve calcification
Introduction
Calcific aortic valve disease (CAVD) is a progressive disorder characterized by fibro-calcific remodeling of the aortic valve and remains a major unmet clinical challenge due to the lack of effective pharmacological therapies [1]. Although increasing evidence supports an active, cell-regulated disease process involving inflammation and osteogenic differentiation of valvular interstitial cells (VICs), the mechanisms coordinating cellular communication within the diseased valve microenvironment remain incompletely understood [2].
In recent years, extracellular vesicles (EVs) are increasingly recognized as important mediators of intercellular signaling in cardiovascular disease. By transferring bioactive cargo such as microRNAs (miRNAs), EVs enable communication between endothelial cells and mesenchymal cell populations, thereby influencing vascular remodeling and calcification [3]. For example, EV-associated miR-126 and miR-143 have been shown to regulate endothelial–smooth muscle cell crosstalk and atherosclerotic lesion stability, supporting a functional driver role in vascular disease progression [3,4]. In the context of calcification, circulating EV-miR-223 has been proposed as a biomarker of vascular calcification and coronary artery disease severity [5]. Although EV-associated miRNAs have been increasingly studied in vascular calcification, much less is known about their role in valvular disease. In particular, how EV-mediated miRNA transfer influences VIC behavior and contributes to valve calcification has only begun to be addressed.
At a mechanistic level, EVs deliver molecular cargo to recipient cells and influence gene expression and cellular behavior. EV cargo is heterogeneous and includes proteins, lipids, and multiple classes of non-coding RNAs. Among these, microRNAs have been studied most extensively because they can modulate defined signaling pathways in recipient cells. By transferring miRNAs between cells, EVs enable regulatory effects that extend beyond the cell of origin and contribute to processes such as vascular remodeling, inflammatory signaling, and calcification in cardiovascular tissues.
MiR-145-5p has been widely studied in the vascular field and is best known for its role in maintaining the differentiated phenotype of vascular smooth muscle cells and supporting vascular integrity. Accordingly, miR-145-5p is generally viewed as a protective factor in vascular disease [4]. Whether this concept also applies to valvular biology has remained uncertain. In particular, little is known about how EV-mediated transfer of miR-145-5p influences VIC behavior or contributes to valve calcification. In this regard, the recent work by Goody et al. [6] provides new mechanistic insights. By combining analyses of human samples with mechanistic in vitro experiments, the authors show that endothelial-derived EVs enriched in miR-145-5p are efficiently taken up by VICs and drive osteogenic reprogramming under calcifying conditions. These observations challenge the assumption of miR-145-5p as a universally anti-calcific miRNA and instead support a model in which its biological effects are strongly dependent on cellular context and mode of intercellular transfer. By positioning EV-associated miR-145-5p as an active signaling molecule rather than a passive biomarker, this work adds a new layer of complexity to current models of valvular calcification. It also raises important questions regarding the determinants of miRNA function in different cardiovascular compartments and underscores the need to consider cellular origin and mode of delivery when interpreting miRNA-based mechanisms in CAVD. This emerging evidence suggests that the biological effects of miR-145-5p cannot be generalized across cardiovascular tissues but are critically shaped by cellular context and mode of delivery.
EV-Mediated miRNA Communication in Cardiovascular and Valvular Calcification
EVs are increasingly appreciated as active conveyors of biological information in cardiovascular tissues, rather than inert by-products of cellular stress or injury. Through the targeted delivery of proteins, lipids, and non-coding RNAs, EVs enable cell-cell communication that can reshape gene regulatory programs in recipient cells [7,8]. Among EV cargoes, miRNAs have attracted particular attention due to their stability, regulatory potency, and ability to reprogram target cells in a context-dependent manner [9].
In cardiovascular pathology, EV-mediated miRNA transfer has been implicated in endothelial dysfunction, vascular inflammation, and smooth muscle cell phenotypic switching [4,10]. Endothelial cell-derived EVs can deliver specific miRNAs to vascular smooth muscle cells, reinforcing contractile gene programs under physiological conditions, whereas EVs released from stressed or diseased smooth muscle cells propagate pro-inflammatory and pro-calcific signals within the vascular wall [11]. These studies establish EV-miRNA signaling as a dynamic and bidirectional regulatory system in cardiovascular disease. Pathological calcification represents a particularly relevant setting for EV-mediated communication. In vascular smooth muscle cells, EVs contribute to calcification through two complementary mechanisms: acting as nucleation sites for mineral deposition and transferring regulatory signals that promote osteogenic differentiation [10–12]. EV-associated miRNAs modulate key osteogenic pathways, including Runx2 signaling, alkaline phosphatase activity, and extracellular matrix remodeling, thereby actively shaping the calcific phenotype rather than merely reflecting it. In contrast, the role of EV-mediated miRNA signaling in valvular calcification has remained less well defined. Although EV accumulation within calcified aortic valves has been documented, most prior studies have emphasized their structural contribution to microcalcification rather than their function as signaling entities. This imbalance has limited the integration of EV biology into contemporary models of CAVD, despite increasing recognition of coordinated intercellular communication as a driver of disease progression [13].
Emerging evidence suggests that EV-mediated miRNA transfer may be particularly relevant in the valvular microenvironment, where signals generated by valvular endothelial cells or infiltrating immune cells must be transmitted to VICs embedded within a dense extracellular matrix [4,14]. EVs provide an efficient mechanism for such signal propagation and may amplify osteogenic programming within VICs under pathological conditions. Recent work, most notably the study by Goody et al., directly supports this concept by demonstrating that EV-associated miRNAs are not only present in calcified valves but functionally active in recipient VIC. By linking EV-mediated miRNA transfer to defined molecular targets and osteogenic outcomes, these findings reposition EVs from passive participants to active regulators of cardiovascular and valvular calcification. Within this conceptual framework, the study by Goody et al. exemplifies how EV-mediated miRNA transfer can exert disease-specific effects in the aortic valve.
Context-Dependent Roles of miR-145-5p in Cardiovascular Biology and Valvular Calcification
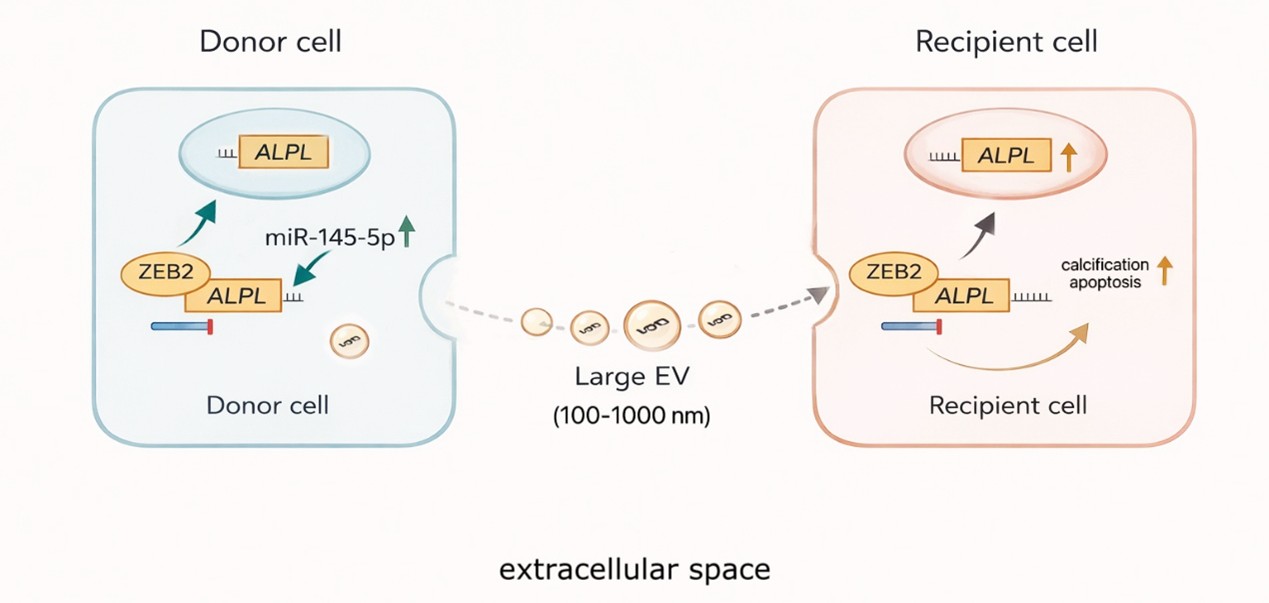
MiR-145-5p has long been regarded as a cornerstone microRNA in cardiovascular biology, primarily based on its well-established role in maintaining vascular smooth muscle cell (VSMC) differentiation and vascular stability. Extensive genetic and experimental evidence demonstrates that miR-145-5p suppresses phenotypic switching in VSMCs by targeting transcription factors such as KLF4, KLF5, and ELK1, and that loss of miR-145-5p is associated with vascular injury, atherosclerosis, and aneurysm formation. These observations have collectively positioned miR-145-5p as a key regulator of vascular smooth muscle cell phenotype and vascular stability [4,15]. Against this prevailing paradigm, the study by Goody et al. introduces a strikingly different role for miR-145-5p in the context of CAVD. The authors provide direct mechanistic evidence that miR-145-5p is enriched in EVs derived from calcified human aortic valves and from the circulation of patients with CAVD. Importantly, EV-mediated transfer of miR-145-5p to VICs promotes osteogenic reprogramming. This effect is mediated, at least in part, through repression of the transcription factor ZEB2, resulting in derepression of alkaline phosphatase expression and enhanced mineralization. These findings establish miR-145-5p not as a passive bystander, but as an active pro-calcific signal within the valvular microenvironment.
The apparent contradiction between the vasoprotective effects of miR-145-5p in VSMCs and its pro-calcific activity in VICs highlights an important principle in microRNA biology: miRNA function is dictated not by the miRNA itself, but by the cellular context in which it operates. In VICs, which possess a distinct developmental origin, mechanical environment, and baseline transcriptional landscape compared with VSMCs, miR-145-5p engages a different repertoire of targets and regulatory networks. As demonstrated by Goody et al., this context enables miR-145-5p to amplify osteogenic signaling rather than restrain pathological remodeling. Beyond its intracellular effects, the mode of miR-145-5p delivery further shapes its biological impact. Previous studies have shown that endothelial cell-derived EVs can transfer miR-145-5p to adjacent VSMCs, reinforcing contractile gene expression and limiting dedifferentiation under physiological or atheroprotective conditions. In contrast, the current study highlights EV-mediated miR-145-5p signaling as a driver of VIC osteogenic differentiation in a diseased valvular environment. These observations emphasize that both cellular origin and recipient cell identity are critical determinants of miR-145-5p function, as illustrated in the proposed model (Figure 1).
The aortic valve thus represents a unique biological niche in which the established rules of vascular miRNA signaling do not necessarily apply [16]. By revealing a pro-calcific role for EV-associated miR-145-5p in VICs, Goody et al. challenge the assumption that augmenting miR-145-5p activity would be uniformly beneficial across cardiovascular tissues. This context-dependent behavior has important implications for the interpretation of circulating miR-145-5p as a biomarker and cautions against systemic miRNA-based therapeutic strategies without tissue-specific targeting.
EV-Associated miR-145-5p as a Biomarker and Therapeutic Target: Opportunities and Challenges
The demonstration by Goody et al. that miR-145-5p is selectively enriched in EVs derived from calcified aortic valves and from the circulation of patients with CAVD immediately raises questions regarding its translational potential. EV-associated miRNAs have gained considerable attention as candidate biomarkers because EV encapsulation confers stability in the circulation and may preferentially reflect active intercellular signaling rather than passive release from damaged cells. Within this framework, the findings by Goody and colleagues position EV-miR-145-5p as a signal closely linked to disease-relevant cellular processes in the valve.
From a biomarker perspective, EV-associated miR-145-5p offers an attractive but still preliminary opportunity. The enrichment of miR-145-5p in circulating EVs from patients with CAVD suggests that valvular pathological signaling can be detected systemically. Given the absence of reliable circulating biomarkers for early-stage disease or progression, this observation is particularly noteworthy. However, as emphasized by the authors’ cross-sectional analysis, the clinical utility of EV-miR-145-5p will ultimately depend on longitudinal validation. Whether EV-miR-145-5p levels track disease severity, predict progression, or anticipate clinical endpoints such as valve replacement remains to be determined. At the same time, the work by Goody et al. highlights important limitations inherent to EV-miRNA-based biomarkers. MiR-145-5p is widely expressed across the cardiovascular system and plays established roles in vascular smooth muscle cell biology and endothelial-smooth muscle communication. Consequently, circulating EV-miR-145-5p likely represents a composite signal arising from multiple cardiovascular compartments rather than a valve-restricted readout. This issue is particularly relevant in CAVD patients, who frequently exhibit concomitant atherosclerosis or vascular remodeling. Improved approaches to define EV cellular origin, including surface marker-based enrichment or single-vesicle profiling, will be critical for enhancing specificity but are not yet ready for routine clinical use.
Beyond its potential value as a biomarker, the therapeutic implications of EV-associated miR-145-5p are even more complex [4,16]. By identifying EV-associated miR-145-5p as a driver of osteogenic reprogramming in VICs, Goody et al. provide a clear mechanistic rationale for targeting this pathway in CAVD. However, any therapeutic strategy must contend with the well-established functions of miR-145-5p in the vascular wall. Systemic inhibition of miR-145-5p could therefore produce unintended and potentially deleterious effects on vascular integrity, underscoring the risks of non-selective miRNA modulation. In this context, the study implicitly shifts attention toward more refined intervention strategies. Rather than global manipulation of miR-145-5p levels, approaches that selectively disrupt EV-mediated miR-145-5p signaling within the valve or that target downstream effectors such as ZEB2 may offer a more viable path forward. While such strategies remain technically challenging and far from clinical translation, the work by Goody et al. provides a conceptual framework for thinking about tissue-specific modulation of miRNA signaling in CAVD.
Collectively, these findings frame EV-associated miR-145-5p as both an opportunity and a cautionary example for translational cardiovascular research. The study by Goody and colleagues highlights the promise of EV-miRNAs as biomarkers and therapeutic targets, while simultaneously underscoring the necessity of context-aware interpretation and tissue-specific strategies. As such, it represents an important step toward, but not yet a solution for, miRNA-based intervention in CAVD.
Methodological Considerations and Unresolved Questions
The study by Goody et al. substantially advances the field by providing mechanistic evidence for extracellular vesicle (EV)-mediated miRNA signaling in CAVD. At the same time, their work brings into sharp focus several methodological limitations and unresolved questions that will need to be addressed before EV-associated miRNAs can be fully integrated into mechanistic models or translational strategies for valvular calcification.
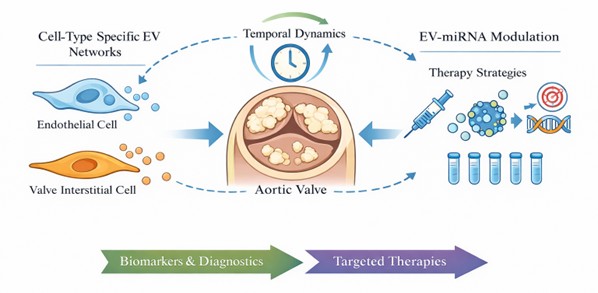
A central challenge highlighted by this study is the intrinsic heterogeneity of EV populations. As in many EV-based investigations, the EV fractions analyzed by Goody and colleagues likely represent a composite of vesicles differing in size, biogenesis, and cargo composition. Although current isolation approaches are sufficient to demonstrate biological activity, they do not allow precise attribution of miRNA signals to defined EV subtypes or biogenetic pathways. This limitation underscores the continued need for rigorous EV characterization, as emphasized by community guidelines, and suggests that future studies building on this work will benefit from integrating complementary isolation strategies and more detailed vesicle profiling. The question of cellular origin and target specificity is equally central to the interpretation of the findings by Goody et al. Within the complex cellular milieu of the aortic valve, EVs may originate from multiple cell types, including valvular endothelial cells, VICs, and infiltrating immune cells. While the authors provide strong evidence for endothelial-to-VIC EV communication in vitro, definitively establishing cell-of-origin-specific EV signaling in vivo remains challenging. Addressing this gap will require refined approaches such as surface marker-based EV enrichment or lineage-resolved EV tracing in experimental models, methodologies that are only beginning to be applied in valvular research.
The temporal dimension of EV-miRNA signaling also emerges as an important unresolved issue. The human valve samples analyzed in this and related studies predominantly reflect end-stage disease, limiting insight into the timing of EV-mediated signaling events during CAVD progression. Whether EV-associated miR-145-5p acts as an initiating signal or primarily amplifies established osteogenic programs cannot be determined from cross-sectional analyses alone. Longitudinal studies and experimental models that capture early disease stages will be essential to define the temporal role of EV-miRNA communication in valve calcification. From a mechanistic perspective, the focus on miR-145-5p provides a clear and compelling entry point, but it also highlights the inherent complexity of EV cargo. EVs deliver a constellation of miRNAs, proteins, and lipids that may converge on shared signaling pathways in recipient VICs. The identification of ZEB2 as a functional target in this study raises the possibility that EV-mediated calcific signaling operates through nodal regulatory hubs rather than single linear pathways. Future work integrating EV transcriptomic and proteomic profiling with functional perturbation experiments will be necessary to delineate these networks more comprehensively.
Finally, the translational implications of the findings by Goody et al. must be considered in light of these methodological constraints. While EV-associated miRNAs represent attractive candidates for biomarker development and therapeutic intervention, their broad expression and context-dependent effects pose significant challenges. The study implicitly argues against simplistic systemic miRNA targeting strategies and instead points toward more selective approaches that modulate EV-mediated signaling or downstream effectors within VICs. Although such strategies remain technically demanding, they represent a logical extension of the conceptual framework established by this work.
In sum, the study by Goody et al. not only provides new mechanistic insight into EV-mediated miRNA signaling in CAVD but also delineates the key methodological and conceptual questions that must be addressed to advance the field. By clarifying both what is now known and what remains uncertain, this work sets a clear agenda for future investigations aimed at translating EV-miRNA biology into meaningful clinical applications for valvular heart disease.
Conclusion
Extracellular vesicle–mediated transfer of miR-145-5p emerges as a context-dependent regulatory mechanism in calcific aortic valve disease rather than a uniformly protective or purely biomarker-associated signal. The available evidence supports a model in which EV-associated miR-145-5p contributes to osteogenic reprogramming of valvular interstitial cells, highlighting the importance of intercellular communication within the valvular microenvironment. At the same time, the dual role of miR-145-5p across cardiovascular tissues underscores the need for careful interpretation of circulating EV-associated miRNAs, as systemic measurements may reflect signals originating from multiple cellular sources. These considerations challenge simplified classifications of miRNAs as either beneficial or detrimental and instead emphasize their context-specific functions. Future progress in this field will depend on resolving key uncertainties, including the cellular origin, temporal dynamics, and target specificity of EV-mediated signaling. Addressing these challenges will be essential for determining whether EV-associated miR-145-5p can be effectively leveraged for disease monitoring or selectively targeted in a valve-specific manner (Figure 2).
Conflict of Interest
None declared.
Funding
This work was supported by the German Society of Cardiology (DGK) (to MRH); the Ernst and Bertha Grimmke Foundation; the Corona Foundation (to MRH); the Deutsche Herzstiftung (to MRH), and the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – Project-ID 397484323 – TRR 259 (Project B04).
References
2. Mathieu P, Arsenault BJ, Boulanger MC, Bossé Y, Koschinsky ML. Pathobiology of Lp(a) in calcific aortic valve disease. Expert Rev Cardiovasc Ther. 2017 Oct;15(10):797–807.
3. Mathiesen A, Hamilton T, Carter N, Brown M, McPheat W, Dobrian A. Endothelial Extracellular Vesicles: From Keepers of Health to Messengers of Disease. Int J Mol Sci. 2021 Apr 28;22(9):4640.
4. Riches-Suman K. Diverse roles of microRNA-145 in regulating smooth muscle (dys)function in health and disease. Biochem Soc Trans. 2021 Feb 26;49(1):353–63.
5. Hernández-López JR, Flores-García M, García-Flores E, Cazarín-Santos BG, Peña-Duque MA, Sánchez-Muñoz F, et al. Circulating Microvesicles Enriched in miR-126-5p and miR-223-3p: Potential Biomarkers in Acute Coronary Syndrome. Biomedicines. 2025 Feb 18;13(2):510.
6. Goody PR, Christmann D, Goody D, Hildebrand S, Billig H, Nehl D, et al. Calcific aortic valve disease augments vesicular microRNA-145-5p to regulate the calcification of valvular interstitial cells via cellular crosstalk. Basic Res Cardiol. 2025 Oct;120(5):991–1010.
7. Krohn JB, Hutcheson JD, Martínez-Martínez E, Aikawa E. Extracellular vesicles in cardiovascular calcification: expanding current paradigms. J Physiol. 2016 Jun 1;594(11):2895–903.
8. Kowal J, Arras G, Colombo M, Jouve M, Morath JP, Primdal-Bengtson B, et al. Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc Natl Acad Sci U S A. 2016 Feb 23;113(8):E968–77.
9. Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018 Nov 23;7(1):1535750.
10. Yáñez-Mó M, Siljander PR, Andreu Z, Zavec AB, Borràs FE, Buzas EI, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015 May 14;4:27066.
11. O'Brien J, Hayder H, Zayed Y, Peng C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front Endocrinol (Lausanne). 2018 Aug 3;9:402.
12. Kapustin AN, Shanahan CM. Emerging roles for vascular smooth muscle cell exosomes in calcification and coagulation. J Physiol. 2016 Jun 1;594(11):2905–14.
13. Ma X, Zhao D, Yuan P, Li J, Yun Y, Cui Y, et al. Endothelial-to-Mesenchymal Transition in Calcific Aortic Valve Disease. Acta Cardiol Sin. 2020 May;36(3):183–94.
14. Wassaifi S, Kaeffer B, Zarrouk S. Cellular Phenotypic Transformation During Atherosclerosis: The Potential Role of miRNAs as Biomarkers. Int J Mol Sci. 2025 Feb 27;26(5):2083.
15. Wang H, Shi J, Li B, Zhou Q, Kong X, Bei Y. MicroRNA Expression Signature in Human Calcific Aortic Valve Disease. Biomed Res Int. 2017;2017:4820275.
16. Moncla LM, Briend M, Bossé Y, Mathieu P. Calcific aortic valve disease: mechanisms, prevention and treatment. Nat Rev Cardiol. 2023 Aug;20(8):546–59.