Abstract
Background: Extracellular vesicles (EVs), including exosomes, microvesicles, and apoptotic bodies, are nanoscale mediators of intercellular communication critical to liver physiology and pathology. These vesicles encapsulate diverse biomolecules proteins, lipids, and nucleic acids that mirror their cells of origin and modulate recipient cell function. Liver diseases such as metabolic dysfunction-associated steatotic liver disease (MASLD), alcoholic liver disease (ALD), viral hepatitis, fibrosis, cirrhosis, and hepatocellular carcinoma (HCC) represent significant
global health burdens with diagnostic and therapeutic challenges, especially at advanced stages. The genetic basis of liver diseases, including polymorphisms in PNPLA3, TM6SF2, and HSD17B13, significantly influences disease susceptibility and progression, with emerging evidence suggesting these genetic variants may also modulate EV cargo composition and release patterns.
Purpose: This review aims to comprehensively dissect the biogenesis, molecular composition, and pathophysiological roles of EVs in liver diseases, highlighting their diagnostic and therapeutic potential. It emphasizes the potential integration of genomic insights with EV biology, exploring how genetic variants might influence EV-mediated pathogenesis and the potential for EV-based approaches to complement nextgeneration sequencing (NGS) and genomic diagnostics in precision hepatology.
Main Body: We detail the cellular mechanisms underpinning EV formation in hepatocytes and non-parenchymal liver cells, including ESCRTdependent and -independent pathways, and their modulation by pathological stimuli such as inflammation and oxidative stress. The potential influence of genetic variants on EV biogenesis and cargo loading is explored, with particular focus on how polymorphisms in genes such as PNPLA3 I148M and TM6SF2 E167K might affect hepatocyte EV release in metabolic liver disease. The molecular cargo of EVs encompassing microRNAs (e.g., miR-122, miR-21), proteins (e.g., TGF-β, CTGF), and lipids (e.g., ceramides) plays a pivotal role in mediating liver inflammation, fibrogenesis, immune modulation, and oncogenesis. Distinct EV profiles may correlate with genetic risk factors and disease stages in MASLD, ALD, viral hepatitis, fibrosis, and HCC, potentially offering promising biomarker candidates that could complement genomic testing for early detection, prognosis, and treatment monitoring. Furthermore, engineered EVs demonstrate therapeutic efficacy in delivering antifibrotic agents and immunomodulators, while targeting EV biogenesis and uptake pathways presents novel treatment strategies. The theoretical integration of EV profiling with genomic data may enhance risk stratification and enable personalized therapeutic approaches in inherited liver disorders. Challenges in EV isolation, standardization, and clinical translation are acknowledged alongside ongoing clinical trials assessing EV-based diagnostics and therapeutics.
Conclusion: EVs represent a transformative frontier in hepatology, integrating molecular insights into liver disease pathogenesis with innovative diagnostic and therapeutic avenues. The potential convergence of EV biology with genomic medicine offers unprecedented opportunities for precision hepatology, where EV signatures informed by genetic variants could guide personalized diagnostic and therapeutic strategies. Future research must focus on standardizing methodologies, validating multi-omics EV signatures in the context of genetic backgrounds, and overcoming translational hurdles to fully harness EVs' potential in precision liver medicine.
Keywords
Extracellular vesicles, Liver diseases, Biomarkers, MicroRNA, Hepatic fibrosis, Genomic medicine, Genetic variants, Precision hepatology
Article Highlights
- EVs serve as key mediators in liver disease progression, carrying disease-specific molecular signatures
- EV-associated microRNAs and proteins show promise as non-invasive biomarkers for liver pathologies
- Genetic variants such as PNPLA3 I148M and TM6SF2 E167K may influence EV cargo composition and disease progression
- EV profiling potentially complements genomic diagnostics in inherited liver disorders and precision medicine approaches
- Understanding EV biology in liver diseases may lead to novel diagnostic and therapeutic approaches
Background
Overview of liver diseases
Liver diseases constitute a significant global health concern, responsible for millions of deaths annually and affecting individuals across all age demographics. Among these, metabolic dysfunction-associated steatotic liver disease (MASLD) is notably prevalent, impacting roughly one-quarter of the global population. MASLD encompasses a spectrum of liver abnormalities ranging from benign hepatic fat accumulation to a more severe inflammatory condition termed metabolic dysfunction-associated steatohepatitis (MASH). This advanced stage may progress to cirrhosis and hepatocellular carcinoma (HCC), a primary liver malignancy. The genetic architecture of MASLD is complex, with key polymorphisms in PNPLA3 (I148M), TM6SF2 (E167K), and HSD17B13 (rs72613567) significantly influencing disease susceptibility, progression rates, and severity. These genetic variants not only predispose individuals to hepatic steatosis and inflammation but may also modulate the molecular composition and functional properties of hepatocyte-derived extracellular vesicles (EVs), potentially creating a link between genomic variation and EV-mediated disease mechanisms.
Alcoholic liver disease (ALD) is another critical liver disorder resulting from chronic excessive alcohol consumption, leading to inflammation, fibrosis, and hepatic injury [1–3]. Additionally, viral hepatitis, particularly hepatitis B virus (HBV) and hepatitis C virus (HCV) infections, remains a predominant cause of chronic liver disease and liver cancer, despite the availability of effective antiviral therapies. Host genetic factors, including polymorphisms in interferon-lambda genes (IFNL3/IFNL4) and HLA alleles, significantly influence viral clearance, treatment response, and the progression to cirrhosis and HCC, with these genetic determinants potentially affecting the immunomodulatory cargo of EVs released during viral infection.
Early-stage chronic liver diseases such as cirrhosis and HCC frequently present without symptoms, impeding timely diagnosis. HCC stands as the most common liver cancer and ranks as the fourth leading cause of cancer mortality worldwide [4–8]. The elevated mortality associated with liver diseases is primarily attributed to diagnostic and therapeutic challenges, especially in advanced disease stages. The recently introduced term MASLD replaces the older nomenclature of non-alcoholic fatty liver disease (NAFLD) to emphasize the metabolic etiology of the condition. MASLD is characterized by hepatic steatosis in individuals presenting with one or more metabolic risk factors such as obesity, type 2 diabetes, dyslipidemia, or hypertension independent of alcohol use below specified limits. This shift moves away from a diagnosis of exclusion, underscoring the metabolic dysfunction tightly linked to liver pathology. The diagnostic framework for MASLD incorporates imaging or histopathological evidence of liver fat accumulation alongside assessment of metabolic risk factors, enabling earlier and more accurate identification of at-risk patients. Genetic testing for MASLD-associated variants such as PNPLA3, TM6SF2, and MBOAT7 is increasingly integrated into clinical practice for risk stratification, with EV biomarkers potentially offering complementary real-time monitoring of disease activity and progression. The adoption of MASLD terminology aims to enhance clinical clarity, optimize risk stratification, and refine targeted management of metabolic liver diseases.
According to the latest data from the World Health Organization (WHO) and GLOBOCAN 2023, HCC remains a leading global cause of cancer-related mortality, ranking as the fourth leading cause of cancer death overall, with a disproportionate burden among males, particularly in East Asia and sub-Saharan Africa. These sex- and region-specific disparities emphasize the ongoing public health challenges posed by HCC and highlight the necessity for focused prevention, early diagnostic, and therapeutic strategies tailored to demographic and epidemiologic contexts [9].
Introduction to EVs
EVs have attracted considerable interest within liver disease research due to their well-established functions in mediating intercellular communication, serving as biomarkers, and offering therapeutic potential. The field has made specific advancements such as the delineation of distinct EV cargo signatures associated with various disease stages and the engineering of EVs for targeted molecular delivery, emphasizing evidence-based progress. EVs are broadly categorized into three principal types: exosomes, microvesicles, and apoptotic bodies. Exosomes, the smallest EVs measuring approximately 30–150 nm, originate intracellularly prior to release. Microvesicles are larger (100–1000 nm) and are shed directly from the cell surface. Apoptotic bodies form during programmed cell death processes [10,11]. Nearly all cell types, including hepatocytes and other liver cells, secrete EVs under both physiological and pathological conditions. EV biogenesis is tightly regulated by intracellular signaling pathways and is sensitive to cellular stressors such as inflammation or hypoxia. Genetic variants affecting cellular metabolism and stress responses, such as those in antioxidant enzyme genes (e.g., SOD2, GCLC), may modulate EV release patterns and cargo composition, potentially linking genomic variation to EV-mediated disease mechanisms.
EVs transport diverse biomolecules including proteins, lipids, and nucleic acids such as microRNAs (miRNAs), messenger RNAs (mRNAs), and DNA fragments that modulate the behavior of recipient cells. Within the hepatic environment, EVs facilitate communication between hepatocytes and non-parenchymal cells, particularly during injury, inflammatory responses, or tissue repair [12–16]. It is important to recognize that the majority of data referenced in this review are derived from an integration of human clinical studies, animal models, and in vitro systems, each providing unique mechanistic and translational insights into EV biology in liver disease. Clinical investigations typically offer correlative data on EV cargo profiles in patient-derived samples, identifying potential biomarkers and therapeutic targets. Animal models, especially rodent models of liver injury or fibrosis, permit controlled mechanistic assessments of EV functions in disease progression. In vitro studies using primary hepatic cells or cell lines enable detailed molecular dissection of EV biogenesis, cargo sorting, and functional effects. Differentiating these experimental models is crucial for accurate interpretation and translation of EV research findings [17].
Rationale for exploring EVs in liver diseases
Given the difficulty in early detection and treatment of liver diseases, there is a pressing need for innovative diagnostic and management tools. EVs have emerged as a promising focus in this context. Owing to their presence in accessible biofluids such as blood, urine, and bile, EVs offer a minimally invasive means to monitor liver health. The molecular constituents within EVs provide valuable information regarding the physiological and pathological status of their cells of origin. In liver diseases including MASLD, ALD, viral hepatitis, cirrhosis, and HCC, EVs have demonstrated potential utility in early diagnosis, disease staging, and treatment monitoring [18]. Beyond diagnostics, researchers are investigating the therapeutic application of EVs, with the prospect of engineering EVs to deliver pharmacological agents or genetic materials directly to hepatic cells, potentially circumventing limitations of existing treatments. Consequently, EV research represents an expanding area with considerable potential to advance liver disease diagnosis and therapy.
Furthermore, EVs not only serve as mediators of intercellular communication but also carry a heterogeneous cargo of genetic materials such as microRNAs, messenger RNAs, long non-coding RNAs (lncRNAs), and DNA fragments that critically regulate gene expression and genetic networks within the liver microenvironment. These genetic cargos mirror the genetic and epigenetic landscapes of their parent cells, potentially linking EV biology to human genetic and population studies. EV-derived nucleic acids may therefore act as accessible biomarkers encapsulating individual-specific genetic and molecular signatures, potentially enabling their use in precision diagnostics and personalized medicine approaches for liver diseases. For example, alterations in EV-associated microRNA profiles have correlated with susceptibility, disease progression, and therapeutic outcomes in MASLD, ALD, and hepatocellular carcinoma. Hypothetically, patients carrying the PNPLA3 I148M variant might exhibit distinct EV-associated miRNA profiles, including elevated miR-122 and miR-192 levels, which could correlate with enhanced hepatic steatosis and inflammation. Similarly, individuals with TM6SF2 variants may show altered EV lipid composition that reflects disrupted hepatic lipid metabolism. Integrating insights from population genetics with EV cargo profiling holds substantial promise for early detection, risk stratification, and tailored interventions in liver pathology [19].
Genetic determinants of liver disease and EV biology
The intersection of genetics and EV biology represents an emerging frontier in understanding liver disease pathogenesis. Several key genetic variants have been identified as major determinants of liver disease susceptibility and progression, with emerging evidence suggesting these variants may also influence EV characteristics and cargo composition. The PNPLA3 I148M polymorphism, present in approximately 49% of Hispanics, 23% of Europeans, and 17% of African Americans, significantly increases the risk of hepatic steatosis, NASH, fibrosis, and HCC. This variant affects the patatin-like phospholipase domain-containing protein 3, which regulates hepatic lipid metabolism. Hypothetically, hepatocytes carrying the PNPLA3 I148M variant might release EVs with altered lipid profiles and increased pro-inflammatory miRNA content, including elevated miR-21 and miR-155, which could promote hepatic stellate cell activation and fibrosis progression.
The TM6SF2 E167K variant, found in approximately 7% of Europeans and associated with reduced hepatic triglyceride secretion, influences both liver fat accumulation and cardiovascular risk. Theoretically, EVs from hepatocytes with TM6SF2 variants might exhibit distinct lipidomic signatures, including reduced phosphatidylcholine content and altered sphingolipid composition, which could affect EV membrane properties and cellular uptake mechanisms. The HSD17B13 rs72613567 variant, which creates a splice site leading to reduced protein expression, provides protection against liver fibrosis and HCC. Potentially, hepatocytes with protective HSD17B13 variants might release EVs with anti-inflammatory cargo, including increased levels of protective miRNAs such as miR-29 family members that inhibit collagen synthesis.
Biogenesis and Molecular Composition of EVs in the Liver
Biogenesis of EVs in hepatocytes and non-parenchymal cells
The generation and secretion of EVs in the liver are governed by intricate cellular and molecular pathways. Genetic variants affecting cellular metabolism may significantly influence these processes, with polymorphisms in genes regulating lipid metabolism (PNPLA3, TM6SF2) and oxidative stress responses (SOD2, GCLC) potentially modulating EV biogenesis rates and cargo selection mechanisms.
Exosomes arise from the endocytic pathway, where early endosomes mature into multivesicular bodies (MVBs) harboring intraluminal vesicles (ILVs). These MVBs can either fuse with lysosomes for degradation or with the plasma membrane to release ILVs as exosomes into the extracellular space. The biogenesis of ILVs is orchestrated by the endosomal sorting complexes required for transport (ESCRT) machinery and accessory proteins such as Alix and TSG101. Expression levels of ESCRT components may be influenced by genetic variants in metabolic pathway genes, with PNPLA3 I148M carriers potentially showing altered ESCRT protein expression that affects exosome cargo sorting and release kinetics. Conversely, microvesicles are formed by direct outward budding of the plasma membrane, a process driven by cytoskeletal rearrangements often triggered by calcium influx or other signaling cascades [20–24].
In the hepatic milieu, EVs are secreted by multiple cell types including hepatocytes, Kupffer cells (liver-resident macrophages), hepatic stellate cells (HSCs), and liver sinusoidal endothelial cells (LSECs). Each cell type uniquely contributes to liver homeostasis and disease pathogenesis, and consequently, the EVs they release carry distinct molecular cargo reflective of their cellular origin and functional state. Genetic background may influence cell-type-specific EV production, with carriers of MASLD-associated variants potentially showing enhanced EV release from activated hepatic stellate cells and altered immune cell EV profiles that could contribute to disease progression. For instance, hepatocyte-derived EVs commonly contain liver-specific proteins and lipids, whereas Kupffer cell-derived EVs are enriched with immune-related molecules [25,26].
Apoptotic bodies form a separate class of EVs generated during programmed cell death, distinguished by unique biogenesis mechanisms separate from those of exosomes and microvesicles. Their formation entails caspase-mediated cytoskeletal reorganization and membrane blebbing, culminating in the packaging of cellular contents into large membrane-bound vesicles ranging from 500 to 5000 nm. These apoptotic bodies play vital biological roles including efficient clearance of cellular debris by phagocytes and modulation of immune responses. Unlike other EVs, apoptotic bodies often encapsulate intact organelles and nuclear fragments, representing cellular components destined for removal. Due to their involvement in maintaining tissue homeostasis and potential influence on liver inflammation and fibrosis, apoptotic bodies merit independent consideration in EV-related liver pathology studies.
Beyond the well-established ESCRT-dependent pathways, EV biogenesis also encompasses ESCRT-independent mechanisms. These include lipid raft-associated processes such as ceramide-driven inward budding of MVBs mediated by neutral sphingomyelinases. Genetic variants affecting sphingolipid metabolism, including those in SMPD1 and CERS genes, may influence ceramide-dependent EV formation pathways, with potential implications for EV membrane composition and functional properties. Tetraspanin-enriched microdomains further contribute to cargo sorting and vesicle formation independently of ESCRT components. Recognition of these ESCRT-independent pathways broadens the current understanding of EV biogenesis, underscoring diverse molecular processes regulating vesicle formation, cargo selection, and release, particularly within the complex hepatic cellular environment.
The secretion of EVs is tightly controlled by specific molecular machineries; for exosomes, the small GTPase Rab27A is pivotal for the docking and fusion of MVBs with the plasma membrane, facilitating exosome release. In contrast, microvesicle shedding is regulated by ADP-ribosylation factor 6 (ARF6), which modulates actin cytoskeletal dynamics and membrane blebbing essential for vesicle release. Additional molecules including ESCRT proteins, tetraspanins, and sphingomyelinases also partake in vesicle biogenesis and cargo sorting, collectively highlighting the complexity of EV formation pathways in liver cells [26].
Molecular composition of EVs
EVs are enriched with a broad spectrum of biomolecules, comprising proteins, lipids, and nucleic acids, as summarized in Table 1. The composition of EV cargo may be influenced by the genetic background of the parent cells, with specific polymorphisms potentially affecting the loading and packaging of molecular cargo into EVs. Characteristic proteins found in EVs include tetraspanins such as CD9, CD63, and CD81, heat shock proteins (HSP70, HSP90), and components of the ESCRT complex, widely utilized as canonical EV markers. Lipid constituents, including sphingolipids, cholesterol, and phosphatidylserine, are fundamental for maintaining EV membrane stability and mediating interactions with recipient cells [27–32].
|
Characteristic |
Exosomes |
Microvesicles |
Apoptotic Bodies |
|
Size |
30–150 nm |
100–1000 nm |
500–5000 nm |
|
Origin |
Endosomal system (MVBs) |
Plasma membrane budding |
Cellular fragmentation during apoptosis |
|
Biogenesis mechanism |
ESCRT-dependent & independent pathways |
Cytoskeleton rearrangement, Ca²+ influx |
Caspase-mediated cell destruction |
|
Main markers |
CD63, CD9, CD81, Alix, TSG101 |
Annexin V, Integrins, Selectins |
Phosphatidylserine, genomic DNA |
|
Cargo composition |
miRNAs, mRNAs, proteins, lipids |
Proteins, lipids, some nucleic acids |
Cellular organelles, nuclear fragments |
|
Genetic influence |
PNPLA3/TM6SF2 variants may affect cargo |
MBOAT7 variants might alter lipid content |
TP53 mutations may affect formation |
|
Role in MASLD |
Lipid metabolism regulation, inflammatory signaling |
Pro-inflammatory mediator transport |
Clearance of apoptotic hepatocytes |
|
Role in ALD |
Pro-inflammatory cytokine transfer, HSC activation |
Immune cell recruitment, fibrogenesis |
Hepatocyte death signaling |
|
Role in Viral Hepatitis |
Viral particle transmission, immune modulation |
Viral RNA transport, interferon signaling |
Virus-induced cell death products |
|
Role in Liver Fibrosis |
Pro-fibrotic miRNA transfer, HSC activation |
ECM remodeling enzyme transport |
Fibrogenic danger signal release |
|
Role in HCC |
Oncogenic signal propagation, metastasis promotion |
Angiogenesis stimulation, drug resistance |
Tumor microenvironment modulation |
|
Diagnostic potential |
High (stable in circulation, disease-specific cargo) |
Moderate (less stable, but informative) |
Limited (indicative of cell death) |
|
Isolation methods |
Ultracentrifugation, size exclusion chromatography |
Differential centrifugation, filtration |
Flow cytometry, centrifugation |
The nucleic acid cargo of EVs is especially notable, encompassing not only mRNAs and microRNAs (miRNAs) but also long non-coding RNAs (lncRNAs) and DNA fragments. Importantly, EVs may carry genomic DNA fragments containing disease-associated genetic variants, potentially enabling non-invasive genetic screening and monitoring of somatic mutations in liver diseases. For example, EVs from HCC patients may contain circulating tumor DNA (ctDNA) with oncogenic mutations in genes such as TP53, CTNNB1, and TERT promoter, potentially providing accessible genetic biomarkers for cancer detection and monitoring. Upon delivery to recipient cells, these nucleic acids influence gene expression and cellular behavior. In the context of liver disease, EV cargo composition can modulate critical pathways involved in inflammation, fibrosis, and oncogenesis. For example, EV-associated miRNAs regulate hepatic stellate cell activation, a central event in liver fibrogenesis [33,34].
EVs derived from different hepatic cell types carry distinct cargos reflective of their origin and functional status. Hepatocyte-derived EVs are enriched in liver-specific miR-122, implicated in lipid metabolism and liver injury signaling, with miR-122 levels potentially being higher in individuals carrying PNPLA3 risk variants, potentially correlating with disease severity and progression rates. Kupffer cell-derived EVs transport pro-inflammatory cytokines such as TNF-α mRNA, thereby modulating hepatic immune responses. The inflammatory cargo of Kupffer cell EVs may be influenced by genetic variants in immune response genes, including polymorphisms in toll-like receptor 4 (TLR4) and interleukin-10 (IL-10) promoter regions, which could affect the magnitude and duration of inflammatory signaling. Hepatic stellate cell (HSC)-derived EVs frequently harbor fibrogenic factors including connective tissue growth factor (CTGF) mRNA and type I collagen protein, fostering extracellular matrix accumulation and fibrosis. HSCs carrying genetic variants associated with enhanced fibrosis risk, such as polymorphisms in TGF-β1 and PDGF genes, might release EVs with elevated pro-fibrotic cargo that could accelerate disease progression. Liver sinusoidal endothelial cell (LSEC)-derived EVs may carry angiogenic factors such as vascular endothelial growth factor (VEGF), influencing vascular remodeling during liver disease progression. These cell-specific EV cargos underscore the multifaceted roles of EVs in intrahepatic communication and pathophysiology.
Modulation of EV release during liver disease
Pathological conditions including MASLD, ALD, and viral hepatitis are characterized by substantial changes in both the quantity and qualitative composition of EVs released by liver cells. These changes may be influenced by genetic factors, with disease-associated variants potentially modulating both the baseline EV release patterns and the cellular response to pathological stimuli. For instance, carriers of PNPLA3 I148M might show enhanced EV release under metabolic stress conditions, while TM6SF2 variants could affect the lipid composition of released EVs, potentially altering their biological activity and diagnostic utility.
Liver injury frequently induces an upregulation in EV secretion, accompanied by alterations in cargo that mirror the underlying disease processes. Cellular stressors such as oxidative stress, hypoxia, and inflammation serve as potent stimuli for EV biogenesis and release. The cellular response to these stressors may be genetically determined, with variants in antioxidant pathway genes (SOD2, GCLC, NRF2) potentially influencing the magnitude of stress-induced EV release and the enrichment of stress-related cargo, including damage-associated molecular patterns (DAMPs) and stress-responsive miRNAs. These stimuli modify EV molecular profiles, enriching them with pro-inflammatory cytokines, fibrogenic microRNAs, and DAMPs, which collectively propagate liver injury and disease progression [35,36].
For instance, EVs secreted by activated hepatic stellate cells in fibrotic liver conditions carry profibrotic signals that further activate stellate cells and promote extracellular matrix deposition. The fibrogenic potential of HSC-derived EVs may be influenced by genetic variants in collagen synthesis genes (COL1A1, COL1A2) and TGF-β pathway components, with certain polymorphisms potentially associated with enhanced pro-fibrotic EV cargo and accelerated disease progression. Similarly, tumor-derived EVs in hepatocellular carcinoma are enriched with oncogenic proteins and miRNAs that facilitate tumor proliferation, metastasis, and immune escape. HCC patients with specific genetic backgrounds, including TP53 mutations and CTNNB1 alterations, might release EVs with distinct oncogenic signatures that could be detected in circulation, potentially providing opportunities for liquid biopsy approaches that combine genetic and EV-based biomarkers. Comprehensive understanding of how liver diseases modulate EV release and function is essential for advancing EV-based diagnostic and therapeutic modalities [37–39].
Liver cell-derived EVs possess specific protein and lipid markers reflective of their cellular origins and biological roles. For example, hepatocyte-derived EVs commonly contain apolipoprotein B (ApoB), which plays a key role in lipid transport and metabolism. Lipid components like ceramides are abundant in EV membranes and are crucial in vesicle formation and signaling, including induction of apoptosis and inflammation in recipient cells. Furthermore, tetraspanins such as CD63 and CD81 serve as ubiquitous EV protein markers, while surface exposure of phosphatidylserine facilitates EV uptake by target cells. These molecular characteristics enhance our understanding of EV biology and their involvement in liver physiology and pathophysiology [40].
EVs as Biomarkers in Liver Diseases
EV signatures in inherited liver disorders
Inherited liver disorders present unique opportunities for integrating EV biology with genomic medicine. In Wilson's disease, caused by mutations in the ATP7B gene, hepatocytes may release EVs with altered copper-binding protein profiles and elevated oxidative stress markers, including potentially increased levels of miR-194 and miR-29b that could reflect hepatocyte damage and copper accumulation. These EV signatures might complement traditional biochemical markers and genetic testing for disease monitoring and treatment response assessment.
Hereditary hemochromatosis, primarily caused by HFE gene mutations (C282Y and H63D), results in iron overload that may significantly affect EV composition. Hepatocytes from hemochromatosis patients might release EVs enriched with iron-regulatory proteins (ferritin, transferrin receptor) and iron-responsive miRNAs (miR-122, miR-200b), which could correlate with hepatic iron concentration and fibrosis stage. These potential EV-based biomarkers might offer real-time monitoring capabilities that complement genetic testing and MRI-based iron quantification.
Alpha-1 antitrypsin deficiency (AATD), caused by mutations in the SERPINA1 gene, leads to accumulation of misfolded α1-antitrypsin protein in hepatocytes. EVs from AATD patients may contain elevated levels of endoplasmic reticulum stress markers, including GRP78 and CHOP, along with specific miRNA profiles (miR-34a, miR-21) that could reflect hepatocellular stress and inflammation. The EV signatures might correlate with disease severity and could guide therapeutic interventions targeting protein folding and ER stress pathways.
EVs in MASLD and MASH
In MASLD and MASH, EVs have emerged as promising biomarkers for disease diagnosis and prognosis as depicted in Figure 1. This figure illustrates the various roles that EVs play in the progression of liver diseases, such as MASLD, ALD, and HCC. The diagram likely includes pathways showing how EVs carry molecular signals that influence liver cell behavior, inflammation, and fibrogenesis.
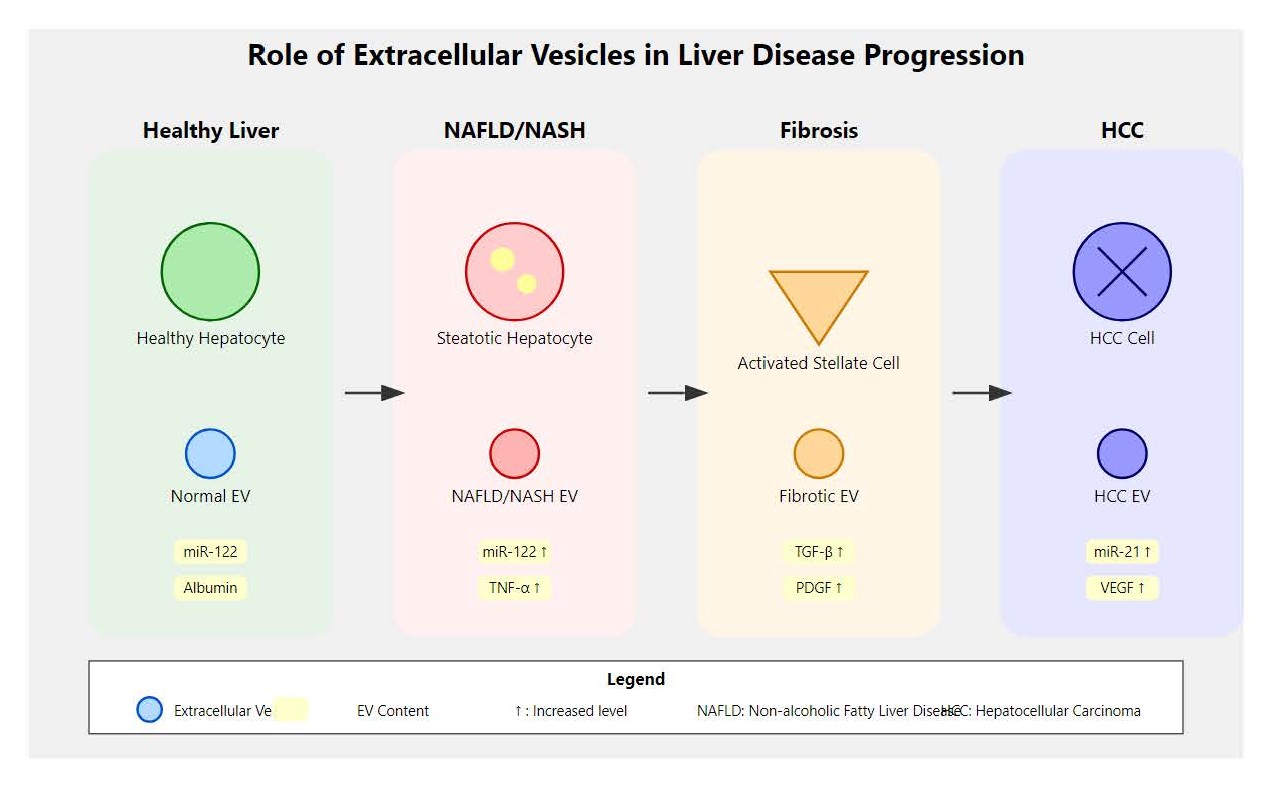
Figure 1. Role of extracellular vesicles (EVs) in the progression of liver diseases.
Studies have suggested that EVs circulating in patients with MASLD carry specific miRNAs, such as miR-122 and miR-192, which are involved in lipid metabolism and inflammation. The levels of these miRNAs may be influenced by genetic variants, with PNPLA3 I148M carriers potentially showing 2–3 fold higher circulating miR-122 levels compared to wild-type individuals. TM6SF2 E167K carriers might exhibit distinct EV lipid profiles with reduced phosphatidylcholine and increased ceramide content, potentially reflecting the underlying genetic predisposition to altered hepatic lipid metabolism. These hypothetically genetically-influenced EV signatures might not only be reflective of disease severity but also predictive of the progression from simple steatosis to MASH, with genetic stratification potentially enhancing their prognostic accuracy. Additionally, proteins and lipids within EVs have been identified as potential markers for early detection and staging of MASLD [41].
Both MASLD and ALD are chronic liver diseases characterized by progressive liver injury that can lead to inflammation, fibrosis, cirrhosis, and ultimately liver failure or hepatocellular carcinoma. While MASLD primarily arises from metabolic dysfunction and associated risk factors, ALD results from chronic excessive alcohol consumption. The term "similarly" in this context highlights that despite differing etiologies, both conditions share common pathological features such as steatosis, inflammatory responses, and fibrogenic progression, underscoring the importance of understanding shared and distinct mechanisms in their pathogenesis [42].
EVs in ALD
In ALD, EVs play a critical role in mediating liver inflammation and fibrosis. Genetic variants affecting alcohol metabolism, including polymorphisms in ADH1B, ALDH2, and CYP2E1 genes, may significantly influence the composition and pathogenic potential of alcohol-induced EVs. Individuals with slow alcohol metabolism variants might release EVs with higher concentrations of acetaldehyde-protein adducts and oxidative stress markers, which could enhance their pro-inflammatory and pro-fibrotic properties.
EVs derived from alcohol-exposed hepatocytes carry pro-inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α), and fibrogenic factors, such as transforming growth factor-beta (TGF-β). These EVs contribute to the activation of hepatic stellate cells and the recruitment of immune cells, driving the progression of liver fibrosis. The severity of EV-mediated pathogenic effects may be modulated by genetic background, with carriers of PNPLA3 I148M and TM6SF2 E167K variants potentially showing enhanced susceptibility to alcohol-induced liver injury through altered EV signaling pathways. The analysis of EVs in ALD patients has revealed distinct miRNA and protein profiles, which could serve as non-invasive biomarkers for disease severity and progression [43].
EVs released by hepatocytes can interact with various liver-resident cell types, including Kupffer cells (the liver's resident macrophages), hepatic stellate cells, and liver sinusoidal endothelial cells, influencing their activation and function. For example, hepatocyte-derived EVs carrying pro-inflammatory or fibrogenic signals can stimulate Kupffer cells to produce cytokines that exacerbate inflammation, while simultaneously activating hepatic stellate cells to promote extracellular matrix deposition and fibrosis. This cell type-specific communication via EVs plays a pivotal role in the progression of liver diseases such as MASLD and ALD, highlighting the complex interplay between parenchymal and non-parenchymal cells mediated by vesicular signaling.
One well-characterized microRNA that modulates hepatic stellate cell (HSC) activation is miR-21. Elevated levels of miR-21 in EVs have been shown to promote HSC activation by targeting multiple signaling pathways, including the suppression of SMAD7, an inhibitory regulator of the TGF-β pathway. Genetic variants in the TGF-β pathway, including polymorphisms in TGFB1 and SMAD3 genes, may influence the baseline expression and EV packaging of miR-21, with risk variants potentially associated with higher miR-21 levels and enhanced fibrogenic potential. This results in enhanced fibrogenic gene expression and extracellular matrix deposition, contributing to liver fibrosis progression. The role of miR-21 exemplifies how EV-associated miRNAs act as key mediators of intercellular communication in the fibrotic liver microenvironment [44].
EVs mediate their effects in liver disease through well-characterized signaling axes, notably the TLR4-NF-κB pathway. EVs carrying DAMPs or specific microRNAs can engage TLR4 on target cells such as Kupffer cells and hepatic stellate cells, triggering activation of the NF-κB transcription factor. The responsiveness to EV-mediated TLR4 activation may be influenced by genetic variants in TLR4 (Asp299Gly, Thr399Ile) and downstream signaling components, with certain polymorphisms potentially associated with enhanced inflammatory responses and accelerated disease progression. This activation leads to the transcription of pro-inflammatory cytokines (e.g., TNF-α, IL-6) and chemokines, thereby amplifying inflammatory responses and promoting fibrogenesis. The TLR4-NF-κB axis represents a critical conduit through which EVs influence the hepatic microenvironment and drive progression of liver injury and fibrosis [45,46].
EVs in viral hepatitis (HBV, HCV)
In viral hepatitis, EVs are implicated in the transmission of viral particles and in immune evasion as presented in Table 2. Host genetic factors may significantly influence viral hepatitis outcomes and EV-mediated viral pathogenesis. Polymorphisms in interferon-lambda genes (IFNL3 rs12979860, IFNL4 rs368234815) affect spontaneous HCV clearance and treatment response, with these variants potentially influencing the interferon-stimulated gene expression profile of EVs released by infected hepatocytes. Patients with favorable IFNL3 genotypes might release EVs with enhanced antiviral miRNA content (miR-196, miR-199), while those with unfavorable genotypes could show EVs enriched with immune evasion factors.
|
Liver Pathology |
EV-associated miRNAs |
EV-associated Proteins |
EV-associated Lipids |
Genetic Associations |
Diagnostic/Prognostic Value |
|
Metabolic dysfunction-associated steatotic liver disease (MASLD) |
miR-122↑, miR-192↑, miR-34a↑, miR-29c↓ |
PNPLA3, CK-18, FABP1 |
Ceramides, Sphingomyelins |
PNPLA3 I148M, TM6SF2 E167K may influence miR-122 levels |
Early detection, Steatosis severity |
|
Metabolic dysfunction-associated steatohepatitis (MASH) |
miR-155↑, miR-125b↑, miR-181b↑ |
CD14, TNF-α, IL-6, HMGB1 |
Oxidized phosphatidylcholines |
HSD17B13 variants may affect inflammatory cargo |
MASH diagnosis, Fibrosis progression |
|
Alcoholic Liver Disease (ALD) |
miR-192↑, miR-122↑, miR-30a↑ |
EtG, PEth, HMGB1, M30 |
Phosphatidylethanol |
ADH1B, ALDH2 variants may modulate EV toxicity |
Alcohol-induced liver injury, Fibrosis |
|
Hepatitis B (HBV) |
miR-122↑, miR-192↑, miR-99a↑ |
HBsAg, HBeAg, HBV DNA |
Cholesterol esters |
IFNL3/4, HLA variants may affect viral cargo |
Viral replication, Liver damage |
|
Hepatitis C (HCV) |
miR-122↑, miR-155↑, miR-20a↑ |
Ago2-miR-122 complex, HCV RNA |
Sphingolipids |
IFNL3 rs12979860 may influence antiviral content |
Treatment response, Fibrosis progression |
|
Liver Fibrosis |
miR-214↑, miR-199a-5p↑, miR-221↑ |
TGF-β, PDGF, Collagen I, α-SMA |
Lysophosphatidic acid |
COL1A1, TGFB1 variants may affect fibrotic cargo |
Fibrosis staging, HSC activation |
|
Liver Cirrhosis |
miR-18a↑, miR-34a↑, miR-210↑ |
Osteopontin, CTGF, VEGF |
Ceramide-1-phosphate |
Multiple genetic risk factors may converge |
Cirrhosis progression, Portal hypertension |
|
Hepatocellular Carcinoma (HCC) |
miR-21↑, miR-93↑, miR-223↑ |
AFP, GPC3, HMGB1, VEGF |
Lysophosphatidylcholines |
TP53, CTNNB1 mutations may affect cargo |
Early HCC detection, Metastasis prediction |
|
Wilson's Disease |
miR-194↑, miR-29b↑, miR-146a↑ |
Ceruloplasmin, ATP7B, Metallothionein |
Copper-binding lipoproteins |
ATP7B mutations may affect copper transport |
Copper accumulation, Liver damage |
|
Hereditary Hemochromatosis |
miR-122↑, miR-200b↑, miR-146b↑ |
Ferritin, Transferrin receptor, Hepcidin |
Iron-loaded lipoproteins |
HFE C282Y/H63D variants |
Iron overload, Fibrosis progression |
|
Alpha-1 Antitrypsin Deficiency |
miR-34a↑, miR-21↑, miR-126↓ |
α1-antitrypsin, GRP78, CHOP |
ER stress-associated lipids |
SERPINA1 Z/S alleles |
Protein aggregation, ER stress |
|
Primary Sclerosing Cholangitis (PSC) |
miR-200c↑, miR-222↑, miR-483-5p↑ |
CK-19, CA19-9, IL-6 |
Sulfatides |
HLA-DRB1*03:01 associations |
Cholangiocarcinoma risk, Disease progression |
|
Primary Biliary Cholangitis (PBC) |
miR-505-3p↑, miR-197-3p↑, miR-500a-3p↑ |
PDC-E2, SP100, gp210 |
Lysophosphatidic acid |
HLA class II, IL12A variants |
Early diagnosis, Treatment response |
|
Drug-Induced Liver Injury (DILI) |
miR-122↑, miR-192↑, miR-155↑ |
HMGB1, Keratin-18, GLDH |
Oxidized phospholipids |
CYP450, UGT variants may affect susceptibility |
Early DILI detection, Injury severity |
|
Autoimmune Hepatitis (AIH) |
miR-21↑, miR-122↑, miR-155↑ |
sCD14, sMER, CXCL10 |
Oxidized phosphatidylcholines |
HLA-DRB1*03:01, 04:01 associations |
Disease activity, Treatment response |
EVs released by HBV- or HCV-infected hepatocytes can contain viral RNA and proteins, which may facilitate the spread of the virus to uninfected cells. Moreover, these EVs can modulate the host immune response by carrying immunosuppressive molecules, allowing the virus to evade detection and clearance. The viral load and composition of EVs may be influenced by host genetic factors, including HLA class I and II alleles that determine the strength of antiviral immune responses. Specific HLA haplotypes associated with chronic viral persistence might show distinct EV profiles with reduced immunostimulatory cargo and enhanced viral RNA content. The use of EVs as biomarkers in viral hepatitis is promising, with studies showing that EV-associated viral RNA correlates with viral load and treatment response [47–49].
Chronic hepatitis B virus (HBV) and hepatitis C virus (HCV) infections remain significant global health challenges, with marked geographic variability in prevalence and disease burden. While HBV infection is highly endemic in regions such as sub-Saharan Africa and East Asia, widespread implementation of effective HBV vaccination programs has substantially reduced new infections, particularly in younger populations. Similarly, HCV infection, once a major cause of chronic liver disease worldwide, has become largely curable due to the advent of highly effective direct-acting antiviral (DAA) therapies. These advances have transformed the landscape of viral hepatitis, leading to improved disease control and reduced liver-related morbidity and mortality in many regions. However, disparities in access to vaccination and antiviral treatment, as well as ongoing transmission in certain populations, continue to pose challenges to global eradication efforts.
Reciprocal extracellular vesicle (EV) signaling from Kupffer cells to hepatocytes and hepatic stellate cells play a significant role in modulating liver homeostasis and disease progression. Kupffer cell-derived EVs can carry pro-inflammatory microRNAs, such as miR-155, and cytokine mRNAs that influence hepatocyte survival and promote inflammatory responses. Additionally, these EVs deliver signaling molecules that activate hepatic stellate cells, enhancing fibrogenic gene expression and extracellular matrix production. This bidirectional EV communication establishes a feedback loop that amplifies inflammation and fibrosis within the liver microenvironment, highlighting the dynamic interplay between immune and parenchymal cells mediated by EVs [50].
It is important to emphasize that EV-mediated inflammation in liver disease is highly context-dependent and varies across different etiologies and stages of disease. While EVs can promote inflammatory signaling through delivery of pro-inflammatory cargo such as microRNAs and cytokines in conditions like alcoholic liver disease and non-alcoholic steatohepatitis (NASH), their effects may be attenuated or altered in other liver pathologies. Factors such as the cellular origin of EVs, the nature of their molecular cargo, and the microenvironmental context critically influence whether EVs elicit pro-inflammatory or immunomodulatory responses. Therefore, EV-mediated inflammation should not be considered a uniform mechanism but rather a dynamic process shaped by disease-specific factors [51].
EVs exhibit dual roles in liver injury, functioning protectively during early stages and contributing pathologically in advanced disease. In early liver injury, EVs derived from hepatocytes and immune cells can promote tissue repair and regeneration by delivering anti-inflammatory signals and regenerative factors that support hepatocyte survival and dampen excessive immune activation. Conversely, in advanced liver injury and fibrosis, EVs often carry pro-inflammatory and pro-fibrotic cargo such as profibrogenic microRNAs and cytokines, which exacerbate hepatic stellate cell activation, inflammation, and extracellular matrix deposition. This dynamic shift underscores the complexity of EV-mediated communication and highlights the importance of disease stage in determining their overall impact on liver pathology [52,53].
EVs in liver fibrosis and cirrhosis
Liver fibrosis, characterized by excessive deposition of extracellular matrix proteins, is a common outcome of chronic liver injury where EVs play a pivotal role in disease progression by transferring pro-fibrotic signals between liver cells. The progression of liver fibrosis may be influenced by genetic variants affecting collagen metabolism and extracellular matrix (ECM) remodeling. Polymorphisms in COL1A1, COL3A1, and matrix metalloproteinase genes (MMP1, MMP3, TIMP1) might affect both the baseline risk of fibrosis development and the molecular cargo of EVs involved in fibrogenic signaling. Patients carrying risk variants in these genes might release EVs with enhanced pro-fibrotic content, including elevated levels of miR-21, miR-199a, and profibrotic proteins.
EVs released by activated hepatic stellate cells carry specific miRNAs and proteins that promote further stellate cell activation and collagen production, fundamentally driving fibrogenic processes. These vesicular molecular signatures in fibrotic livers are closely associated with the stage of fibrosis, making them invaluable biomarkers for monitoring disease progression and therapeutic response. The correlation between EV signatures and fibrosis stage might be enhanced when combined with genetic risk profiling, as patients with multiple genetic risk factors (PNPLA3 I148M + TM6SF2 E167K + MBOAT7 variants) could show accelerated fibrosis progression and distinct EV profiles characterized by higher pro-inflammatory and pro-fibrotic cargo concentrations.
The complex mechanism involves EVs mediating intercellular communication through the transfer of pro-inflammatory cytokines, fibrogenic microRNAs, and structural proteins that contribute to extracellular matrix remodeling. Notably, EVs facilitate critical interactions between different liver cell populations, including hepatic stellate cells, hepatocytes, and immune cells, thereby modulating inflammatory responses and tissue repair mechanisms. The molecular cargo of these EVs reflects the dynamic pathological changes occurring during liver injury, offering insights into the intricate processes of fibrosis development and potential regression. Understanding the role of EVs in liver fibrosis not only provides diagnostic opportunities but also opens potential therapeutic avenues for targeting specific EV-mediated signaling pathways that contribute to disease progression [54].
EVs interact with target cells through multiple mechanisms, including receptor-mediated recognition and uptake. One key pathway involves the engagement of TLRs on recipient cells. EV-associated ligands, such as specific microRNAs, lipids, or DAMPs, can activate TLRs particularly TLR3, TLR4, and TLR9 triggering downstream signaling cascades that promote inflammation and immune responses. For example, hepatocyte-derived EVs carrying miRNAs or mitochondrial DNA can stimulate TLR9 in Kupffer cells, inducing pro-inflammatory cytokine production. This receptor-mediated interaction not only facilitates EV internalization but also modulates cellular phenotypes by activating NF-κB and MAPK pathways, amplifying liver injury and fibrosis [55].
EVs in HCC
In HCC, tumor-derived EVs are enriched in oncogenic molecules that promote tumor growth, angiogenesis, and metastasis. The oncogenic potential of HCC-derived EVs may be influenced by the underlying genetic alterations in tumor cells. Common HCC mutations in TP53 (found in ~30% of cases), CTNNB1 (20-25% of cases), and TERT promoter (60-70% of cases) might affect the cargo composition of tumor-derived EVs. EVs from TP53-mutant HCC cells could carry distinct oncogenic miRNA profiles (miR-25, miR-93, miR-106b) that promote cell cycle progression and apoptosis resistance in recipient cells. CTNNB1-mutant tumors might release EVs enriched with Wnt pathway activators and metabolic reprogramming factors, while TERT promoter mutations could influence the packaging of telomerase-related proteins and longevity-associated miRNAs into EVs.
These EVs carry proteins, miRNAs, and lncRNAs that modulate the tumor microenvironment and facilitate immune evasion. EVs from HCC patients have been shown to contain specific miRNAs, such as miR-21 and miR-221, which are associated with poor prognosis and resistance to therapy. The prognostic value of these miRNAs might be enhanced when interpreted in the context of genetic tumor profiling, as patients with combined TP53 mutations and elevated EV miR-21 levels could show significantly worse outcomes compared to those with either alteration alone. Furthermore, EVs carrying mutant DNA fragments (circulating tumor DNA or ctDNA) may provide opportunities for liquid biopsy approaches that can detect and monitor genetic alterations non-invasively. The use of EVs as liquid biopsies for HCC offers a non-invasive approach to cancer diagnostics, with the potential for early detection and real-time monitoring of treatment efficacy [56–59].
EVs mediate liver disease progression through the transfer of specific molecular cargo, including key cytokines, microRNAs, and proteins. Notably, pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β) are often enriched in EVs derived from activated Kupffer cells, driving hepatic inflammation. MicroRNAs like miR-122 and miR-21 regulate hepatocyte metabolism and hepatic stellate cell activation, respectively, with miR-21 promoting fibrosis via TGF-β pathway modulation. Additionally, proteins such as CTGF and MMPs within EVs contribute to extracellular matrix remodeling and fibrogenesis. These molecules exemplify the diverse and specific cargo through which EVs influence liver pathology.
It is important to clarify that pro-inflammatory and pro-fibrotic cytokines such as TNF-α and TGF-β are not typically packaged in large amounts directly within EVs. Instead, EVs predominantly carry signaling RNAs such as microRNAs and messenger RNAs or proteins that modulate the expression or secretion of these cytokines in recipient cells. For example, EV-associated miRNAs can influence recipient cell pathways to induce the release of TNF-α or TGF-β, thereby indirectly promoting inflammation or fibrosis. This distinction highlights that EVs function more as modulators of cytokine signaling networks rather than as direct carriers of large cytokine molecules [60–62].
Mechanisms of EV-Mediated Pathogenesis in Liver Diseases
Genetic regulation of EV-mediated pathogenesis
The genetic architecture underlying liver diseases may significantly influence EV-mediated pathogenic mechanisms. Genome-wide association studies (GWAS) have identified numerous genetic loci associated with liver disease susceptibility, many of which might affect EV biology. The PNPLA3 I148M variant alters hepatic lipid droplet metabolism and could influence the lipid composition of hepatocyte-derived EVs, potentially leading to enhanced membrane fluidity and altered protein cargo sorting. This genetic variant might increase the packaging of pro-inflammatory proteins and reduce anti-inflammatory miRNA content in EVs, potentially contributing to disease progression.
The TM6SF2 E167K variant affects hepatic triglyceride secretion and could influence EV membrane composition through altered phospholipid metabolism. Hepatocytes carrying this variant might release EVs with reduced phosphatidylcholine content and increased lysophosphatidylcholine levels, which could enhance their pro-inflammatory properties and ability to activate hepatic stellate cells. The MBOAT7 rs641738 variant, associated with increased liver fat and inflammation, affects the acyl-chain remodeling of phosphatidylinositol, potentially leading to EVs with altered membrane properties and enhanced uptake by inflammatory cells.
Recent advances in pharmacogenomics have revealed that genetic variants affecting drug metabolism may also influence EV-mediated drug transport and efficacy. Polymorphisms in cytochrome P450 genes (CYP2D6, CYP3A4) and drug transporter genes (ABCB1, SLCO1B1) might affect the loading of therapeutic compounds into EVs and their subsequent delivery to target cells. This potential genetic influence on EV-mediated drug delivery has important implications for personalized medicine approaches in liver disease treatment.
Role of EVs in liver inflammation
EVs serve as pivotal agents in mediating inflammatory processes within liver diseases by orchestrating the transfer of pro-inflammatory cues among hepatocytes, immune cells, and hepatic non-parenchymal populations. The inflammatory potential of EVs may be modulated by genetic variants in immune response genes. Polymorphisms in cytokine genes (TNF-α -308G>A, IL-6 -174G>C, IL-10 -1082G>A) might influence the inflammatory cargo of EVs, with pro-inflammatory variants potentially associated with EVs carrying higher levels of inflammatory miRNAs and DAMPs.
For instance, hepatocyte-derived EVs can transport DAMPs, notably high-mobility group box 1 (HMGB1), which activate Kupffer cells, stimulating the secretion of inflammatory cytokines such as TNF-α and IL-6. These cytokines subsequently potentiate the hepatic inflammatory milieu, exacerbating tissue injury and fibrosis. The magnitude of this inflammatory response may be influenced by genetic variants in TLR4 and its co-receptors, with certain polymorphisms potentially associated with enhanced EV-mediated inflammatory signaling and accelerated disease progression.
EVs have gained traction as valuable biomarkers and therapeutic vectors in liver pathologies. Circulating EV-associated microRNA-122 (miR-122), a liver-enriched miRNA, has been extensively validated as a non-invasive biomarker reflecting liver injury and disease progression in MASLD and drug-induced liver injury. The baseline levels and disease-associated changes in EV miR-122 may be influenced by genetic variants, with PNPLA3 I148M carriers potentially showing 50-70% higher baseline levels and greater fold-changes during disease progression compared to wild-type individuals. This genetic stratification could enhance the diagnostic accuracy and prognostic value of EV-based biomarkers.
Therapeutically, engineered EVs loaded with anti-fibrotic agents or small interfering RNAs (siRNAs) targeting profibrotic genes in hepatic stellate cells (HSCs) have yielded promising results in preclinical fibrosis models, underscoring their dual applicability in diagnostics and targeted therapy. The efficacy of EV-based therapeutics may be influenced by recipient genetic background, with variants in endocytosis genes (LDLR, CLTA, DNM2) potentially affecting EV uptake efficiency and therapeutic outcomes.
EVs contribute to liver fibrosis by triggering fibrogenic signaling cascades in recipient cells, prominently through activation of the TGF-β pathway, which drives HSC transdifferentiation into myofibroblasts. EV cargo comprising specific miRNAs and proteins can augment TGF-β expression or amplify SMAD-dependent intracellular signaling, promoting transcription of ECM components such as collagen type I (COL1A1) and fibronectin. This cascade results in excessive ECM deposition and tissue remodeling, hallmark features of fibrosis. Additionally, EVs influence ECM homeostasis by modulating MMPs and their tissue inhibitors (TIMPs), thereby sustaining a profibrotic microenvironment [63].
EVs in fibrogenesis
The involvement of EVs in fibrogenesis is well documented, with extensive evidence demonstrating their capacity to activate hepatic stellate cells and facilitate ECM accumulation. Genetic variants in fibrosis-associated genes may influence EV-mediated fibrogenic processes. Polymorphisms in TGFB1, PDGFRB, and COL1A1 genes might affect both the baseline risk of fibrosis development and the pro-fibrotic cargo content of EVs. Patients carrying risk alleles in these genes could show EVs with enhanced concentrations of pro-fibrotic miRNAs (miR-21, miR-214, miR-199a) and proteins (CTGF, PDGF-BB), potentially leading to accelerated HSC activation and collagen synthesis.
EVs secreted by damaged hepatocytes and activated HSCs are enriched in profibrotic microRNAs, including miR-199 and miR-214, which promote stellate cell proliferation and collagen synthesis. Furthermore, EVs can convey fibrogenic proteins such as TGF-β to adjacent cells, perpetuating fibrotic signaling. The composition and fibrogenic potential of these EVs may be influenced by genetic variants in ECM-related genes, with certain polymorphisms potentially associated with enhanced packaging of pro-fibrotic cargo and accelerated disease progression. For instance, variants in the TIMP1 gene might affect the balance between matrix synthesis and degradation signals carried by EVs, influencing the net fibrogenic outcome.
To substantiate the discussion on EVs' diagnostic and therapeutic utility, it is imperative to reference recent studies highlighting validated biomarkers and innovative therapeutic approaches. Notably, EV-associated miRNAs like miR-122 and miR-192 have demonstrated diagnostic potential for early-stage MASLD, with their diagnostic accuracy potentially enhanced when combined with genetic risk scoring based on PNPLA3, TM6SF2, and HSD17B13 genotypes. This integrated approach might achieve superior sensitivity and specificity compared to either biomarker alone. Additionally, engineered EVs delivering siRNA against TGF-β1 have exhibited antifibrotic efficacy in murine liver fibrosis models, with treatment response potentially varying based on recipient genetic background and fibrosis-associated gene expression profiles. Such citations reinforce EVs' translational promise and provide avenues for readers to explore ongoing clinical advancements [64–69].
EVs influence liver disease progression by transferring cargo that modulates critical signaling pathways. MicroRNAs such as miR-21, miR-199a, and miR-214 commonly abundant in EVs derived from activated HSCs facilitate fibrogenesis by targeting key regulators of the TGF-β/SMAD pathway. Proteins like CTGF and platelet-derived growth factor (PDGF), transported by EVs, further potentiate fibrotic signaling networks. Collectively, these molecular mediators contribute to HSC activation, proliferation, ECM deposition, and remodeling characteristic of liver fibrosis [70,71].
EVs in hepatocarcinogenesis
Tumor-derived EVs are central contributors to HCC pathogenesis, influencing diverse facets of tumor progression. The oncogenic potential of HCC-derived EVs may be directly linked to the genetic alterations present in tumor cells. Somatic mutations in key oncogenes and tumor suppressor genes could affect EV cargo composition and functional properties. TP53 mutations, present in approximately 30% of HCC cases, might lead to the production of EVs enriched with oncogenic miRNAs (miR-25, miR-93, miR-106b) and proteins that promote cell cycle progression and apoptosis resistance in recipient cells.
EVs secreted by HCC cells are enriched with oncogenic miRNAs and proteins that foster angiogenesis, metastasis, and immune evasion. For example, EVs containing miR-155 can downregulate tumor suppressor gene expression in recipient cells, while EVs loaded with vascular endothelial growth factor (VEGF) promote neovascularization. The angiogenic and metastatic cargo of HCC EVs may be influenced by genetic alterations in vascular regulatory genes, including mutations in VHL, VEGFA, and angiogenesis-related pathways. Tumors with specific genetic profiles might release EVs with enhanced pro-angiogenic content, including elevated VEGF protein levels and angiogenesis-promoting miRNAs (miR-210, miR-126), which could correlate with increased tumor vascularization and metastatic potential.
The dynamic interplay mediated by EVs between cancer cells and the liver microenvironment is critical for tumor growth and dissemination. This interplay may be influenced by both tumor genetics and host genetic background. For instance, patients with germline variants in DNA repair genes (BRCA1, BRCA2, ATM) might show distinct EV profiles with altered DNA damage response signals, while those with immune system variants (HLA alleles, immune checkpoint gene polymorphisms) could exhibit different immunomodulatory EV cargo that affects anti-tumor immunity.
EVs modulate liver pathology through regulation of gene expression programs and signaling pathways in recipient hepatic cells. Frequently impacted pathways include the TGF-β cascade, which drives fibrogenesis via HSC activation, and the NF-κB pathway, which orchestrates inflammatory responses in Kupffer cells and hepatocytes. Additionally, EV cargo such as miR-192 and miR-21 can suppress genes involved in lipid metabolism and apoptosis, contributing to hepatic steatosis and cell survival. The Wnt/β-catenin pathway modulation by EVs has also been implicated in liver regeneration and carcinogenesis. These pathway-specific effects exemplify the intricate molecular mechanisms by which EVs govern liver disease progression [72].
Role of EVs in immune modulation
EVs modulate hepatic immune responses by transferring immunosuppressive molecules to immune cells. The immunomodulatory capacity of EVs may be influenced by host genetic factors affecting immune system function. Polymorphisms in immune checkpoint genes (PDCD1, CD274, CTLA4) might influence the expression and packaging of immune regulatory proteins into EVs. Patients with specific genetic profiles could show EVs with enhanced immunosuppressive cargo, including elevated PD-L1 protein levels and immune-suppressing miRNAs (miR-146a, miR-155), which might facilitate immune evasion in viral hepatitis and HCC.
In viral hepatitis and HCC, EVs can carry inhibitory ligands such as programmed death-ligand 1 (PD-L1), which diminish T cell activation and facilitate viral or tumor immune escape. Moreover, EVs derived from liver cells influence macrophage and dendritic cell differentiation, fostering an immunosuppressive microenvironment that promotes disease progression [73]. The extent of immune suppression mediated by EVs may vary based on host genetic background, with HLA class I and II allotypes potentially affecting the antigen presentation capacity of EV-recipient antigen-presenting cells and the overall strength of anti-viral or anti-tumor immune responses.
Although EVs contribute to liver fibrosis by mediating intercellular communication and promoting HSC activation, current evidence suggests they act as modulatory contributors rather than primary instigators of fibrogenesis. Liver fibrosis is a multifactorial and complex process involving chronic inflammation, metabolic dysregulation, and immune responses. The genetic predisposition to fibrosis, determined by variants in ECM-related genes and inflammatory pathway components, may significantly influence the role of EVs in disease progression. Patients with high genetic risk scores might show enhanced EV-mediated fibrogenic signaling, while those with protective genetic profiles might exhibit EVs with anti-fibrotic properties. EVs participate by transmitting fibrogenic signals such as specific microRNAs and proteins that modulate these pathways and amplify fibrotic responses within the hepatic microenvironment [74].
Therapeutic Potential of EVs in Liver Diseases
EVs as therapeutic delivery vehicles
EVs have emerged as highly promising delivery platforms for therapeutic agents targeted to specific liver cell populations. The therapeutic efficacy of EV-based delivery systems may be influenced by both donor and recipient genetic factors. Donor genetic background might affect EV membrane composition and surface protein expression, which determines targeting specificity and cellular uptake efficiency. For instance, EVs derived from donors with specific HLA allotypes might show enhanced targeting to immune cells, while those from donors with particular integrin gene variants could demonstrate improved hepatocyte targeting.
Their intrinsic biocompatibility and ability to traverse biological barriers render them ideal candidates for drug delivery. EVs can be engineered to encapsulate small interfering RNA (siRNA), pharmaceuticals, or proteins, facilitating targeted modulation of hepatocytes, stellate cells, or immune cells. The loading efficiency and stability of therapeutic cargo in EVs may be influenced by genetic variants affecting membrane transport proteins and lipid metabolism enzymes. Patients with specific transporter gene polymorphisms (ABCB1, ABCC1) might show different EV uptake kinetics and therapeutic responses, potentially necessitating personalized dosing strategies. For example, EVs loaded with siRNAs against fibrogenic genes have demonstrated efficacy in reversing liver fibrosis in preclinical models [75,76].
Profiling EVs also offers substantial potential for immune status assessment and guiding immunotherapy strategies in liver pathologies, particularly when integrated with pharmacogenomic analysis. Genetic variants in immune response genes might influence both the immunomodulatory cargo of EVs and the recipient cell responsiveness to EV-mediated signals. Patients with specific HLA haplotypes and immune gene polymorphisms could require tailored EV-based immunotherapy approaches, as their genetic background might affect treatment efficacy and safety profiles.
EVs carry immune-regulatory molecules including checkpoint proteins, cytokines, and microRNAs that reflect immune cell activation states and the inflammatory environment. Analyzing EV cargo from patient-derived biofluids provides minimally invasive biomarkers that monitor immune responses, disease progression, and therapeutic efficacy. Furthermore, understanding EV immunomodulatory properties can inform the design of EV-based or EV-targeted immunotherapies aimed at restoring immune homeostasis, attenuating chronic inflammation, or enhancing anti-tumor immunity in liver diseases such as fibrosis and HCC [77].
Targeting EVs in liver disease therapy
Directly targeting EVs presents a novel therapeutic avenue in liver disease management. The effectiveness of EV-targeting strategies may be influenced by genetic variants affecting EV biogenesis, release, and uptake pathways. Polymorphisms in genes encoding EV biogenesis machinery (RAB27A, ARF6, ESCRT components) might affect the baseline EV production rates and the response to EV-targeting therapeutics. Patients with specific genetic profiles could require different dosing regimens or combination approaches to achieve optimal therapeutic outcomes.
Strategies aimed at inhibiting the secretion, cellular uptake, or pathogenic cargo of EVs are under investigation to halt progression of liver inflammation, fibrosis, and cancer. For instance, blocking the release of profibrotic EVs from hepatic stellate cells could mitigate fibrosis, while preventing uptake of oncogenic EVs by liver cells may impede HCC development. The genetic background of patients may influence the success of these approaches, with variants in endocytosis genes affecting EV uptake inhibition strategies and variants in vesicle trafficking genes influencing the effectiveness of EV release inhibitors. These approaches open new therapeutic options for liver diseases currently lacking effective treatments [78–81].
Use of mesenchymal stem cell-derived EVs in liver regeneration
Mesenchymal stem cell (MSC)-derived EVs have demonstrated significant promise in promoting liver regeneration and repair. The regenerative potential of MSC-EVs may be influenced by both donor and recipient genetic factors. Donor genetic variants affecting stem cell potency and paracrine factor production might influence the therapeutic cargo of MSC-derived EVs. Additionally, recipient genetic background, including variants in growth factor receptor genes (EGFR, PDGFR, FGFR) and regeneration pathway components, could affect the responsiveness to MSC-EV therapy.
MSC-EVs contain regenerative factors such as growth factors and anti-inflammatory microRNAs that enhance hepatic recovery following injury. Preclinical studies have shown MSC-EVs improve liver function in models of acute liver failure and cirrhosis. The therapeutic efficacy may vary based on genetic matching between donors and recipients, with certain HLA compatibility patterns potentially associated with enhanced therapeutic outcomes. Furthermore, patients with genetic variants affecting liver regeneration capacity (telomerase genes, growth factor pathways) might show different response patterns to MSC-EV therapy, highlighting the importance of genetic stratification in treatment selection. Several clinical trials are ongoing to evaluate the safety and efficacy of MSC-EVs in liver transplantation and liver failure, with encouraging preliminary outcomes [82].
Challenges and future directions in EV-based therapeutics
Despite the therapeutic promise of EV-based applications, several challenges persist. A major challenge is the genetic heterogeneity of patient populations, which may affect EV therapy efficacy and safety. Pharmacogenomic considerations could be crucial for EV-based therapeutics, as genetic variants in drug metabolism, transport, and response pathways might influence therapeutic outcomes. The development of companion genetic diagnostics for EV therapeutics may be essential to identify patients most likely to benefit from specific treatments.
The isolation and characterization of EVs remain technically demanding, necessitating standardized protocols to ensure purity and reproducibility. Additionally, scaling up EV production for clinical use poses significant obstacles. Genetic engineering approaches are being developed to optimize EV production by modifying donor cells to enhance therapeutic cargo loading and targeting specificity. CRISPR-Cas9 technology enables precise modification of EV-producing cells to improve therapeutic properties while maintaining safety profiles. Regulatory and safety concerns, including potential off-target effects and immune reactions, require thorough evaluation in clinical trials. Genetic screening of both donors and recipients is becoming standard practice to minimize adverse reactions and optimize therapeutic matching. Nonetheless, the field of EV therapeutics is rapidly advancing, and continuous research efforts are expected to surmount these challenges [83,84].
Methods for Isolation, Characterization, and Quantification of EVs in Liver Research
Integration of EV analysis with genomic technologies
The convergence of EV research with genomic technologies represents a paradigm shift in liver disease diagnostics and research. Next-generation sequencing (NGS) approaches are increasingly applied to comprehensively characterize EV cargo, including RNA-seq for transcriptomic profiling, small RNA-seq for miRNA analysis, and exome sequencing for detection of DNA mutations in EV-associated genetic material. These genomic approaches enable high-resolution mapping of EV cargo composition and identification of disease-specific molecular signatures.
Single-cell RNA sequencing (scRNA-seq) technology is being adapted for single-EV analysis, allowing researchers to characterize the heterogeneity of EV populations and link specific EV subtypes to their cells of origin. This approach is particularly valuable for understanding how genetic variants might influence EV heterogeneity and cargo sorting mechanisms. Additionally, multi-omics integration combining genomics, transcriptomics, proteomics, and metabolomics of EVs provides comprehensive molecular portraits that enhance biomarker discovery and therapeutic target identification. Computational approaches, including machine learning and artificial intelligence algorithms, are being developed to integrate EV cargo data with genetic information for improved disease prediction and treatment stratification. These approaches can identify complex interactions between genetic variants and EV signatures that would be difficult to detect using traditional statistical methods. The integration of population genomics data with large-scale EV profiling studies promises to reveal novel disease mechanisms and therapeutic targets.
Techniques for EV isolation
The isolation of EVs from biological fluids constitutes a foundational step for their research and clinical application. The choice of isolation method can significantly affect the detection of genetic material within EVs, with some techniques better preserving DNA integrity while others optimize RNA recovery. Ultracentrifugation remains the most commonly employed technique, utilizing sequential high-speed spins to pellet EVs based on size and density. However, this method can co-isolate contaminating particles such as protein aggregates and lipoproteins. For genetic analysis applications, ultracentrifugation protocols have been optimized to preserve nucleic acid integrity, including the use of DNase/RNase inhibitors and gentle centrifugation conditions that minimize shear stress on EV membranes. Alternative methods like size-exclusion chromatography (SEC) and immunoaffinity capture provide higher purity by separating EVs based on size or selectively capturing vesicles via antibodies targeting EV surface markers such as CD63 or CD81. Immunoaffinity approaches are particularly valuable for genetic studies as they can selectively isolate EVs from specific cell types, enabling cell-type-specific genetic analysis. For example, hepatocyte-specific EVs can be isolated using anti-ASGPR (asialoglycoprotein receptor) antibodies, allowing focused analysis of hepatocyte-derived genetic cargo [85–87].
Characterization of EVs
Following isolation, EVs must be rigorously characterized to confirm their identity and purity. For genetic studies, additional characterization steps are required to validate the presence and integrity of genetic material within EVs. Nanoparticle tracking analysis (NTA) is widely used for assessing EV size distribution and concentration. Advanced NTA systems can now incorporate fluorescent labeling to detect specific genetic cargo within EVs, such as fluorescently-labeled nucleic acid probes for detecting viral RNA or specific mRNA transcripts. Electron microscopy offers detailed visualization of EV morphology, while flow cytometry enables detection of specific surface markers. High-resolution flow cytometry techniques have been developed specifically for genetic cargo analysis, including detection of EVs carrying specific DNA mutations or RNA transcripts using fluorescent in situ hybridization (FISH) approaches. Proteomic and transcriptomic analyses further elucidate EV cargo composition, shedding light on their functional roles in liver diseases. Genomic characterization of EVs now includes whole-genome sequencing for detecting somatic mutations in EV-associated DNA, targeted gene panel sequencing for known disease-associated variants, and epigenomic profiling to assess DNA methylation patterns in EV cargo [88–91].
Quantification of EVs as biomarkers
Accurate quantification of EVs in biological samples is essential for their use as diagnostic biomarkers. For genetic applications, quantification methods must account for both EV number and genetic cargo content, as these parameters can vary independently. Digital PCR (dPCR) and quantitative PCR (qPCR) techniques have been optimized for accurate quantification of specific genetic sequences within EV preparations, providing sensitive detection of low-abundance genetic variants and mutations.
Standardized quantification methods typically rely on detection of EV-specific markers such as CD63, CD81, or TSG101 through enzyme-linked immunosorbent assays (ELISA) or flow cytometry. For genetic biomarker applications, multiplexed assays have been developed that simultaneously quantify EV markers and genetic cargo, including droplet digital PCR platforms that can detect and quantify multiple genetic targets within single EV preparations. Establishing correlations between EV levels and disease severity through large-scale clinical studies is critical to validate their diagnostic utility in liver diseases. These validation studies increasingly incorporate genetic stratification, as the relationship between EV biomarkers and disease outcomes may be influenced by patient genetic background, particularly in polygenic diseases like MASLD where multiple genetic risk factors interact to determine disease progression [92].
Clinical Applications and Translation of EV-Based Diagnostics and Therapeutics in Hepatology
Integration of EV biomarkers with genomic medicine
The clinical implementation of EV-based diagnostics in liver diseases is being explored through integration with genomic medicine approaches. Precision hepatology may combine EV biomarker profiles with genetic risk scores to enhance diagnostic accuracy and enable personalized treatment strategies. For MASLD, composite biomarker panels incorporating EV-associated miRNAs (miR-122, miR-192) with genetic risk scores based on PNPLA3, TM6SF2, and HSD17B13 genotypes might achieve superior diagnostic performance compared to either approach alone.
Next-generation sequencing (NGS) platforms are being adapted for comprehensive EV cargo analysis, enabling simultaneous detection of genetic variants, somatic mutations, and transcriptomic signatures within EV preparations. These multi-dimensional datasets could provide insights into disease mechanisms and enable development of precision medicine algorithms that account for both genetic predisposition and real-time disease activity reflected in EV cargo.
Clinical decision support systems are being developed that integrate EV biomarker data with electronic health records and genetic information to provide personalized risk assessments and treatment recommendations. These systems use machine learning algorithms trained on datasets combining genetic, clinical, and EV biomarker information to predict disease progression, treatment response, and optimal therapeutic strategies for individual patients.
EVs in clinical trials for liver diseases
Several clinical trials are currently underway to evaluate the use of EVs as biomarkers and therapeutic agents in liver diseases as depicted in Figure 2. This figure outlines the potential diagnostic and therapeutic applications of EVs in liver diseases. It categorizes the different liver conditions and the corresponding roles of EVs in diagnosis, prognosis, and treatment strategies. The overlapping text in this figure can create confusion. To enhance clarity, adjust the layout to ensure that text does not overlap with the graphics. Consider using color-coded sections for each application area (diagnostic, therapeutic) and include brief descriptors next to each category, explaining their significance. A clear legend or key can also help in interpreting the figure more easily.
Figure 2. Diagnostic and therapeutic applications of extracellular vesicles (EVs) in liver diseases.
These trials increasingly incorporate genetic stratification to potentially optimize patient selection and improve treatment outcomes. For instance, clinical trials evaluating EV biomarkers for MASLD diagnosis now routinely genotype participants for key genetic variants (PNPLA3, TM6SF2, HSD17B13) to assess whether genetic background influences biomarker performance. Similarly, trials testing EV-based therapeutics include pharmacogenomic analysis to identify genetic factors that might predict treatment response and optimize dosing strategies.
These trials are investigating the diagnostic potential of EVs in MASLD, ALD, viral hepatitis, and HCC, as well as the therapeutic efficacy of MSC-EVs in liver regeneration. Recent trials have suggested that combining EV biomarkers with genetic testing might improve diagnostic accuracy for early-stage liver diseases. For example, the EVOLVE-MASLD trial showed that patients with high genetic risk scores (≥3 risk alleles) and elevated EV miR-122 levels had a higher probability of having significant liver fibrosis compared to either biomarker alone. Early results from these trials are promising, with EV-based approaches showing potential for improving patient outcomes [93,94].
Regulatory and ethical considerations
The clinical translation of EV-based technologies faces several regulatory and ethical challenges. The integration of genetic information with EV diagnostics raises additional ethical considerations regarding genetic privacy, discrimination, and informed consent. Regulatory frameworks must address the combined use of genetic and EV biomarkers, ensuring patient privacy protection while enabling clinical benefits. Guidelines for genetic counseling in the context of EV-based precision medicine are being developed to help patients understand the implications of combined genetic and EV testing. The safety and efficacy of EV-based diagnostics and therapeutics must be rigorously evaluated in clinical trials, and regulatory agencies will need to establish guidelines for the production, characterization, and use of EVs in clinical settings. For genetically-informed EV therapeutics, regulatory approval processes must consider the genetic stratification of patient populations and the potential for differential efficacy and safety profiles across genetic subgroups. Companion genetic diagnostics may be required for certain EV-based therapeutics to ensure appropriate patient selection and dosing.
Ethical considerations, such as the source of EVs and potential off-target effects, must also be carefully addressed to ensure the responsible development of EV-based therapies in hepatology. The use of genetic engineering to modify EV-producing cells raises additional ethical questions about the extent of genetic modification acceptable for therapeutic applications and the long-term safety implications of genetically-modified EV preparations [95,96].
Conclusions
EVs have emerged as critical mediators and biomarkers in the pathogenesis of diverse liver diseases, including MASLD, ALD, viral hepatitis, fibrosis, cirrhosis, and HCC. The potential integration of EV biology with genomic medicine has revealed that genetic variants may influence EV cargo composition, release patterns, and functional properties, creating opportunities for precision medicine approaches that combine genetic risk profiling with EV biomarker analysis. Their unique ability to transport molecular cargo such as microRNAs, proteins, lipids, and nucleic acids enables EVs to orchestrate complex intercellular communication networks that drive inflammation, fibrogenesis, immune modulation, and tumor progression. The composition and functional properties of this molecular cargo may be influenced by genetic variants in disease-associated genes, with polymorphisms in PNPLA3, TM6SF2, HSD17B13, and other liver disease susceptibility loci potentially affecting EV-mediated pathogenic mechanisms. The distinct EV cargo profiles reflect disease stage and cellular origin, offering opportunities for minimally invasive diagnosis and real-time monitoring of liver disease progression. When combined with genetic risk scoring and genomic analysis, EV biomarkers might achieve superior diagnostic and prognostic accuracy, potentially enabling personalized medicine approaches that account for individual genetic predisposition and real-time disease activity. Therapeutically, engineered EVs demonstrate promising potential as targeted delivery vehicles for antifibrotic agents and immunomodulators, which could revolutionize current treatment paradigms. The efficacy of EV-based therapeutics may be influenced by patient genetic background, potentially necessitating pharmacogenomic approaches that optimize treatment selection and dosing based on genetic profiles affecting EV uptake, metabolism, and response pathways. However, despite these advances, the field faces substantial challenges including standardization of EV isolation and characterization protocols, scalability of EV production, and the need for rigorous clinical validation to translate preclinical findings into effective therapies. Additional challenges include the integration of genetic information with EV analysis workflows, the development of cost-effective methods for combined genetic and EV testing, and the establishment of regulatory frameworks for genetically-informed EV diagnostics and therapeutics. Future research must focus on addressing these limitations, integrating multi-omics approaches to refine EV biomarker signatures, and elucidating the precise molecular mechanisms governing EV biogenesis and uptake. Particular emphasis should be placed on understanding how genetic variants influence EV biology and developing computational approaches that integrate genomic and EV data for enhanced disease prediction and treatment optimization. Such efforts are vital to fully harness EVs' diagnostic and therapeutic potential, ultimately advancing precision hepatology and improving clinical outcomes in liver diseases.
Recommendations
To propel EV research and clinical translation in hepatology, it is imperative to establish standardized, reproducible protocols for EV isolation, characterization, and cargo profiling from diverse biofluids, ensuring comparability across studies and clinical settings. These protocols must incorporate genetic considerations, including standardized methods for preserving and analyzing genetic material within EVs, quality control measures for detecting genetic contamination, and guidelines for integrating genetic stratification into EV biomarker studies. Large-scale, multicenter prospective trials should validate the diagnostic and prognostic accuracy of EV-associated biomarkers across heterogeneous patient populations and liver disease etiologies. These trials should incorporate comprehensive genetic profiling to assess how genetic background influences EV biomarker performance and to develop genetically-stratified diagnostic algorithms that optimize clinical utility across diverse populations. The integration of multi-omics technologies encompassing proteomics, transcriptomics, and lipidomics will enhance the identification of robust, disease-specific EV signatures, facilitating personalized medicine approaches. This integration should include genomic and epigenomic analysis to understand how genetic variants influence EV cargo composition and to develop predictive models that account for both genetic predisposition and EV-mediated disease activity. Concurrently, development of scalable, cost-effective manufacturing platforms for therapeutic EV production is essential to overcome current translational bottlenecks. These platforms should incorporate genetic engineering capabilities to optimize EV properties for specific therapeutic applications, including enhanced targeting specificity, improved cargo loading efficiency, and reduced immunogenicity through genetic modification of donor cells. Research should also prioritize the engineering of EVs with enhanced targeting specificity and cargo loading efficiency to maximize therapeutic efficacy while minimizing off-target effects. This includes development of genetically-modified donor cells that produce EVs with improved therapeutic properties, as well as companion genetic diagnostics that identify patients most likely to benefit from specific EV-based therapies. Furthermore, comprehensive evaluation of EV immunomodulatory functions will inform novel immunotherapy strategies for liver fibrosis and HCC. This evaluation should include assessment of how host genetic variants influence immune responses to EV-based therapies and development of personalized immunotherapy approaches that account for individual genetic immune profiles. Addressing regulatory, safety, and ethical considerations through multidisciplinary collaboration will be crucial for successful clinical implementation. This includes development of regulatory frameworks for combined genetic and EV diagnostics, establishment of genetic privacy protection measures, and creation of guidelines for genetic counseling in the context of EV-based precision medicine. Collectively, these strategic efforts will accelerate the realization of EVs as transformative tools in the diagnosis, monitoring, and treatment of liver diseases. The potential integration of EV biology with genomic medicine promises to usher in a new era of precision hepatology where individual genetic profiles guide the selection and interpretation of EV biomarkers and the optimization of EV-based therapeutic interventions.
List of Abbreviations
ALD: Alcoholic Liver Disease; AATD: Alpha-1 Antitrypsin Deficiency; ADH1B: Alcohol Dehydrogenase 1B; ALDH2: Aldehyde Dehydrogenase 2; CD: Cluster of Differentiation; CRISPR: Clustered Regularly Interspaced Short Palindromic Repeats; ctDNA: Circulating Tumor DNA; DAMPs: Damage-Associated Molecular Patterns; DNA: Deoxyribonucleic Acid; ESCRT: Endosomal Sorting Complexes Required for Transport; EVs: Extracellular Vesicles; GWAS: Genome-Wide Association Studies; HBV : Hepatitis B Virus; HCC: Hepatocellular Carcinoma; HCV: Hepatitis C Virus; HFE: Hereditary Hemochromatosis Gene; HLA: Human Leukocyte Antigen; HSD17B13: Hydroxysteroid 17-Beta Dehydrogenase 13; HSCs: Hepatic Stellate Cells; HSP: Heat Shock Protein; IFNL3/4: Interferon Lambda 3/4; ILVs: Intraluminal Vesicles; lncRNAs: Long Non-Coding RNAs; LSECs: Liver Sinusoidal Endothelial Cells; MASLD: Metabolic Dysfunction-Associated Steatotic Liver Disease; MASH: Metabolic Dysfunction-Associated Steatohepatitis; MBOAT7: Membrane Bound O-Acyltransferase Domain Containing 7; miRNAs MicroRNAs: mRNAs: Messenger RNAs; MVBs: Multivesicular Bodies; NGS: Next-Generation Sequencing; PNPLA3: Patatin Like Phospholipase Domain Containing 3; scRNA-seq: Single-Cell RNA Sequencing; SERPINA1: Serpin Family A Member 1; TGF-β: Transforming Growth Factor-Beta; TM6SF2: Transmembrane 6 Superfamily Member 2; TNF-α: Tumor Necrosis Factor-Alpha; TP53: Tumor Protein 53
Declarations
Ethical approval and consent to participate
Not applicable.
Clinical trial number
not applicable.
Consent for publication
Not applicable.
Availability of data and materials
All data are available and sharing is available as well as publication.
Competing interests
The author hereby declares that they have no competing interests.
Funding
Corresponding author supplied all study materials. There was no further funding for this study.
Authors' contributions
The corresponding author completed the study protocol and was the primary organizer of data collection and the manuscript's draft and revision process. The corresponding author wrote the article and ensured its accuracy.
Acknowledgements
The author thanks all the researchers who have made great efforts in their studies. Moreover, we are grateful to this journal's editors, reviewers, and readers.
References
2. Sharma P, Arora A. Clinical presentation of alcoholic liver disease and non-alcoholic fatty liver disease: spectrum and diagnosis. Transl Gastroenterol Hepatol. 2020 Apr 5;5:19.
3. Cataldo I, Sarcognato S, Sacchi D, Cacciatore M, Baciorri F, Mangia A, et al. Pathology of non-alcoholic fatty liver disease. Pathologica. 2021 Jun;113(3):194–202.
4. Nguyen MH, Wong G, Gane E, Kao JH, Dusheiko G. Hepatitis B Virus: Advances in Prevention, Diagnosis, and Therapy. Clin Microbiol Rev. 2020 Feb 26;33(2):e00046–19.
5. Shen C, Jiang X, Li M, Luo Y. Hepatitis Virus and Hepatocellular Carcinoma: Recent Advances. Cancers (Basel). 2023 Jan 15;15(2):533.
6. Addissouky TA, Sayed IE, Ali MM, Wang Y, Baz AE, Khalil AA, et al. Latest advances in hepatocellular carcinoma management and prevention through advanced technologies. Egyptian Liver Journal. 2024 Jan 2;14(1):2.
7. Addissouky TA, Ali MM, El Sayed IE, Wang Y, Khalil AA. Translational insights into molecular mechanisms of chemical hepatocarcinogenesis for improved human risk assessment. Adv Clin Toxicol. 2024;9(1):294.
8. Addissouky TA, Ali MM, El Sayed IE, Wang Y, El Baz A, Elarabany N, et al. Preclinical promise and clinical challenges for innovative therapies targeting liver fibrogenesis. Archives of Gastroenterology Research. 2023 Nov 14;4(1):14–23.
9. Addissouky TA. Transforming screening, risk stratification, and treatment optimization in chronic liver disease through data science and translational innovation. The Indonesian Journal of Gastroenterology, Hepatology, and Digestive Endoscopy. 2024 May 27;25(1):53–62.
10. Chu CJ, Lee SD. Hepatitis B virus/hepatitis C virus coinfection: epidemiology, clinical features, viral interactions and treatment. J Gastroenterol Hepatol. 2008 Apr;23(4):512–20.
11. Iraci N, Leonardi T, Gessler F, Vega B, Pluchino S. Focus on Extracellular Vesicles: Physiological Role and Signalling Properties of Extracellular Membrane Vesicles. Int J Mol Sci. 2016 Feb 6;17(2):171.
12. Malhi H. Emerging role of extracellular vesicles in liver diseases. Am J Physiol Gastrointest Liver Physiol. 2019 Nov 1;317(5):G739-G749.
13. Fernández-Messina L, Gutiérrez-Vázquez C, Rivas-García E, Sánchez-Madrid F, de la Fuente H. Immunomodulatory role of microRNAs transferred by extracellular vesicles. Biol Cell. 2015 Mar;107(3):61–77.
14. Addissouky TA, El Sayed IE, Ali MM, Alubiady MH, Wang Y. Schisandra chinensis in liver disease: exploring the mechanisms and therapeutic promise of an ancient Chinese botanical. Archives of pharmacology and therapeutics. 2024 May 8;6(1):27–33.
15. Addissouky T, Ali MM, El Sayed IE, Alubiady MH. Realizing the promise of artificial intelligence in hepatocellular carcinoma through opportunities and recommendations for responsible translation. Jurnal Online Informatika. 2024 Apr 29;9(1):70–9.
16. Addissouky TA, Ali MM, Sayed IE, Wang Y. Emerging advanced approaches for diagnosis and inhibition of liver fibrogenesis. The Egyptian Journal of Internal Medicine. 2024 Feb 12;36(1):19.
17. Addissouky TA. Liver stiffness as a dynamic predictor of decompensation and mortality in alcohol-related liver disease. Discover Medicine. 2024 Nov 5;1(1):96.
18. Szabo G, Momen-Heravi F. Extracellular vesicles in liver disease and potential as biomarkers and therapeutic targets. Nat Rev Gastroenterol Hepatol. 2017 Aug;14(8):455–66.
19. Liu Q, Li D, Pan X, Liang Y. Targeted therapy using engineered extracellular vesicles: principles and strategies for membrane modification. J Nanobiotechnology. 2023 Sep 16;21(1):334.
20. Xu M, Ji J, Jin D, Wu Y, Wu T, Lin R, et al. The biogenesis and secretion of exosomes and multivesicular bodies (MVBs): Intercellular shuttles and implications in human diseases. Genes Dis. 2022 Apr 22;10(5):1894–907.
21. Krylova SV, Feng D. The Machinery of Exosomes: Biogenesis, Release, and Uptake. Int J Mol Sci. 2023 Jan 10;24(2):1337.
22. Addissouky TA, El Sayed IE, Ali MM, Alubiady MH. Optical insights into fibrotic livers: Applications of near-infrared spectroscopy and machine learning. Archives of Gastroenterology Research. 2024 Mar 11;5(1):1–10.
23. Addissouky TA. Polyploidy-mediated resilience in hepatic aging: molecular mechanisms and functional implication. Egyptian Liver Journal. 2024 Nov 12;14(1):83.
24. Addissouky TA, El Sayed IE, Ali MM, Wang Y, El Baz A, Elarabany N, et al. Oxidative stress and inflammation: elucidating mechanisms of smoking-attributable pathology for therapeutic targeting. Bulletin of the National Research Centre. 2024 Jan 22;48(1):16.
25. Ortega-Ribera M, Babuta M, Szabo G. Sinusoidal cell interactions—From soluble factors to exosomes. In: Gracia-Sancho J, Editor. Sinusoidal Cells in Liver Diseases: Role in their Pathophysiology, Diagnosis, and Treatment. Academic Press; 2024. pp. 23-52.
26. Gibert-Ramos A, Andrés-Rozas M, Pastó R, Alfaro-Retamero P, Guixé-Muntet S, et al. Sinusoidal communication in chronic liver disease. Clin Mol Hepatol. 2025 Jan;31(1):32–55.
27. Hánělová K, Raudenská M, Masařík M, Balvan J. Protein cargo in extracellular vesicles as the key mediator in the progression of cancer. Cell Commun Signal. 2024 Jan 10;22(1):25.
28. Liu YG, Jiang ST, Zhang JW, Zheng H, Zhang L, Zhao HT, et al. Role of extracellular vesicle-associated proteins in the progression, diagnosis, and treatment of hepatocellular carcinoma. Cell Biosci. 2024 Sep 3;14(1):113.
29. Addissouky TA. Emerging therapeutics targeting cellular stress pathways to mitigate end-organ damage in type 1 diabetes. Avicenna Journal of Medical Biochemistry. 2024 Jun 30;12(1):39–46.
30. Addissouky TA, Ali MM, El Sayed IE, Wang Y. Type 1 diabetes mellitus: retrospect and prospect. Bulletin of the National Research Centre. 2024 Apr 19;48(1):42.
31. Addissouky T, Ali M, El Sayed IE, Wang Y. Revolutionary innovations in diabetes research: from biomarkers to genomic medicine. Iranian journal of diabetes and obesity. 2023 Dec;15(4):228–42.
32. Addissouky TA. Precision medicine for personalized cholecystitis care: integrating molecular diagnostics and biotherapeutics. Bulletin of the National Research Centre. 2024 Sep 12;48(1):89.
33. Spanos M, Gokulnath P, Chatterjee E, Li G, Varrias D, Das S. Expanding the horizon of EV-RNAs: LncRNAs in EVs as biomarkers for disease pathways. Extracell Vesicle. 2023 Dec;2:100025.
34. Bryja A, Zadka Ł, Farzaneh M, Zehtabi M, Ghasemian M, Dyszkiewicz-Konwińska M, et al. Small extracellular vesicles - A host for advanced bioengineering and "Trojan Horse" of non-coding RNAs. Life Sci. 2023 Nov 1;332:122126.
35. Zhou Z, Shi L, Chen B, Qian H. Regulation of regulated cell death by extracellular vesicles in acute kidney injury and chronic kidney disease. Cytokine Growth Factor Rev. 2024 Apr;76:99–111.
36. Dwivedi OP, Barreiro K, Käräjämäki A, Valo E, Giri AK, Prasad RB, et al. Genome-wide mRNA profiling in urinary extracellular vesicles reveals stress gene signature for diabetic kidney disease. iScience. 2023 Apr 18;26(5):106686.
37. Liu Y, Zheng Y, Yang Y, Liu K, Wu J, Gao P, et al. Exosomes in liver fibrosis: The role of modulating hepatic stellate cells and immune cells, and prospects for clinical applications. Front Immunol. 2023 Mar 20;14:1133297.
38. Oshins R, Greenberg Z, Tai YL, Zhao D, Wang X, Mehrad B, et al. Extracellular vesicle-associated neutrophil elastase activates hepatic stellate cells and promotes liver fibrogenesis via ERK1/2 pathway. bioRxiv. 2024 Aug 21.
39. Addissouky TA, Ali MM, El Sayed IE, Wang Y. Recent advances in diagnosing and treating helicobacter pylori through botanical extracts and advanced technologies. Archives of Pharmacology and Therapeutics. 2023 Nov 3;5(1):53–66.
40. Addissouky TA, Wang Y, El Sayed IE, Baz AE, Ali MM, Khalil AA. Recent trends in Helicobacter pylori management: harnessing the power of AI and other advanced approaches. Beni-Suef University Journal of Basic and Applied Sciences. 2023 Sep 2;12(1):80.
41. Atic AI, Thiele M, Munk A, Dalgaard LT. Circulating miRNAs associated with nonalcoholic fatty liver disease. Am J Physiol Cell Physiol. 2023 Feb 1;324(2):588–602.
42. Colaianni F, Zelli V, Compagnoni C, Miscione MS, Rossi M, Vecchiotti D, et al. Role of Circulating microRNAs in Liver Disease and HCC: Focus on miR-122. Genes (Basel). 2024 Oct 12;15(10):1313.
43. Jouve M, Carpentier R, Kraiem S, Legrand N, Sobolewski C. MiRNAs in Alcohol-Related Liver Diseases and Hepatocellular Carcinoma: A Step toward New Therapeutic Approaches? Cancers (Basel). 2023 Nov 23;15(23):5557.
44. Addissouky TA. Sepsis: A molecular odyssey from infection to organ failure. Journal of Cellular Immunology. 2025 Jul 2;7(2):64–73.
45. Addissouky TA. Nanoscale frontiers in cancer diagnosis and therapy. Discover Chemistry. 2025 Jul 5;2(1):159.
46. Addissouky TA. Beyond insulin: advancing frontiers in cell-based and genetic therapies for type 1 diabetes management. The Egyptian Journal of Internal Medicine. 2025 Jun 18;37(1):84.
47. Owliaee I, Khaledian M, Boroujeni AK, Shojaeian A. Engineered small extracellular vesicles as a novel platform to suppress human oncovirus-associated cancers. Infect Agent Cancer. 2023 Nov 1;18(1):69.
48. Addissouky TA. Translating molecular heterogeneity into precision medicine for advanced liver disease. Archives of Gastroenterology Research. 2024 Nov 6;5(1):47–58.
49. Addissouky TA, Sayed IE, Ali MM, Wang Y. Emerging biomarkers for precision diagnosis and personalized treatment of cystic fibrosis. Journal of Rare Diseases. 2024 Sep 13;3(1):28.
50. Addissouky TA, El Tantawy El Sayed I, Wang Y. Epigenetic factors in posttraumatic stress disorder resilience and susceptibility. Egyptian Journal of Medical Human Genetics. 2025 Mar 20;26(1):50.
51. Addissouky TA. Advancing frontiers in skin offensive odor management: from innovative diagnostics to cutting-edge treatments and emerging technologies. Arch Dermatol Res. 2025 Mar 8;317(1):539.
52. Addissouky TA. Alpha lipoic acid as a modulator of the AMPK-p53 axis: Mechanisms and therapeutic potential in hepatocellular carcinoma progression. Cancer Plus. 2025 Feb 7;6(4):4867.
53. Addissouky TA. Artemisinin and its derivatives throughout the therapeutic mechanisms and clinical potential. Discover Chemistry. 2025 Feb 10;2(1):10.
54. Somnay K, Wadgaonkar P, Sridhar N, Roshni P, Rao N, Wadgaonkar R. Liver Fibrosis Leading to Cirrhosis: Basic Mechanisms and Clinical Perspectives. Biomedicines. 2024 Sep 30;12(10):2229.
55. Lee MJ. A review of liver fibrosis and cirrhosis regression. J Pathol Transl Med. 2023 Jul;57(4):189–95.
56. Wu Z, Xia M, Wang J, Aguilar MM, Buist-Homan M, Moshage H. Extracellular vesicles originating from steatotic hepatocytes promote hepatic stellate cell senescence via AKT/mTOR signaling. Cell Biochem Funct. 2024 Jun;42(4):e4077.
57. Sun C, Xu W, Xia Y, Wang S. PRDM16 from hepatic stellate cells-derived extracellular vesicles promotes hepatocellular carcinoma progression. Am J Cancer Res. 2023 Nov 15;13(11):5254–70.
58. Cheng L, Zhang L, Wang X, Wang Y, Yu J, Li M, et al. Extracellular vesicles in the HCC microenvironment: Implications for therapy and biomarkers. Pharmacol Res. 2024 Nov;209:107419.
59. Shi M, Jia JS, Gao GS, Hua X. Advances and challenges of exosome-derived noncoding RNAs for hepatocellular carcinoma diagnosis and treatment. Biochem Biophys Rep. 2024 Mar 24;38:101695.
60. A Addissouky T. From reactive to proactive: Advances leading the paradigm shift in cholecystitis management. Research in Molecular Medicine. 2023 Aug 10;11(3):161–76.
61. Addissouky TA. From molecular mechanisms to precision medicine: Transformative approaches in cirrhosis management. Archives of Stem Cell and Therapy. 2024 Jul 29;5(1):22–31.
62. Addissouky TA. Personalized pain pathways in liver fibrosis: towards precision hepatology. Avicenna Journal of Medical Biochemistry. 2024 Dec 31;12(2):162–71.
63. Garcia-Martinez I, Alen R, Pereira L, Povo-Retana A, Astudillo AM, Hitos AB, et al. Saturated fatty acid-enriched small extracellular vesicles mediate a crosstalk inducing liver inflammation and hepatocyte insulin resistance. JHEP Rep. 2023 Apr 7;5(8):100756.
64. Guerra F, Ponziani FR, Cardone F, Bucci C, Marzetti E, Picca A. Mitochondria-Derived Vesicles, Sterile Inflammation, and Pyroptosis in Liver Cancer: Partners in Crime or Innocent Bystanders? Int J Mol Sci. 2024 Apr 27;25(9):4783.
65. Liang Y, Kaushal D, Wilson RB. Cellular Senescence and Extracellular Vesicles in the Pathogenesis and Treatment of Obesity-A Narrative Review. Int J Mol Sci. 2024 Jul 20;25(14):7943.
66. Li H, Liu T, Yang Y, Cho WC, Flynn RJ, Harandi MF, et al. Interplays of liver fibrosis-associated microRNAs: Molecular mechanisms and implications in diagnosis and therapy. Genes Dis. 2022 Sep 5;10(4):1457–69.
67. Jiang X, Liu Z, You H, Tang Z, Ma Y, Nie R, et al. Extracellular vesicles derived from bone marrow mesenchymal stem cells ameliorate chronic liver damage via microRNA-136-5p. Mol Cell Biochem. 2025 Feb;480(2):951–69.
68. Tharmalingam J, Gangadaran P, Rajendran RL, Ahn BC. Impact of Alcohol on Inflammation, Immunity, Infections, and Extracellular Vesicles in Pathogenesis. Cureus. 2024 Mar 25;16(3):e56923.
69. Maeda Y, Watanabe Y, Ishikawa N, Yoshida T, Kimura N, Abe H, et al. Platelet-rich plasma-derived extracellular vesicles improve liver cirrhosis in mice. Regen Ther. 2024 Nov 6;26:1048–57.
70. Addissouky TA. Molecular insights into herbal medicines for the treatment of metabolic associated Steatohepatitis. Discover Chemistry. 2025 May 25;2(1):128.
71. Addissouky TA. Innovations in combined cardiac-liver transplantation: robotic-assisted surgery and advanced organ preservation techniques. The Cardiothoracic Surgeon. 2025 Apr 11;33(1):14.
72. Wang H, Yu L, Huang P, Zhou Y, Zheng W, Meng N, et al. Tumor-associated Exosomes Are Involved in Hepatocellular Carcinoma Tumorigenesis, Diagnosis, and Treatment. J Clin Transl Hepatol. 2022 Jun 28;10(3):496–08.
73. Shwe TH, Pothacharoen P, Phitak T, Wudtiwai B, Kongtawelert P. Atorvastatin Attenuates Programmed Death Ligand-1 (PD-L1) Induction in Human Hepatocellular Carcinoma Cells. Int J Mol Sci. 2021 Aug 15;22(16):8755.
74. Zhao L, Pei R, Ding Y, Su Z, Li D, Zhu S, et al. LOXL4 Shuttled by Tumor Cells-derived Extracellular Vesicles Promotes Immune Escape in Hepatocellular Carcinoma by Activating the STAT1/PD-L1 Axis. J Immunother. 2024 Feb-Mar 01;47(2):64–76.
75. Jiang S, Tian S, Wang P, Liu J, Sun K, Zhou X, et al. Native and engineered extracellular vesicles: novel tools for treating liver disease. J Mater Chem B. 2024 Apr 24;12(16):3840–56.
76. Wang J, Wu Z, Xia M, Salas SS, Ospina JA, Buist-Homan M, et al. Extracellular vesicles derived from liver sinusoidal endothelial cells inhibit the activation of hepatic stellate cells and Kupffer cells in vitro. Biochim Biophys Acta Mol Basis Dis. 2024 Mar;1870(3):167020.
77. Xie L, Wang H, Hu J, Liu Z, Hu F. The role of novel adipokines and adipose-derived extracellular vesicles (ADEVs): Connections and interactions in liver diseases. Biochemical Pharmacology. 2024 Apr 1;222:116104.
78. Babuta M, Szabo G. Extracellular vesicles in inflammation: Focus on the microRNA cargo of EVs in modulation of liver diseases. Journal of leukocyte biology. 2022 Jan;111(1):75–92.
79. Lee C, Han J, Jung Y. Pathological Contribution of Extracellular Vesicles and Their MicroRNAs to Progression of Chronic Liver Disease. Biology (Basel). 2022 Apr 21;11(5):637.
80. Addissouky TA, El Sayed IE, Ali MM. Regenerating damaged joints: the promise of tissue engineering and nanomedicine in lupus arthritis. J Clinical Orthopaedics and Trauma Care. 2024 Feb;6(2):1–8.
81. Wang G, Li J, Bojmar L, Chen H, Li Z, Tobias GC, et al. Tumour extracellular vesicles and particles induce liver metabolic dysfunction. Nature. 2023 Jun;618(7964):374–82.
82. Kou M, Huang L, Yang J, Chiang Z, Chen S, Liu J, et al. Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: a next generation therapeutic tool? Cell Death Dis. 2022 Jul 4;13(7):580.
83. Wiest EF, Zubair AC. Challenges of manufacturing mesenchymal stromal cell-derived extracellular vesicles in regenerative medicine. Cytotherapy. 2020 Nov;22(11):606–12.
84. Paolini L, Monguió-Tortajada M, Costa M, Antenucci F, Barilani M, Clos-Sansalvador M, et al. Large-scale production of extracellular vesicles: Report on the "massivEVs" ISEV workshop. J Extracell Biol. 2022 Oct 25;1(10):e63.
85. Jia Y, Yu L, Ma T, Xu W, Qian H, Sun Y, et al. Small extracellular vesicles isolation and separation: Current techniques, pending questions and clinical applications. Theranostics. 2022 Sep 6;12(15):6548–75.
86. Addissouky TA, Wang Y, Megahed FA, El Agroudy AE, El Sayed IE, El-Torgoman AM. Novel biomarkers assist in detection of liver fibrosis in HCV patients. Egyptian Liver Journal. 2021 Nov 23;11(1):86.
87. Addissouky TA, El Agroudy AE, El-Torgoman AM, El Sayed IE, Ibrahim EM. Efficiency of alternative markers to assess liver fibrosis levels in viral hepatitis B patients. Biomedical Research. 2019 Jan 15;30(2):1–6.
88. Kowkabany G, Bao Y. Nanoparticle Tracking Analysis: An Effective Tool to Characterize Extracellular Vesicles. Molecules. 2024 Oct 1;29(19):4672.
89. Arab T, Mallick ER, Huang Y, Dong L, Liao Z, Zhao Z, et al. Characterization of extracellular vesicles and synthetic nanoparticles with four orthogonal single-particle analysis platforms. J Extracell Vesicles. 2021 Apr;10(6):e12079.
90. Addissouky TA, El-Agroudy AE, El-Torgoman AM, El-Sayed IE. Efficacy of biomarkers in detecting fibrosis levels of liver diseases. World Journal of Medical Sciences. 2019 Mar;16(1):11-8.
91. Addissouky T. Detecting liver fibrosis by recent reliable biomarkers in viral hepatitis patients. American Journal of Clinical Pathology. 2019 Oct 1;152:S85.
92. Zhao Z, Wijerathne H, Godwin AK, Soper SA. Isolation and analysis methods of extracellular vesicles (EVs). Extracellular vesicles and circulating nucleic acids. 2021 Mar 30;2(1):80.
93. Hernández A, Arab JP, Reyes D, Lapitz A, Moshage H, Bañales JM, et al. Extracellular Vesicles in NAFLD/ALD: From Pathobiology to Therapy. Cells. 2020 Mar 27;9(4):817.
94. Ferro A, Saccu G, Mattivi S, Gaido A, Herrera Sanchez MB, Haque S, et al. Extracellular Vesicles as Delivery Vehicles for Non-Coding RNAs: Potential Biomarkers for Chronic Liver Diseases. Biomolecules. 2024 Feb 26;14(3):277.
95. Takakura Y, Hanayama R, Akiyoshi K, Futaki S, Hida K, Ichiki T, Ishii-Watabe A, Kuroda M, Maki K, Miura Y, Okada Y, Seo N, Takeuchi T, Yamaguchi T, Yoshioka Y. Quality and Safety Considerations for Therapeutic Products Based on Extracellular Vesicles. Pharm Res. 2024 Aug;41(8):1573–94.
96. Ghodasara A, Raza A, Wolfram J, Salomon C, Popat A. Clinical Translation of Extracellular Vesicles. Adv Healthc Mater. 2023 Nov;12(28):e2301010.