Abstract
Methylphenidate (MPD) is a widely prescribed psychostimulant used to treat behavioral disorders such as attention deficit hyperactivity disorder (ADHD), as well as for cognitive and memory enhancement across all age groups. In addition, MPD is often used—and abused—recreationally by adolescents, adults, and older individuals. The increasing use of MPD among healthy individuals has raised concerns regarding its potential neurological and behavioral consequences, including dependence and abuse. This study examines the behavioral and neuronal effects of MPD by simultaneously recording neural activity from three key monoaminergic brain regions—the locus coeruleus (LC), dorsal raphe nucleus (DR), and ventral tegmental area (VTA)—in freely behaving adolescent and adult rats. Recording electrodes were implanted in these regions, and both behavior and neural activity were recorded using a wireless telemetry system over ten consecutive days. Rats from each age group were assigned to one of the following five experimental groups: time control, saline control, or one of three MPD treatment groups receiving acute and repeated (chronic) doses of 0.6, 2.5, or 10.0 mg/kg. MPD was administered daily from experimental recording (ER) days 1 to 6 (ER1–6), followed by a three-day washout period (ER7–9), and a rechallenge with saline and MPD on ER10. A total of 2,679 neurons were recorded from 311 adolescent and adult male rats. The results revealed significant age- and brain region-dependent differences in both behavioral and neuronal responses to MPD. These findings emphasize the importance of considering both age and specific brain regions when evaluating the effects of psychostimulants. The data suggests that MPD differentially affects the noradrenergic, serotonergic, and dopaminergic systems in adolescents and adults. Furthermore, repeated MPD exposure produced dependency-like behavioral and neuronal patterns in both age groups, underscoring the need for caution in its therapeutic and non-therapeutic use.
Keywords
Methylphenidate, Ritalin, Neuronal and behavior, Age, Monoamines
Introduction
Why study methylphenidate?
Methylphenidate (MPD), commonly known by its trade name Ritalin, is a widely prescribed psychostimulant for the treatment of behavioral disorders such as attention deficit hyperactivity disorder (ADHD) [1–7]. It is primarily used to enhance attention, cognition, planning, and memory [8,9]. Despite its widespread clinical use, its mechanisms of action through which MPD exerts its therapeutic effects remain only partially understood [10]. According to a National Institutes of Health (NIH) study [11], nearly one in five adolescents in the United States has been diagnosed with ADHD and receives psychostimulant treatment. Beyond its medical use, MPD is increasingly used non-medically by students and professionals seeking to enhance cognitive performance and productivity [2,12–15]. In the face of growing academic and professional competition, many individuals turn to MPD to improve cognitive abilities, planning, and memory—and, in some cases, for recreational use [3,14,16–22]. Moreover, adolescents and young adults around the world engage in the illicit sale and use of MPD, not only for its euphoric effects but also to gain an academic advantage [13,23,24]. The concept of enhancing memory and cognition with a pill is no longer science fiction, it is a growing reality.
What is methylphenidate (MPD)?
MPD is a central nervous system (CNS) stimulant structurally related to amphetamine. Neuropharmacological, its profile is similar to that of cocaine [25,26]. Due to its increasing use across all age groups, including adolescents, adults, and the elderly [18,27–31], it is crucial to investigate its long-term effects through behavioral and neurophysiological studies in animal models. MPD primarily affects the brain’s monoaminergic system [32–45], acting as an indirect monoaminergic agonist. It binds to the transporters for norepinephrine (NE), serotonin (5-HT), and dopamine (DA), inhibiting their reuptake from the synaptic cleft into the presynaptic terminals. This inhibition increases monoamine concentrations in the synaptic cleft, thereby enhancing their activity and leading to MPD’s stimulant effects.
What is a drug of abuse?
A drug of abuse is defined as a substance consumed outside of therapeutic contexts to induce pleasure, euphoria, or altered perception. Drug abuse is considered a brain disorder characterized by behaviors such as craving, tolerance, sensitization, withdrawal, dependence, and addiction. Addiction involves compulsive, persistent drug-seeking behavior despite harmful consequences. Craving—a powerful urge to use a drug—can persist for years after drug use has stopped, indicating long-term alterations in brain circuitry [25].
What experimental criteria indicate abuse potential?
A substance may be considered to have abuse potential if it produces the following phenomena in animal models:
- Tolerance: The need to increase the dose to maintain the initial effect.
- Sensitization: A progressive and lasting increase in behavioral response following its repeated exposure.
- Withdrawal: A set of unpleasant or severe behavioral symptoms that appear when drug use stops.
- Dependence: Continued drug use for non-medical purposes [25,42,46].
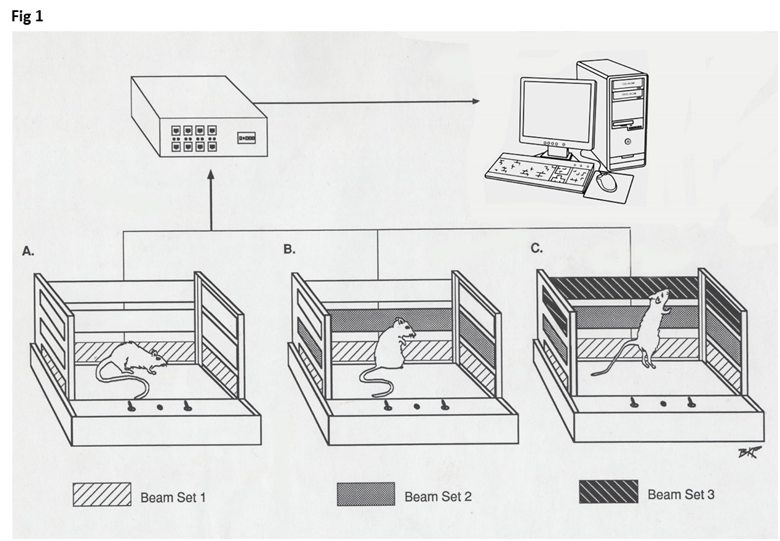
These behavioral responses are widely accepted as experimental indicators of a drug’s potential for dependence and abuse [25,47,48]. Most substances known to be addictive induce these responses, reflecting shared underlying neurobiological mechanisms. Such behavioral manifestations represent complex integrative responses to chronic drug exposure. The open-field recording system (Figure 1), widely used in behavioral pharmacology research due to its simplicity and reliability, captures spontaneous locomotor and exploratory activity and serves as a standard method for evaluating drug-induced behavioral changes.
Figure 1. The figure shows three open-field cages, each equipped with three levels of infrared panels (marked in black). Each cage measures 40 cm in length, 40 cm in width, and 30 cm in height. Each panel contains 16 infrared beams aligned with 16 corresponding sensors on the opposite side to detect beam interruptions caused by the animal's movements. Panels are positioned at 4 cm, 8 cm, and 12 cm above the cage floor. The lowest panel records the number of movements (NM) and total distance (TD) traveled in centimeters. The middle panel records the number of stereotypic movements (NOS). The top panel records vertical activity (VA) before and after drug administration respectively.
Primary research question and hypothesis
The central research question of this study is: Does repeated exposure to MPD produce dependence-like behavioral effects?
Hypothesis
Chronic (repeated) MPD administration will result in behavioral and neurophysiological expression of tolerance, sensitization, and withdrawal. Furthermore, responses to MPD will differ depending on the dose and the age of the subject.
The findings from this study support the hypothesis that chronic MPD exposure in both adolescent and adult rats lead to neuronal and behavioral sensitization, tolerance, and withdrawal. Additionally, the neuronal activity recorded in animals exhibiting behavioral sensitization differed significantly from that observed in animals showing behavioral tolerance.
Materials and Methods
Animals
Adolescent and adult male Sprague-Dawley (SD) rats were obtained from Harlan Laboratories (Indianapolis, IN, USA). Animals were housed individually in standard home cages under controlled temperature and lighting conditions (12-hour light/dark cycle; lights on at 06:00). Food and water were available ad libitum. Rats were allowed to acclimate for 5–6 days prior to electrode implantation. To minimize novelty-induced behavioral effects, all testing was conducted in the animals' home cages, as previously described [36,47–50]. Each age group included five experimental subgroups. The time-control group consisted of eight adolescent and twelve adult rats, while the saline control group included eight adolescent and thirteen adult rats [10,36,47–50]. The methylphenidate (MPD)-treated groups comprised 153 adolescent and 158 adult rats. These rats received 0.6, 2.5, or 10.0 mg/kg MPD once daily over six consecutive experimental recording days (ER1–ER6), followed by a three-day washout period (ER7–ER9) and a final rechallenge on ER10 with either saline or 0.6, 2.5 or 10.0 mg/kg MPD injection.
Surgical procedure and electrode implantation
Rats were anesthetized with intraperitoneal (i.p) pentobarbital. The scalp was shaved and disinfected with lidocaine hydrochloride gel. Each animal was then secured in a stereotaxic frame, and a midline scalp incision exposed the skull. Bilateral electrodes (60 µm, Teflon-coated nickel-chromium, exposed only at the tip) were stereotaxic ally implanted into the locus coeruleus (LC), dorsal raphe (DR), and ventral tegmental area (VTA) [36,47–50]. Implantation coordinates were determined using age-specific brain atlases: Sherwood and Timiras [51] for adolescent rats and Paxinos and Watson [52] for adults. Following surgery, animals recovered for 5–7 days and were habituated daily (~2 hours/day) to the wireless recording system (TBSI; Triangle BioSystems Inc., Durham, NC). Recordings began on postnatal day P - 40 for adolescents and P - 60 for adults. All procedures were approved by the University of Texas Health Science Center Animal Welfare Committee and complied with NIH guidelines.
Locomotor behavioral activity
Behavioral activity was recorded in the home cage using the Computerized Animal Activity Monitoring (CAAM) system (Accuscan Instruments, Columbus, OH), which consist of an array of three levels of 8 infrared beams in each of the three levels (Figure 1). Data were collected in 10-minute bins (six bins/hour) and analyzed using OASIS software. Four locomotor indices were evaluated based on infrared beam breaks by the animals at different vertical levels: Number of movements (NM). Total distance (TD) traveled (in cm). Number of stereotypic (NOS) repetitive movements and Vertical activity (VA) as previously described [36,47–50].
Neuronal activity
Neuronal activity from the LC, DR, and VTA was recorded simultaneously using a lightweight wireless headstage (<4.5 g) that captured and transmitted spike activity to a receiver outside the testing cage. The receiver was connected to a Micro1402-3 analog-to-digital converter and a PC, where the data were stored for offline analysis using Spike2 v7 software (CED, Cambridge Electronic Design, UK).
Experimental protocol
On ER1, rats were placed in a Faraday cage within their home cage and allowed to acclimate for 30 minutes. Baseline recordings (ER1 BL) of both behavior and neuronal activity were then collected for 60 minutes following a saline injection, followed by 60 minutes of post-MPD recording (ER1 MPD). Daily MPD administration continued from ER1 through ER6. No injections were administered during the washout period (ER7–ER9). On ER10, the ER1 protocol was repeated: 60 minutes of baseline recording after saline injection (ER10 BL), followed by 60 minutes of recording after MPD injection (ER10 MPD; 0.6, 2.5, or 10.0 mg/kg). A complete timeline is provided in Table 1.
|
Treatment Groups Adolescent |
ER 1* |
ER 2–6 |
ER 7–9 |
ER 10* |
|
|
1 |
Saline |
Saline/Saline |
Saline |
Washout |
Saline/Saline |
|
2 |
0.6 mg/kg MPD |
Saline/0.6 mg/kg MPD |
0.6 mg/kg MPD |
Washout |
Saline/0.6 mg/kg MPD |
|
3 |
2.5 mg/kg MPD |
Saline/2.5 mg/kg MPD |
2.5 mg/kg MPD |
Washout |
Saline/2.5 mg/kg MPD |
|
4 |
10.0 mg/kg MPD |
Saline/10.0 mg/kg MPD |
10.0 mg/kg MPD |
Washout |
Saline/10.0 mg/kg MPD |
|
|
|
|
|
|
|
|
|
Treatment Groups Adult |
ER 1* |
ER 2–6 |
ER 7–9 |
ER 10* |
|
1 |
Saline |
Saline/Saline |
Saline |
Washout |
Saline/Saline |
|
2 |
0.6 mg/kg MPD |
Saline/0.6 mg/kg MPD |
0.6 mg/kg MPD |
Washout |
Saline/0.6 mg/kg MPD |
|
3 |
2.5 mg/kg MPD |
Saline/2.5 mg/kg MPD |
2.5 mg/kg MPD |
Washout |
Saline/2.5 mg/kg MPD |
|
4 |
10.0 mg/kg MPD |
Saline/10.0 mg/kg MPD |
10.0 mg/kg MPD |
Washout |
Saline/10.0 mg/kg MPD |
Methylphenidate (MPD) administration
MPD hydrochloride (Mallinckrodt, Hazelwood, MO, USA) was dissolved in 0.9% isotonic saline. MPD doses of 0.6, 2.5, and 10.0 mg/kg (free base) were administered i.p. in 0.8 ml volumes. Prior studies have demonstrated significant behavioral effects at doses as low as 0.6 mg/kg (34, 91). MPD brain concentrations are similar across i.p., intravenous (i.v.), and oral routes [47,48,53–55]. These three doses represent low, moderate, and high MPD treatment levels [36,47–49].
Electrode placement verification
After ER10 recordings, animals were euthanized with an overdose of sodium pentobarbital and perfused with 10% formalin containing 3% potassium ferrocyanide. A 10 µA direct current was passed through each electrode for 20 seconds to mark the electrode tip. Brains were sectioned, fixed, and examined histologically using the Paxinos and Watson [52] brain atlas. Only data from animals with verified electrode placements in the LC, DR, and VTA and consistent spike activity patterns from ER1 to ER10 were included in the final analysis [36,47,49,50,56,57].
Data acquisition and analysis
Behavioral data
Each session consisted of two 60-minute periods: post-saline and post-MPD administration. Acute MPD effects were evaluated by comparing behavioral and neuronal activity during ER1 MPD to ER1 BL (ER1 MPD / ER1 BL). Chronic MPD effects were assessed by comparing ER10 MPD to ER1 MPD (ER10 MPD / ER1 MPD). Paired t and the CR tests were used for comparisons [36,47–50,53,55–59,]. Rats exhibiting significance increased activity on ER10 MPD vs. ER1 MPD (p<0.05) were classified as sensitized, while those expressing significance (p<0.05) reduced activity were classified as tolerant [36,47–50,53,56–59].
Neuronal data
Spike sorting was conducted offline using CED Spike2 v7. Signals were bandpass-filtered (0.3–3.0 kHz) and sorted using dual-window discrimination. Templates were generated from 1,000 waveform samples with ≥95% match accuracy. ER1 templates were reused in ER10 to ensure consistent evaluation [36,47–50,53,55–59]. Neuronal firing rates were compared across: Acute effect: ER1 MPD vs. ER1 BL. Withdrawal effect: ER10 BL vs. ER1 BL. Chronic effect: ER10 MPD vs. ER1 MPD. Statistical analyses included Student’s t-test and the Critical Ratio (CR) test [36,47–50,53,57]. Results were summarized by brain region and behavioral subgroup (All, Sensitized, Tolerant; see Table 3).
Statistical analysis
Individual rat behaviors and neuronal firing patterns were analyzed using paired t-tests and CR tests, respectively. For group-level comparisons, Chi-square tests were used to assess differences in the proportion of animals or neurons exhibiting facilitation or attenuation. Group data were analyzed using repeated-measures ANOVA with Tukey’s post hoc tests and Chi-square analysis, using SPSS v21 software [36,47–50,53,55,57].
Results
A total of 311 male Sprague-Dawley (SD) rats were used in this study: 153 adolescents and 158 adults. Each age group was divided into five subgroups: time control, saline control, and three MPD-treated groups (0.6, 2.5, and 10.0 mg/kg).
Locomotor behavioral observations
Time and saline (Sal) control groups
Across all recoding days, both adolescent and adult rats in the time control groups exhibited consistent levels of locomotor activity—measured by number of movements (NM), total distance traveled (TD, in cm), number of stereotypic movements (NOS), and vertical activity (VA). Comparisons between ER1 and subsequent experimental days showed no significant changes in locomotor measures [10,36,47,49,53,55,60].
Similarly, single or repeated saline injections did not significantly alter locomotor activity relative to ER1 baseline (BL) values. These findings indicate that handling, injection procedures, and environmental conditions did not influence locomotor behavior [36,47–50,53,57].
Behavioral effects of MPD
In previous studies [54,55], some animals groups did not show statistically significant changes in locomotor activity upon MPD rechallenge (ER10 MPD vs. ER1 MPD). However, individual analyses revealed two distinct patterns: i.e., some individual rats expressed behavioral sensitization. i.e., the rats displayed increased locomotor activity at ER10 MPD compared to ER1 MPD and some individual rats expressed behavioral tolerance. These rats exhibited reduced activity at ER10 MPD compared to ER1 MPD. When both subgroups were analyzed together (All group), the opposing effects masked any overall changes, resulting in no significant MPD effect. Therefore, in this study rats were categorized into three behavioral outcome groups for each MPD dose and age: All groups, include all the rats in the study. Sensitized groups, included only rats that expressed behavioral sensitization and Tolerance groups, included only rats expressing behavioral tolerance respectively.
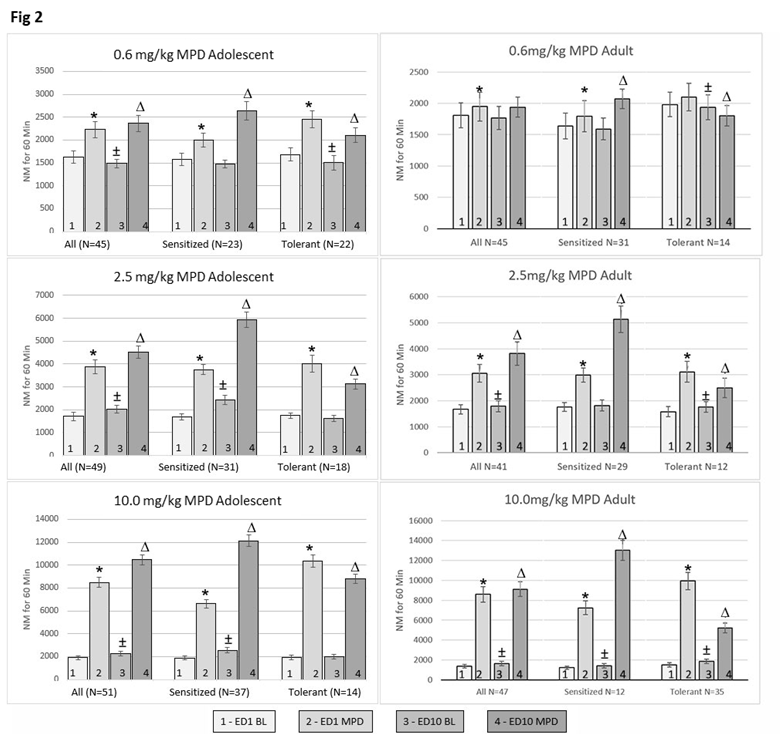
Behavioral effects of MPD (Figure 2)
All groups
Acute MPD effects (ER1 MPD/ER1 BL): Administration of 0.6, 2.5, or 10.0 mg/kg MPD significantly increased NM (P < 0.01) in both adolescent and adult rats, indicating acute locomotor stimulation (Figure 2).
Chronic MPD effects on BL activity (ER10 BL/ER1 BL): Adolescents rats showed significant increases in ER10 BL/ER1 BL NM across all MPD doses groups after six days of treatment and a three-day washout (P<0.01) while adults rats groups showed no change at 0.6 mg/kg MPD group, but significant increases in NM at 2.5 and 10.0 mg/kg MPD groups (Figure 2).
Chronic MPD rechallenge (ER10 MPD/ER1 MPD): Both age groups demonstrated significant increases in NM (P<0.01) at ER10 MPD/ ER1 MPD indicating expression of behavioral sensitization.
Sensitized groups
Acute MPD effects (ER1 MPD/ER1 BL): Significant increases in NM were observed in both age groups across all MPD doses (P<0.01) groups (Figure 2 – middle panel).
Chronic effects on baseline (ER10 BL/ER1 BL): Adolescents rats show no change at ER10 BL/ER1 BL at 0.6 mg/kg MPD group, but significant (P<0.01) increases in NM at 2.5 and 10.0 mg/kg MPD groups, while adult rats at 10.0 mg/kg group showed a significant (P<0.01) increase in NM at ER10 BL/ER1 BL.
MPD rechallenge (ER10 MPD/ER1 MPD): All sensitized animals exhibited significant increases in NM after MPD rechallenge across all doses and age groups (P<0.01) (Figure 2 – middle panel).
Tolerant groups
Acute MPD effects (ER1 MPD/ER1 BL): Significant increases in NM were observed in both adolescent and adult tolerant rats for all acute MPD doses (P<0.01) groups (Figure 2 – right panel).
Chronic effects on BL (ER10 BL/ER1 BL): Adolescents rats treated for six daily doses of 0.6 mg/kg MPD and three days of washout exhibited significant decrease (P<0.01) in NM on ER10 BL/ER1 BL and no changes in NM at 2.5 or 10.0 mg/kg MPD groups while adult rats groups, exhibit no change at 0.6 mg/kg MPD group and significant increases at the 2.5 and 10.0 mg/kg MPD (P<0.01) groups respectively.
MPD rechallenge (ER10 MPD/ER1 MPD): Both adolescent and adult behavioral tolerant rat groups showed significant reductions in NM at ER10 compared to ER1 (P<0.01), indicating behavioral tolerance.
Sensitization vs. tolerance ratios (Chi-square analysis)
The proportion of adolescent rats compared to adult rats expressing behavioral tolerance vs behavioral sensitization was significantly (P<0.01 )different using Chi-square analysis: Adolescent rats exhibit the following behavioral sensitization vs. tolerance ratio 23/22, 31/18, and 37/14 following 0.6, 2.5, and 10.0 mg/kg MPD respectively, while the adult rats groups exhibit behavioral sensitization vs. tolerance ratio 31/14, 29/12, and 12/35, following 0.6, 2.5, and 10.0 mg/kg MPD, respectively (Figure 2 – Sensitized/Tolerant)
Figure 2. This figure summarizes the number of movements (NM) for adolescent and adult rats across treatment groups. For each MPD dose (0.6, 2.5, and 10.0 mg/kg), rats were classified into three subgroups: “All animals”, animals expressing behaviorally “sensitized”, and animals expressing behaviorally “tolerant”. The “All” animals group includes all animals that received a given MPD dose. The "Sensitized" and "Tolerant" groups include only those animals that exhibited behavioral sensitization or tolerance, respectively, based on comparisons between the initial (acute) MPD exposure (ER1) and after six daily MPD exposures followed by three drug-free days (ER10 MPD/ER1 MPD). Each histogram displays four bars: ER1 Baseline (BL), ER1 MPD, ER10 BL, and ER10 MPD. These are used in three comparisons: ER1 MPD vs. ER1 BL (ER1 MPD/ER1 BL) – to assess the acute MPD effect. ER10 BL/ ER1 BL – to assess changes after repeated exposure and withdrawal. ER10 MPD/ER1 MPD – to assess the chronic MPD effect. Additionally, ER1 MPD values are compared between adolescents and adults to evaluate age-related differences in the acute response; likewise, ER10 MPD values are compared to ER1 MPD to assess chronic response of MPD differences. SD values are shown above each column. * = indicates significant difference from ER1 BL (acute effect, p<0.05). ‡ = indicates significant difference between ER10 BL/ER1 BL (withdrawal effect, p<0.05). ^ = indicates significant difference from ER1 MPD (chronic effect, p<0.05). N = number of rats per group.
Neuronal recordings
A total of 2,679 neurons were recorded: 1,460 from adolescents and 1,219 from adults, following saline or MPD administration.
Saline controls
Saline and repetitive daily saline injections did not alter neuronal activity in the LC, DR, or the VTA compared to untreated control recording, indicating that handling and injection procedures did not modify the LC, DR, and VTA neuronal firing rates.
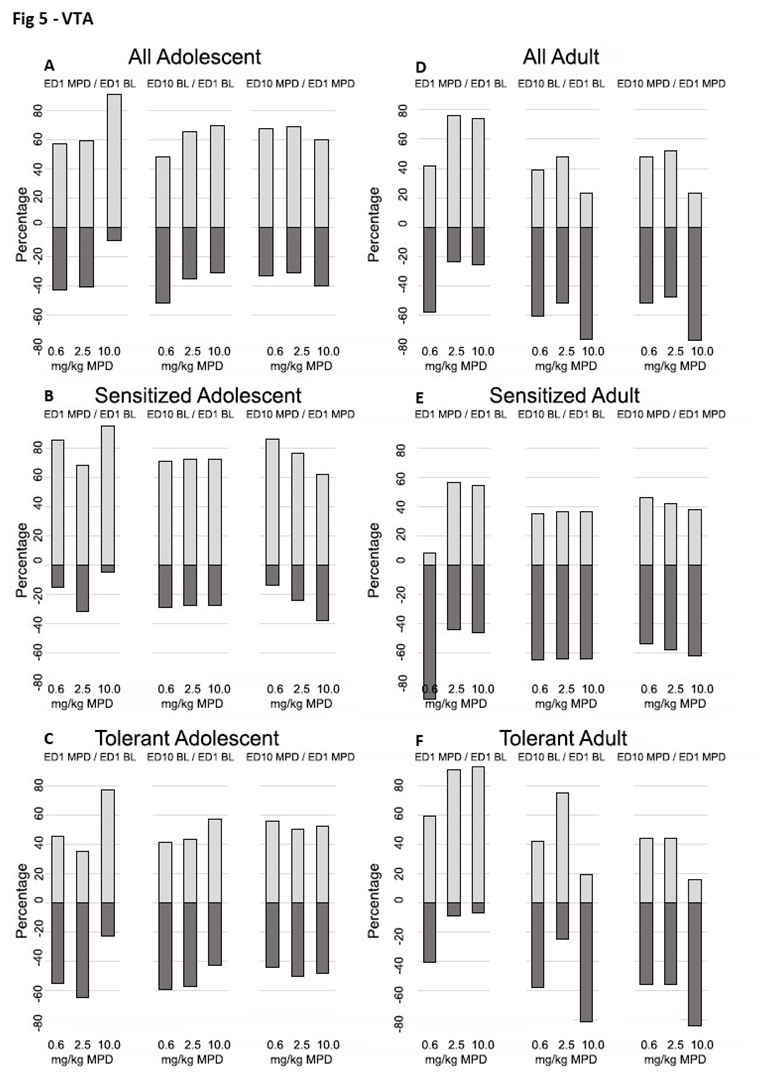
Neuronal responses to 0.6, 2.5, and 10.0 mg/kg MPD – All rats groups (Figures 3–5)
Neuronal recordings before and following acute 0.6, 2.5 and 10.0 mg/kg MPD exposure (Table 2) and chronic MPD exposure included 420, 447, and 435 neurons recorded from adolescent LC, DR, and VTA regions, respectively, and 405, 329, and 372 neurons from adult rats across the same regions respectively. The tables summarize how many LC, DR and VTA neurons responded to acute MPD i.e., ER1 MPD/ER1 BL and chronic MPD compared to acute MPD, ER10 MPD/ER1 MPD. Figures 3–5 summarized the numbers of LC, DR, and VTA neurons that respond significantly (p<0.01) in percentage of the number of the recorded neurons from each brain area and MPD dose groups respectively by increase as compared to decrease in firing rates as result of 0.6, 2.5 and 10.0 mg/kg MPD administration at ER10 MPD/ER1 MPD, and ER10 BL/ER1 BL and show significant (p<0.01 different within the different MPD doses groups and between the two age groups (Figures 3–5). In adolescent rats, LC neurons showed higher responsiveness to MPD application than the DR and VTA recorded neurons. In adult rats the VTA neurons were more responsive than LC or DR neurons to MPD treatment (Tables 2 and 3).
|
Teenage |
LC |
DR |
VTA |
|||
|
Dose |
n |
Responsible |
n |
Responsible |
n |
Responsible |
|
Sal |
56 |
4 (7.1%) |
57 |
3 (3.5%) |
45 |
2 (4.4%) |
|
0.6 mg/kg MPD |
134 |
83 (61.9%) |
137 |
71 (57.8%) |
141 |
40 (28.3%) |
|
2.5 mg/kg MPD |
146 |
92 (63.0%) |
142 |
89 (62.7%) |
142 |
78 (54.9%) |
|
10 mg/kg MPD |
140 |
128 (91.4%) |
168 |
134 (79.8%) |
152 |
105 (69.1%) |
|
Total = 480 |
Total = 476 |
Total = 504 |
||||
|
Adult |
LC |
DR |
VTA |
|||
|
Dose |
n |
Responsible |
n |
Responsible |
n |
Responsible |
|
Sal |
60 |
2 (3.3%) |
27 |
1 (3.7%) |
26 |
1 (3.8%) |
|
0.6 mg/kg MPD |
128 |
50 (39.0%) |
122 |
58 (47.5%) |
171 |
112 (65.5%) |
|
2.5 mg/kg MPD |
136 |
106 (77.9%) |
94 |
54 (57.4%) |
102 |
80 (78.4%) |
|
10 mg/kg MPD |
141 |
128 (90.8%) |
113 |
83 (73.5%) |
99 |
70 (70.7%) |
|
Total = 465 |
Total = 356 |
Total = 398 |
||||
|
EDIO BL,'EDI BL |
||||||||||||
|
|
VTA |
LC |
DR |
|||||||||
|
MPD dose, mg/kg |
n |
↑ |
↓ |
↔ |
n |
↑ |
↓ |
↔ |
n |
↑ |
↓ |
↔ |
|
A. units recorded from altogether animals |
||||||||||||
|
saline |
36 |
0 (0%) |
1 (2.8%) |
35 (97.2%) |
56 |
3 (5.4%) |
3 (5.4%) |
50 (89.2%) |
27 |
1 (3.7%) |
2 (7.4) |
24 (88.9%) |
|
0.6 |
115 |
28 (24.3%) |
30 (26.0%) |
57 (49.6%) |
109 |
21 (19.3%) |
34 (31.2%) |
54 (49.5%) |
122 |
51 (41.8%) |
56 (46.7%) |
14 (11.5%) |
|
2.5 |
135 |
55 (40.8%) |
30 (22.2%) |
50 (37.0%) |
132 |
59 (44.7%) |
40 (30.3%) |
33 (25%) |
94 |
35 (37.2%) |
32 (34.0%) |
27 (28.8%) |
|
10 |
117 |
74 (63.3%) |
34 (29.0%) |
9 (7.7%) |
112 |
84 (75.0%) |
14 (12.5%) |
14 (12.5%) |
113 |
37 (32.7%) |
61 (54.0%) |
15 (13.3%) |
|
B. units recorded from sensitized animals |
||||||||||||
|
0.6 |
38 |
10 (26.3%) |
4 (10.5%) |
24 (63.2%) |
47 |
909.1%) |
13 (27.7%) |
25 (53.2%) |
27 |
11 (40.7%) |
9 (33.3%) |
7 (26%) |
|
2.5 |
80 |
46 (57.5%) |
18 (22.5%) |
16 (20%) |
61 |
36 (59.0%) |
12 (19.7%) |
13 (21.3%) |
42 |
13 (31.0%) |
22 (52.4%) |
7 (16.6%) |
|
10 |
92 |
61 (66.3%) |
24 (26.1%) |
7 (7.6%) |
81 |
63 (77.8%) |
9(11.1%) |
9(11.1%) |
20 |
4 (20.0%) |
15 (75.0%) |
1 (5.0%) |
|
C. units recorded from tolerant animals |
||||||||||||
|
0.6 |
77 |
18 (23.4%) |
26 (33.8%) |
33 (42.8%) |
62 |
12 (19.4%) |
21 (33.9%) |
29 (46.8%) |
95 |
38 (40.0%) |
49 (51.6%) |
8 (8.4%) |
|
2.5 |
55 |
9 (16.4%) |
12 (21.8%) |
34 (61.8%) |
71 |
23 (32.4%) |
28 (39.4%) |
20 (28.2%) |
52 |
21 (40.4%) |
11 (21.2%) |
20 (38.4%) |
|
10 |
25 |
13 (52%) |
10 (40%) |
2 (8%) |
31 |
21 (67.8%) |
5 (16.1%) |
5 (16.1%) |
93 |
32 (34.4%) |
46 (49.5%) |
15 (16.1%) |
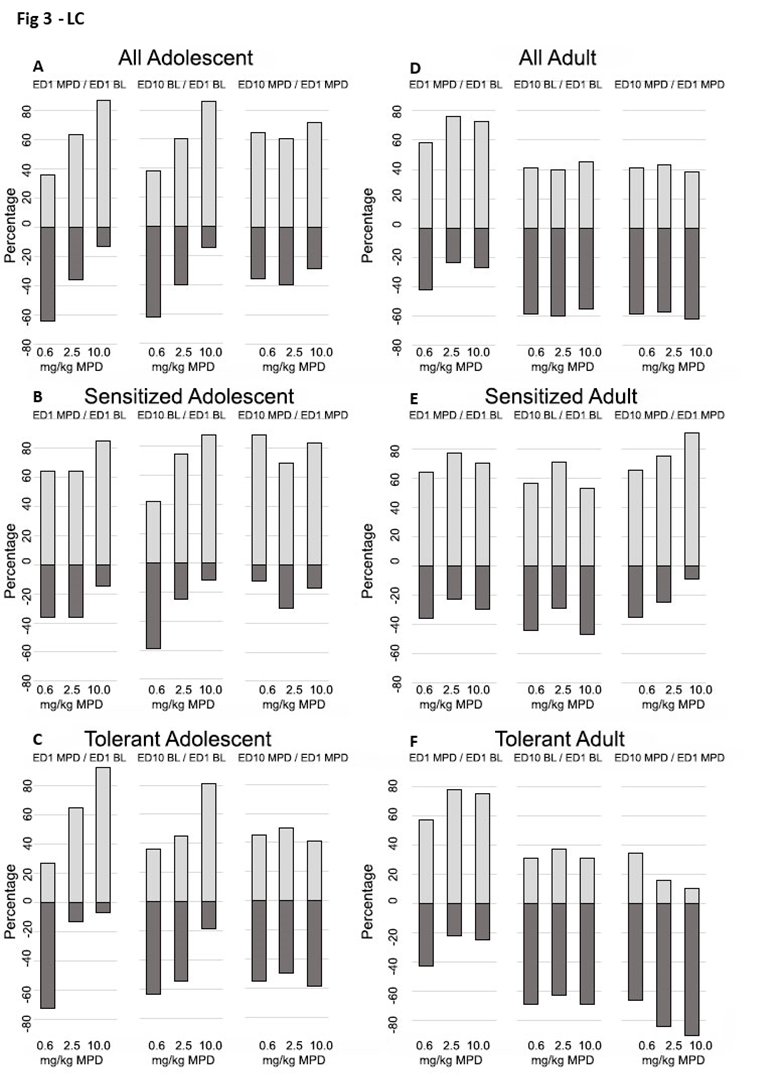
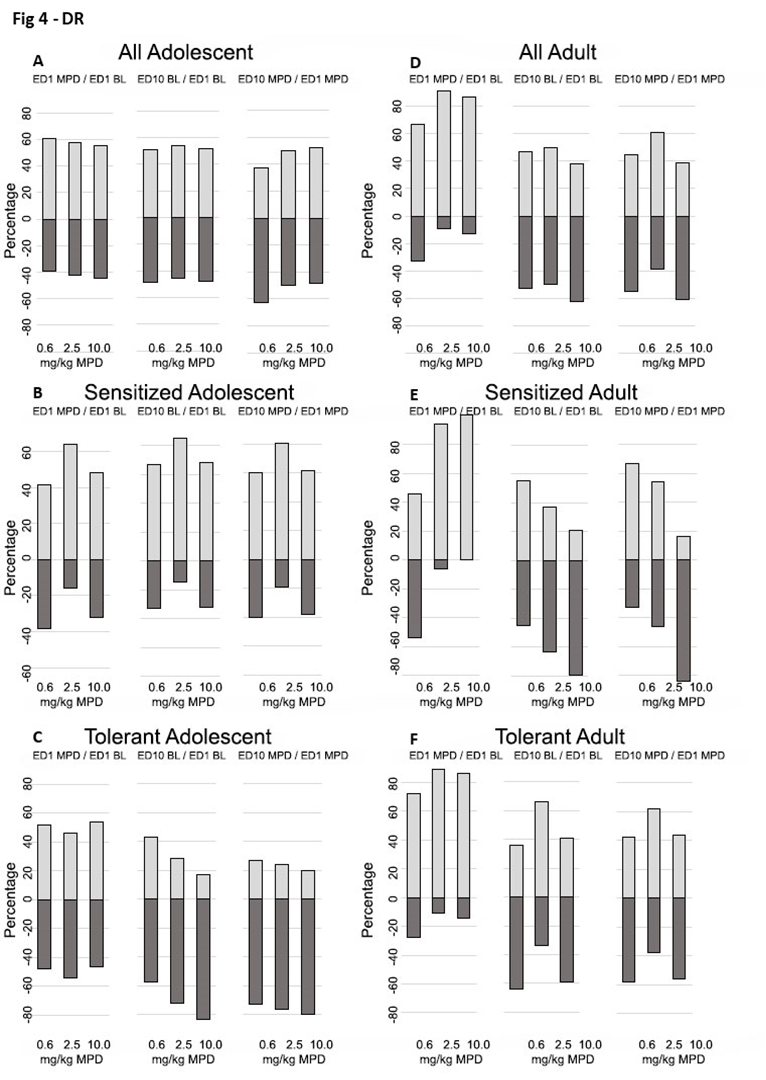
Neuronal responses recorded from behavioral sensitization group to acute and chronic MPD (Figures 3–5)
Figures 3–5 summarizes how many neurons (in percentage) respond significantly (p<0.01) to 0.6 mg, 2.5 mg and 10.0 mg/kg MPD by significant (p<0.01) increase or decrease in their firing rates recorded from the LC (Figure 3), DR (Figure 4), and VTA (Figure 5) on ER1 MPD/ER1 BL and ER10 MPD/ER1 MPD respectively. The ratio of how many LC, DR, and VTA neurons responded by significant (p<0.01) attenuation or facilitation and magnitude in percentage to acute and chronic MPD were significantly different (P<0.01) between the LC, DR, and VTA neurons within each MPD dose groups and between adolescent and adult groups.
Neuronal responses recorded from behavioral tolerance group to acute and chronic MPD (Figures 3–5)
In animals expressing behavioral tolerant groups the LC, DR, and VTA neuronal responses to MPD were significantly different (P<0.01) how many neurons from each of this area responded by attenuation versus facilitation to different MPD doses. The proportion of LC, DR, and VTA neurons that responded to MPD exposure with increased vs. decreased activity also varied significantly within each age group (i.e., those treated with 0.6 mg/kg MPD compared to the other two MPD dose groups and between the adolescent and the adult groups (P<0.01).
Neuronal activity of the ER10 BL/ER1 BL after six daily MPD exposure and three wash out days (Figures 3–5)
All group: The LC and VTA recorded neurons exhibited similar ratio how many of them exhibited modification in their neuronal firing rates at ER10 vs, ER1 after six daily MPD exposure and three washout days while the DR neuronal recording exhibited different ratio in the neurons that recorded from adolescent All group. In the sensitized and tolerance groups each MPD dose elicit different ratio how many neurons exhibited increase or decrease in their ER10 BL/ER1 BL. In the adult rats’ groups, the neurons recorded from the LC, DR and the VTA exhibited different ratios of how many neurons expressed facilitation to attenuation.
Sensitization group: Significant differences (P<0.01) in ER10 BL/ER1 BL were observed across the neurons recorded from the three brain regions and age groups after six daily 0.6, or 2.5 or 10.0 mg/kg MPD and three washout days.
Tolerance group: Significant (P<0.01) difference in how many neurons in ED10 BL after six daily MPD exposure and three wash out days (ER10 BL/ER1 BL) exhibited significant different changes (P<0.01) within each age group following 0.6, 2.5, or 10.0 mg/kg MPD and between the two age groups respectively.
Figure 3. This figure summarizes the percentage of LC neurons that showed significant (p<0.05) increases or decreases in firing rates in response to acute or chronic MPD treatment. Each segment includes three columns representing: ER1 MPD/ER1 BL (acute MPD effect), ER10 BL/ER1 BL (baseline change indicate withdrawal, ER10 MPD/ER1 MPD (chronic MPD effect). Results are grouped into: Panels A and D: All recorded LC neurons. Panels B and E: LC neurons from behaviorally sensitized animals. Panels C and F: LC neurons from behaviorally tolerant animals, in adolescent and adult groups, respectively.
Figure 4. This figure summarizes the percentage of DR neurons that showed significant (p<0.05) increases or decreases in firing rates in response to acute or chronic MPD treatment. Each segment includes three columns representing: ER1 MPD/ER1 BL (acute MPD effect), ER10 BL/ER1 BL (baseline change indicate withdrawal, ER10 MPD/ER1 MPD (chronic MPD effect). Results are grouped into: Panels A and D: All recorded DR neurons. Panels B and E: DR neurons from behaviorally sensitized animals. Panels C and F: DR neurons from behaviorally tolerant animals, in adolescent and adult groups, respectively.
Figure 5. This figure summarizes the percentage of VTA neurons that showed significant (p<0.05) increases or decreases in firing rates in response to acute or chronic MPD treatment. Each segment includes three columns representing: ER1 MPD/ER1 BL (acute MPD effect), ER10 BL/ER1 BL (baseline change indicate withdrawal, ER10 MPD/ER1 MPD (chronic MPD effect). Results are grouped into: Panels A and D: All recorded VTA neurons. Panels B and E: VTA neurons from behaviorally sensitized animals. Panels C and F: VTA neurons from behaviorally tolerant animals, in adolescent and adult groups, respectively.
Discussion
Methylphenidate (MPD) is a psychostimulant widely used to treat behavioral disorders and to enhance cognitive functions such as attention, memory, and planning in adolescents, adults, and the elderly [4,9,61,62]. MPD exerts its effects by binding to monoamine transporters—norepinephrine (NE) [63,64], serotonin (5-HT) [65–67], and dopamine (DA) [68,69]—at the synaptic cleft, thereby preventing their reuptake into presynaptic terminals. This inhibition prolongs the action of these neurotransmitters on postsynaptic receptors [26,45–48]. NE is primarily synthesized in the LC [70,71], 5-HT in the DR [58,72–74], and DA in the VTA [53,56,59,75]. These nuclei project to both subcortical and cortical regions, modulating diverse brain functions. Studying the effects of MPD on neuronal activity in LC, DR, and VTA alongside behavioral responses is essential to understand the mechanisms underlying MPD’s actions. NE, 5-HT, and DA are critical neurotransmitters in both the central and peripheral nervous systems, each playing distinct roles in regulating behavior, mood, arousal, cognition, motor function, and physiological responses. NE is central to attention, wakefulness, memory, and the “fight or flight” response, and regulates sympathetic nervous system activity [34,37,42,63,69,70]. 5-HT is involved in mood regulation, appetite, sleep, and emotional processing [21,32,33,35,38,42,44,45,57,65,73], while DA contributes to motivation, reward, learning, executive function, and motor activity [39,40,42,44,69].
Neurophysiological findings
Acute MPD exposure
Acute administration of MPD induced both excitatory and inhibitory responses in LC, DR, and VTA neurons, in a dose-dependent manner. Higher doses produced more pronounced effects, and these responses differed between adolescent and adult rats (Table 1; Figures 3–5 B–F; ER1 MPD/ER1 BL).
Baseline shift post-chronic MPD
After six consecutives daily MPD injections (ER1–ER6) and a three-day washout (ER7–ER9), baseline neuronal activity at ER10 differed significantly from initial ER1 baseline, indicating neurophysiological withdrawal (ER10 BL/ER1 BL).
Chronic MPD effects
Repeated MPD exposure (0.6, 2.5, 10.0 mg/kg) resulted in either neuronal tolerance (reduced response) or sensitization (enhanced response) at ER10 compared to ER1 (ER10 MPD/ER1 MPD; Figures 3–5). These changes were dose-dependent and varied by brain region and age.
Differential responsivity
The proportion of neurons showing altered firing (either facilitation or attenuation) varied across brain regions and age groups, indicating both region- and age-dependent neurophysiological responses to MPD.
Brain region- and age-specific MPD dose effects
The ratio of how many LC, DR, or VTA neurons responded by excitatory tor inhibitory responses to MPD (0.6, 2.5, 10.0 mg/kg) differed significantly across the LC, DR, and VTA, and between adolescents and adults and MPD doses groups suggesting age, brain site and dose-specific patterns of MPD modulation.
Behavioral findings
Acute MPD effects
Acute MPD administration resulted in dose-dependent alterations in locomotor activity—either stimulation or suppression—depending on dose and age group (Figure 2).
Baseline locomotor activity shift post-chronic MPD
After six days of MPD treatment and three days of washout, BL locomotor activity at ER10 was significantly different from ER1 BL, indicating behavioral withdrawal (ER10 BL/ER1 BL).
Chronic MPD effects
Chronic MPD exposure induced either behavioral tolerance or sensitization compared to ER1 MPD effect responses in a dose- and age-dependent manner (ER10 MPD/ER1 MPD; Figure 2).
Differential responsivity
The proportion of animals showing behavioral attenuation or facilitation differed by MPD dose within each age group and between adolescents and adults.
Dose- and age-dependent responses
The number of animals that developed tolerance or sensitization to chronic MPD exposure as compared to the initial MPD effect varied by dose and age, emphasizing the importance of developmental stage in MPD responsiveness.
Age-dependent behavioral profiles
Behavioral sensitivity to MPD differed consistently between adolescents and adults at all tested doses, reinforcing the influence of age on MPD dose effects.
Interpretation and implications
The finding that MPD doses (0.6, 2.5, or 10.0 mg/kg) can lead to divergent behavioral outcomes—tolerance in some animals and sensitization in others—highlights the complexity of MPD’s action. These results underscore the importance of concurrently examining neuronal and behavioral responses during chronic MPD exposure whether the recording was obtained from animals expressing behavioral tolerance versus sensitization. Neurophysiological data confirm that the LC, DR, and VTA all participate in MPD’s effects, but each contributes uniquely. NE, 5-HT, and DA systems are differentially engaged based on MPD dose and age. Each brain region exhibited a distinct ratio of excitatory to inhibitory neurons, suggesting that the different ratio of the dual mechanism (excitation and inhibition) in the LC, DR, and the VTA underpinning region-specific outcomes.
These findings of age-related differences emphasize the need for age-specific clinical guidelines when prescribing MPD. Similar brain region specific and age dependent were reported using PET imaging in humans’ study in response to MPD [76]. MPD demonstrates markedly higher affinity for norepinephrine (NET) and dopamine (DAT) transporters compared to serotonin (5-HTT) [45,77,78]. Moreover, the cellular composition and neurochemical profile of each region determine its response to MPD [79]. Repeated MPD exposure leads to region-specific alterations in NE, 5-HT, and DA transmission. These monoaminergic systems—and their respective nuclei (LC, DR, VTA)—play crucial roles in neuropsychiatric health, drug response, and neuroadaptations linked to psychostimulant exposure.
Relevance to drug dependence and neuroplasticity
Chronic exposure to psychostimulants like MPD leads to long-term behavioral and neurochemical adaptations, reflecting underlying neuroplastic changes associated with drug dependence. Tolerance and sensitization are well-established indicators of such neuroadaptations and are considered early markers of addictive potential [80–85]. The tolerance and sensitization expressing are considered early indicators of neuroplastic changes associated with addiction and are essential for understanding the development of compulsive drug use [82]. Tolerance and sensitization are used widely as an experimental model for studying changes in the neural circuitry of motivation and reward systems [47,48,83,84]. Developing tolerance and behavioral sensitization include: increased self-administration of psychostimulants [86,87]; enhanced conditioned place preference [42,88-90]; increased locomotor activity [28,55,89,90]. Locomotor activity recording serves as a practical experimental measure for assessing whether behavioral, tolerance, and sensitization has developed, providing a reliable model for the enduring effects of psychostimulant exposure [24,47-50,53-60,81].
Conclusions
Most previous studies on methylphenidate (MPD) have focused on the acute effects of a single dose on behavioral responses or on activity within a single brain structure. In contrast, this study investigated the dose–response effects of both acute and chronic MPD administration, alongside its influence on neuronal activity across multiple brain regions that serve as major sources of dopamine (DA), norepinephrine (NE), and serotonin (5-HT). While published reports suggest that MPD primarily affects the dopaminergic system, the findings indicate that the NE and 5-HT systems also play significant roles in MPD’s actions. Each of these monoamine systems (DA, NE, and 5-HT) contribute to the overall pharmacological and behavioral effects of MPD. Significant age-related differences were observed in behavioral responses to MPD. Repeated (chronic) exposure to MPD at doses of 0.6, 2.5, and 10.0 mg/kg produced divergent behavioral outcomes: some animals developed tolerance, others exhibited sensitization, and all displayed withdrawal behaviors. These findings suggest that MPD induces behavioral expressions consistent with criteria for substance use disorder and drug dependence—important considerations for clinicians prescribing MPD. Although neurons in the locus coeruleus (LC), dorsal raphe (DR), and ventral tegmental area (VTA) of both adolescent and adult animals responded similarly to acute MPD exposure, their responses diverged following chronic treatment. Chronic exposure at 0.6, 2.5, and 10.0 mg/kg produced age-dependent differences in both behavioral outcomes and neuronal activity patterns, particularly in the ratio of neurons showing excitation versus inhibition in response to MPD. These results underscore the importance of simultaneously correlating neuronal recordings from multiple brain regions with behavioral observations to fully characterize MPD’s effects. In summary, adolescents and adults display distinct different neurophysiological profiles following chronic MPD exposure—differences that clinicians should carefully consider in therapeutic contexts.
Acknowledgements
This study was supported by the National Institute of Health (NIH) grant DA R01 027222.
The authors thank S.M. Gordon and J. Concha for reviewing the manuscript and helping to prepare the figures.
References
2. Arnsten AF. Stimulants: Therapeutic actions in ADHD. Neuropsychopharmacology. 2006 Nov;31(11):2376–83.
3. Bogle KE, Smith BH. Illicit methylphenidate use: a review of prevalence, availability, pharmacology, and consequences. Curr Drug Abuse Rev. 2009 May;2(2):157–76.
4. Challman TD, Lipsky JJ. Methylphenidate: its pharmacology and uses. Mayo Clin Proc. 2000 Jul;75(7):711–21.
5. Jaeschke RR, Sujkowska E, Sowa-Kućma M. Methylphenidate for attention-deficit/hyperactivity disorder in adults: a narrative review. Psychopharmacology (Berl). 2021 Oct;238(10):2667–91.
6. Sroubek A, Kelly M, Li X. Inattentiveness in attention-deficit/hyperactivity disorder. Neurosci Bull. 2013 Feb;29(1):103–10.
7. Storebø OJ, Storm MRO, Pereira Ribeiro J, Skoog M, Groth C, Callesen HE, et al. Methylphenidate for children and adolescents with attention deficit hyperactivity disorder (ADHD). Cochrane Database Syst Rev. 2023 Mar 27;3(3):CD009885.
8. Kapur A. Is Methylphenidate Beneficial and Safe in Pharmacological Cognitive Enhancement? CNS Drugs. 2020 Oct;34(10):1045–62.
9. Newcorn JH. A glimpse into key issues in ADHD. CNS Spectr. 2000 Jun;5(6):25.
10. Dafny N, Yang PB. The role of age, genotype, sex, and route of acute and chronic administration of methylphenidate: a review of its locomotor effects. Brain Res Bull. 2006 Feb 15;68(6):393–405.
11. National Institute on Drug Abuse. Update, 2018 and 2019.
12. Arria AM, Caldeira KM, O'Grady KE, Vincent KB, Johnson EP, Wish ED. Nonmedical use of prescription stimulants among college students: associations with attention-deficit-hyperactivity disorder and polydrug use. Pharmacotherapy. 2008 Feb;28(2):156–69.
13. Faraone SV, Rostain AL, Montano CB, Mason O, Antshel KM, Newcorn JH. Systematic Review: Nonmedical Use of Prescription Stimulants: Risk Factors, Outcomes, and Risk Reduction Strategies. J Am Acad Child Adolesc Psychiatry. 2020 Jan;59(1):100–12.
14. Greely H, Sahakian B, Harris J, Kessler RC, Gazzaniga M, Campbell P, et al. Towards responsible use of cognitive-enhancing drugs by the healthy. Nature. 2008 Dec 11;456(7223):702–5.
15. Teter CJ, McCabe SE, Cranford JA, Boyd CJ, Guthrie SK. Prevalence and motives for illicit use of prescription stimulants in an undergraduate student sample. J Am Coll Health. 2005 May-Jun;53(6):253–62.
16. Cavaco AM, Ribeiro J, Nørgaard LS. Exploring the use of cognitive enhancement substances among Portuguese university students. Explor Res Clin Soc Pharm. 2021 Dec 14; 5:100097.
17. Gray JD, Punsoni M, Tabori NE, Melton JT, Fanslow V, Ward MJ, et al. Methylphenidate administration to juvenile rats alters brain areas involved in cognition, motivated behaviors, appetite, and stress. J Neurosci. 2007 Jul 4;27(27):7196–207.
18. Klassen A, Miller A, Raina P, Lee SK, Olsen L. Attention-deficit hyperactivity disorder in children and youth: a quantitative systematic review of the efficacy of different management strategies. Can J Psychiatry. 1999 Dec;44(10):1007–16.
19. Malík M, Tlustoš P. Nootropics as Cognitive Enhancers: Types, Dosage and Side Effects of Smart Drugs. Nutrients. 2022 Aug 17;14(16):3367.
20. McCabe SE, Knight JR, Teter CJ, Wechsler H. Non-medical use of prescription stimulants among US college students: prevalence and correlates from a national survey. Addiction. 2005 Jan;100(1):96–106.
21. Salman T, Afroz R, Nawaz S, Mahmood K, Haleem DJ, Zarina S. Differential effects of memory enhancing and impairing doses of methylphenidate on serotonin metabolism and 5-HT1A, GABA, glutamate receptor expression in the rat prefrontal cortex. Biochimie. 2021 Dec;191:51–61.
22. Thevapalan L, Stasiak M, Harris L, Pandit AS. Smart drug among surgeons: Future cause for concern? Brit J Surgery. 2023;139.
23. Kim MG, Kim J, Kim SC, Jeong J. Twitter analysis of the nonmedical use and side effects of methylphenidate: machine learning study. J Med Internet Res. 2020 Feb 24;22(2):e16466.
24. Lakhan SE, Kirchgessner A. Prescription stimulants in individuals with and without attention deficit hyperactivity disorder: misuse, cognitive impact, and adverse effects. Brain Behav. 2012 Sep;2(5):661–77.
25. O’Brien CP. Drug addiction and drug abuse. Goodman and Gilman’s the Pharmacological Basis of Therapeutics. 2006;11:607–27.
26. Volkow ND, Wang G, Fowler JS, Logan J, Gerasimov M, Maynard L, et al. Therapeutic doses of oral methylphenidate significantly increase extracellular dopamine in the human brain. J Neurosci. 2001 Jan 15;21(2):RC121.
27. Bolaños CA, Barrot M, Berton O, Wallace-Black D, Nestler EJ. Methylphenidate treatment during pre- and periadolescence alters behavioral responses to emotional stimuli at adulthood. Biol Psychiatry. 2003 Dec 15;54(12):1317–29.
28. Ruocco LA, Carnevale UA, Treno C, Sadile AG, Melisi D, Arra C, et al. Prepuberal subchronic methylphenidate and atomoxetine induce different long-term effects on adult behaviour and forebrain dopamine, norepinephrine and serotonin in Naples high-excitability rats. Behav Brain Res. 2010 Jun 26;210(1):99–106.
29. Schachter HM, Pham B, King J, Langford S, Moher D. How efficacious and safe is short-acting methylphenidate for the treatment of attention-deficit disorder in children and adolescents? A meta-analysis. CMAJ. 2001 Nov 27;165(11):1475–88.
30. Storebø OJ, Ramstad E, Krogh HB, Nilausen TD, Skoog M, Holmskov M, et al. Methylphenidate for children and adolescents with attention deficit hyperactivity disorder (ADHD). Cochrane Database Syst Rev. 2015 Nov 25;2015(11):CD009885.
31. Vertessen K, Luman M, Swanson JM, Bottelier M, Stoffelsen R, Bet P, et al. Methylphenidate dose-response in children with ADHD: evidence from a double-blind, randomized placebo-controlled titration trial. Eur Child Adolesc Psychiatry. 2024 Feb;33(2):495–504.
32. Alvarez BD, Morales CA, Amodeo DA. Impact of specific serotonin receptor modulation on behavioral flexibility. Pharmacol Biochem Behav. 2021 Oct;209:173243.
33. Barnes NM, Sharp T. A review of central 5-HT receptors and their function. Neuropharmacology. 1999 Aug;38(8):1083–152.
34. Berridge CW, Waterhouse BD. The locus coeruleus-noradrenergic system: modulation of behavioral state and state-dependent cognitive processes. Brain Res Brain Res Rev. 2003 Apr;42(1):33–84.
35. Borycz J, Zapata A, Quiroz C, Volkow ND, Ferré S. 5-HT 1B receptor-mediated serotoninergic modulation of methylphenidate-induced locomotor activation in rats. Neuropsychopharmacology. 2008 Feb;33(3):619–26.
36. Dafny N, Reyes-Vasquez C, Liu Y. The Serotonergic Signaling and the Dorsal Raphe (DR) Neurons in Adolescent Rats are the Most Engaging in Response to Acute and Chronic Methylphenidate as Compared to Other Neuronal Activities Recorded from Other Five Brain Areas. J. Clin. Pharmacol. Ther. 2022; 3:1026.
37. Dahl MJ, Mather M, Werkle-Bergner M. Noradrenergic modulation of rhythmic neural activity shapes selective attention. Trends Cogn Sci. 2022 Jan;26(1):38–52.
38. Daniali S, Madjd Z, Shahbazi A, Niknazar S, Shahbazzadeh D. Chronic Ritalin administration during adulthood increases serotonin pool in rat medial frontal cortex. Iran Biomed J. 2013;17(3):134–9.
39. Del Campo N, Chamberlain SR, Sahakian BJ, Robbins TW. The roles of dopamine and noradrenaline in the pathophysiology and treatment of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2011 Jun 15;69(12):e145–57.
40. Gottlieb S. Methylphenidate works by increasing dopamine levels. BMJ. 2001 Feb 3;322(7281):259.
41. Koda K, Ago Y, Cong Y, Kita Y, Takuma K, Matsuda T. Effects of acute and chronic administration of atomoxetine and methylphenidate on extracellular levels of noradrenaline, dopamine and serotonin in the prefrontal cortex and striatum of mice. J Neurochem. 2010 Jul;114(1):259–70.
42. Kuczenski R, Segal DS. Effects of methylphenidate on extracellular dopamine, serotonin, and norepinephrine: comparison with amphetamine. J Neurochem. 1997 May;68(5):2032–7.
43. Oades RD. Role of the serotonin system in ADHD: treatment implications. Expert Rev Neurother. 2007 Oct;7(10):1357–74.
44. Oades RD. Dopamine-serotonin interactions in attention-deficit hyperactivity disorder (ADHD). Prog Brain Res. 2008;172:543–65.
45. Volkow ND, Gatley SJ, Fowler JS, Wang GJ, Swanson J. Serotonin and the therapeutic effects of ritalin. Science. 2000 Apr 7;288(5463):11.
46. Kuczenski R, Segal DS. Exposure of adolescent rats to oral methylphenidate: preferential effects on extracellular norepinephrine and absence of sensitization and cross-sensitization to methamphetamine. J Neurosci. 2002 Aug 15;22(16):7264–71.
47. Dafny N, Claussen C, Frazier E, Lin Y. Neurophysiological evidence that the: ventral tegmental area, locus coeruleus, dorsal raphe, nucleus accumbens, caudate nucleus, and prefrontal cortex neuronal populations each participate differently to regulate the response to psychostimulant methylphenidate. Neuronal and behavioral recording from freely behaving adult male rats. Int J Mol Sci. 2024;25:1–29.
48. Dafny N, Claussen C, Frazier E, Liu Y. Differential Roles of Key Brain Regions: Ventral Tegmental Area, Locus Coeruleus, Dorsal Raphe, Nucleus Accumbens, Caudate Nucleus, and Prefrontal Cortex in Regulating Response to Methylphenidate: Insights from Neuronal and Behavioral Studies in Freely Behaving Rats. Int J Mol Sci. 2024 May 29;25(11):5938.
49. Medina AC, Kabani A, Reyes-Vasquez C, Dafny N. Age differences to methylphenidate-NAc neuronal and behavioral recordings from freely behaving animals. J Neural Transm (Vienna). 2022 Aug;129(8):1061–76.
50. Tang B, Dafny N. Locus coeruleus neuronal and behavioral activity following acute and chronic methylphenidate. Brain Sci. 2015;1(1):24–42.
51. Sherwood, Nancy M, and Paola S Timiras. A Stereotaxic Atlas of the Developing Rat Brain [by] Nancy M. Sherwood and Paola S. Timiras. Berkeley: University of California Press; 1970.
52. Paxinos G, Watson C. The rat brain in stereotaxic coordinates: hard cover edition. Elsevier; 2006 Nov 2.
53. Broussard E, Reyes-Vazquez C, Dafny N. Methylphenidate dose-response behavioral and neurophysiological study of the ventral tegmental area and nucleus accumbens in adolescent rats. Eur J Neurosci. 2019 Aug;50(4):2635–52.
54. Gaytan O, Ghelani D, Martin S, Swann A, Dafny N. Dose response characteristics of methylphenidate on different indices of rats' locomotor activity at the beginning of the dark cycle. Brain Res. 1996 Jul 15;727(1-2):13–21.
55. Yang PB, Swann AC, Dafny N. Chronic administration of methylphenidate produces neurophysiological and behavioral sensitization. Brain Res. 2007 May 11;1145:66–80.
56. Floren S, King N, Carrasco A, Dafny N. Glutamate and dopamine in the VTA participate differently in the acute and chronic effect of methylphenidate. Behav Brain Res. 2020 Feb 17;380:112390.
57. Tang B, Dafny N. Dorsal raphe neuronal activities are modulated by methylphenidate. J Neural Transm (Vienna). 2013 May;120(5):721–31.
58. Kharas N, Whitt H, Reyes-Vasquez C, Dafny N. Methylphenidate modulates dorsal raphe neuronal activity: Behavioral and neuronal recordings from adolescent rats. Brain Res Bull. 2017 Jan;128:48–57.
59. Dafny N, Claussen C, Frazier E, Liu Y. Differential Roles of Key Brain Regions: Ventral Tegmental Area, Locus Coeruleus, Dorsal Raphe, Nucleus Accumbens, Caudate Nucleus, and Prefrontal Cortex in Regulating Response to Methylphenidate: Insights from Neuronal and Behavioral Studies in Freely Behaving Rats. Int J Mol Sci. 2024 May 29;25(11):5938.
60. Medina AC, Reyes-Vasquez C, Kharas N, Dafny N. Adolescent rats respond differently to methylphenidate as compared to adult rats- concomitant VTA neuronal and behavioral Recordings. Brain Res Bull. 2022 Jun 1;183:1–12.
61. Spencer TJ, Brown A, Seidman LJ, Valera EM, Makris N, Lomedico A, et al. Effect of psychostimulants on brain structure and function in ADHD: a qualitative literature review of magnetic resonance imaging-based neuroimaging studies. J Clin Psychiatry. 2013 Sep;74(9):902–17.
62. Thapar A, Cooper M, Eyre O, Langley K. What have we learnt about the causes of ADHD?. J Child Psychol Psychiatry. 2013 Jan;54(1):3–16.
63. Drouin C, Page M, Waterhouse B. Methylphenidate enhances noradrenergic transmission and suppresses mid- and long-latency sensory responses in the primary somatosensory cortex of awake rats. J Neurophysiol. 2006 Aug;96(2):622–32.
64. Hannestad J, Gallezot JD, Planeta-Wilson B, Lin SF, Williams WA, van Dyck CH, et al. Clinically relevant doses of methylphenidate significantly occupy norepinephrine transporters in humans in vivo. Biol Psychiatry. 2010 Nov 1;68(9):854–60.
65. Cook EH Jr, Stein MA, Ellison T, Unis AS, Leventhal BL. Attention deficit hyperactivity disorder and whole-blood serotonin levels: effects of comorbidity. Psychiatry Res. 1995 Jun 29;57(1):13–20.
66. Kirby LG, Zeeb FD, Winstanley CA. Contributions of serotonin in addiction vulnerability. Neuropharmacology. 2011 Sep;61(3):421–32.
67. Segal DS, Kuczenski R. Escalating dose-binge treatment with methylphenidate: role of serotonin in the emergent behavioral profile. J Pharmacol Exp Ther. 1999 Oct;291(1):19–30.
68. Di Miceli M, Omoloye A, Gronier B. Characterisation of methylphenidate-induced excitation in midbrain dopamine neurons, an electrophysiological study in the rat brain. Prog Neuropsychopharmacol Biol Psychiatry. 2022 Jan 10;112:110406.
69. Esposito E, Di Matteo V, Di Giovanni G. Serotonin-dopamine interaction: an overview. Prog Brain Res. 2008; 172:3–6.
70. Aston-Jones G, Waterhouse B. Locus coeruleus: From global projection system to adaptive regulation of behavior. Brain Res. 2016 Aug 15; 1645:75–8.
71. Poe GR, Foote S, Eschenko O, Johansen JP, Bouret S, Aston-Jones G, et al. Locus coeruleus: a new look at the blue spot. Nat Rev Neurosci. 2020 Nov;21(11):644–59.
72. Allers KA, Sharp T. Neurochemical and anatomical identification of fast- and slow-firing neurones in the rat dorsal raphe nucleus using juxtacellular labelling methods in vivo. Neuroscience. 2003;122(1):193–204.
73. Heidenreich BA, Basse-Tomusk AE, Rebec GV. Serotonergic dorsal raphe neurons: subsensitivity to amphetamine with long-term treatment. Neuropharmacology. 1987 Jul;26(7A):719–24.
74. Nakamura K, Matsumoto M, Hikosaka O. Reward-dependent modulation of neuronal activity in the primate dorsal raphe nucleus. J Neurosci. 2008 May 14;28(20):5331–43.
75. Jones Z, Vazquez CR, Dafny N. Ventral tegmental area neuronal activity correlates to animals’ behavioral response to chronic methylphenidate recorded from adolescent SD male rats. Journal of Behavioral and Brain Science. 2014;4(04):168–89.
76. Chang JC, Lin HY, Gau SS. Age-dependent effects of cumulative methylphenidate exposure on brain structure and symptom amelioration in youth with ADHD: A longitudinal MRI study. Prog Neuropsychopharmacology Biol Psychiatry. 2025 Jul 13; 140:111429.
77. Ding YS, Fowler JS, Volkow ND, Dewey SL, Wang GJ, Logan J, et al. Chiral drugs: comparison of the pharmacokinetics of [11C]d-threo and L-threo-methylphenidate in the human and baboon brain. Psychopharmacology (Berl). 1997 May;131(1):71–8.
78. Stevens T, Sangkuhl K, Brown JT, Altman RB, Klein TE. PharmGKB summary: methylphenidate pathway, pharmacokinetics/pharmacodynamics. Pharmacogenet Genomics. 2019 Aug;29(6):136–54.
79. Sharma A, Couture J. A review of the pathophysiology, etiology, and treatment of attention-deficit hyperactivity disorder (ADHD). Ann Pharmacother. 2014 Feb;48(2):209–25.
80. Kalivas PW, Duffy P, DuMars LA, Skinner C. Behavioral and neurochemical effects of acute and daily cocaine administration in rats. J Pharmacol Exp Ther. 1988 May;245(2):485–92.
81. Laakso A, Mohn AR, Gainetdinov RR, Caron MG. Experimental genetic approaches to addiction. Neuron. 2002 Oct 10;36(2):213–28.
82. Robinson TE, Berridge KC. The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Brain Res Rev. 1993 Sep-Dec;18(3):247–91.
83. Robinson TE. Behavioral sensitization: characterization of enduring changes in rotational behavior produced by intermittent injections of amphetamine in male and female rats. Psychopharmacology (Berl). 1984;84(4):466–75.
84. Robinson TE. Persistent sensitizing effects of drugs on brain dopamine systems and behavior: implications for addiction and relapse. Biological Basis of Substance Abuse. 1993:373–402.
85. Wolf ME. The role of excitatory amino acids in behavioral sensitization to psychomotor stimulants. Prog Neurobiol. 1998 Apr;54(6):679–720.
86. Berridge KC, Robinson TE. The mind of an addicted brain: neural sensitization of wanting versus liking. Current Directions in Psychological Science. 1995 Jun;4(3):71–5.
87. Brandon CL, Marinelli M, Baker LK, White FJ. Enhanced reactivity and vulnerability to cocaine following methylphenidate treatment in adolescent rats. Neuropsychopharmacology. 2001 Nov;25(5):651–61.
88. Andersson M, Westin JE, Cenci MA. Time course of striatal DeltaFosB-like immunoreactivity and prodynorphin mRNA levels after discontinuation of chronic dopaminomimetic treatment 1. Eur J Neurosci 17:661–6.2003.
89. Kuczenski R, Segal DS. Sensitization of amphetamine-induced stereotyped behaviors during the acute response. J Pharmacol Exp Ther. 1999 Feb;288(2):699–709.
90. Kuczenski R, Segal DS. Locomotor effects of acute and repeated threshold doses of amphetamine and methylphenidate: relative roles of dopamine and norepinephrine. J Pharmacol Exp Ther. 2001 Mar;296(3):876–83.