Abstract
Morphine influences multiple brain nuclei that regulate key physiological systems, including the endocrine and immune systems, as well as behavioral expression. Opioid consumption has been shown to suppress both the hypothalamic-pituitary-adrenal (HPA) axis and immune function—systems that play a critical role in the development of opioid tolerance and the manifestation of withdrawal symptoms. Evidence from animal studies indicates that immune suppression can prevent naloxone-precipitated behavioral withdrawal in opioid-dependent subjects, suggesting that both dependence and withdrawal are mediated, at least in part, by neuroendocrine and immune mechanisms. Early research proposed that the central nervous system (CNS) modulates immune responses, aligning with the anti-inflammatory properties of opioids. In experiments where the immune system was ablated via irradiation, chronic morphine exposure failed to induce withdrawal symptoms upon naloxone administration. Conversely, restoring immune function through donor cell transplantation reinstated typical withdrawal behaviors. Notably, a single administration of cortisol—an immunomodulatory hormone—prior to chronic morphine exposure or in morphine-dependent animals significantly reduced the severity of behavioral withdrawal symptoms. Collectively, these findings highlight a bidirectional interaction between the nervous and immune systems in regulating opioid dependence and withdrawal, pointing to potential therapeutic strategies for mitigating addiction and relapse.
Keywords
Opiate, Addiction, Withdrawal, Treatment, Cortisol
Introduction
Opioid addiction is recognized as a chronic brain disease. The death due to opioid overdose in the USA since 1980 increases exponentially at 76% per year, reaching 100,000 per year [1]. The opioid epidemic began in the 1990s with over-prescription of opioids as pain relieving medications swiftly became the most prescribed medication in the country. About 20 to 30 percent of patients who are prescribed opioids for chronic pain misuse them [2]. More than four million Americans used opiates for nonmedical purposes, and it increase every year [3,4]. There are more opioid overdose deaths in the US every year than deaths due to car accidents and gun shots [5]. Misuse of opioids affects millions of Americans. Overdose deaths related to morphine and its analogs increased from year to year [4]. Sadly, the treatments that exist, including medications in the case of opioids, help only a fraction of those who could benefit from them. The need for new treatments is real and urgent. The primary drugs currently used for the detoxification of opioid addiction are themselves opioid agonists, such as methadone, or partial agonists like buprenorphine. However, these drugs may simply substitute one opioid for another, potentially prolonging a type of "dependence." Additionally, patients treated with these drugs face an increased risk of relapses and subsequent overdoses. Additionally, most of these drugs are used primarily for managing opiate withdrawal symptoms and do not adequately address the cravings that often lead to relapse. Consequently, there is concern that these treatments merely substitute one opioid for another, perpetuating a state of dependency. Patient compliance with the available long-term treatment regimens is often poor, limiting the overall effectiveness of these therapies. Furthermore, patients undergoing these treatments are at an increased risk of relapse and subsequent overdoses [6]. These limitations highlight the urgent need for additional and more effective remedies for opioid dependence. Additionally, most of available drugs are used primarily for managing opiate withdrawal symptoms and do not adequately address the cravings that often lead to relapse. Consequently, there is concern that these treatments merely substitute one opioid for another, perpetuating a state of dependency. Patient compliance with long-term treatment regimens is often poor, limiting the overall effectiveness of these therapies. Furthermore, patients undergoing these treatments are at an increased risk of relapse and subsequent overdoses [6]. These limitations highlight the urgent need for additional and more effective remedies for opioid dependence.
Recent animal studies highlight the potential of immunotherapy as a novel approach to treating opioid addiction [7–12]. Two primary immunotherapeutic strategies have emerged: 1. Passive immunization, involving monoclonal antibody administration [10,11] and 2. Active immunization, using vaccine-based interventions [12]. Both approaches have shown promise in promoting abstinence from substances such as cocaine, nicotine, methamphetamine, and heroin [10–12]. However, concerns remain regarding opiate overdose risks associated with these treatments [12]. Immunotherapy represents a promising adjunct to existing treatment modalities for opioid addiction. Building on this rationale, experimental studies have tested whether cortisol administration—before or after repeated morphine exposure—can prevent or attenuate the severity of opioid withdrawal symptoms using six behavioral expressions involved in the expression of tolerance and behavioral withdrawal [13,14]. These findings highlight the potential role of immunomodulation in mitigating opioid dependence and withdrawal, paving the way for future research into non-opioid therapeutic interventions.
Methods
Induction of morphine dependence and tolerance in rats
Several experimental procedures are used to induce physical dependence on morphine. 1. Repeated daily morphine administration: Rats received single dose daily intraperitoneal (i.p) injections of morphine (10 or 20 mg/kg) for 7 to 21 days—testing for expressing tolerance and/or withdrawal behavior. 2. Incremental “staircase” morphine dosing schedule: Morphine was administered three times daily (morning, noon, and evening) in progressively increasing doses over five consecutive days: Day 1: 10, 10, and 20 mg/kg i.p morphine. Day 2: 20, 20, and 30 mg/kg i.p morphine. Day 3: 30, 30, and 40 mg/kg i.p morphine. Day 4: 40, 40, and 50 mg/kg i.p morphine. Day 5: testing for the expression of tolerance and/or withdrawal behavior. 3. Morphine Pellet Implantation: A 75–100 mg morphine pellet was implanted subcutaneously for 72 hours, followed by tolerance or withdrawal or assessment (Figure 1). These procedures have been previously validated in our laboratory [15–21]. In the present report, implanting morphine pellet was used to elicit morphine dependent animal models and naloxone (1mg/kg i.p) was used to elicit behavioral withdrawal symptoms to shorten the experimental days (Figure 2).
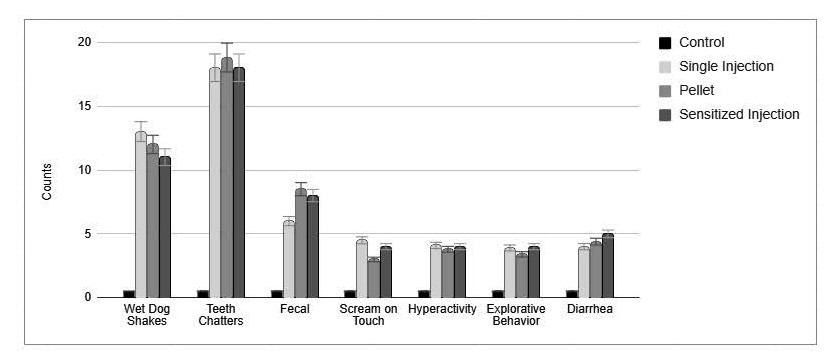
Figure 1. The figure shows the result of three experimental protocols used to elicit morphine dependence followed by Naloxone injection and the seven behavioral withdrawal expressions. The first column (N=8) is the result of control animals treated daily for seven days with saline and on day eight with Naloxone 1.0 mg/kg i.p. The second column (N=8) is the result of 20.0 mg/kg morphine injection i.p given daily for seven days and on experimental day 8 treated with Naloxone 1.0 mg/kg i.p. The third column (N=8) is the result of the five-morphine escalating /day (morning, noon and evening) as follow: Day 1 – 10, 10, and 20 mg/kg morphine i.p. Day 2 – 20, 20, and 30 mg/kg morphine i.p. Day 3 – 30, 30, and 40 mg/kg morphine i.p. Day 4 – 40, 40, and 50 mg/kg morphine i.p. Day 5 – 50 mg/kg morphine i.p followed two hrs. later with Naloxone 1.0 mg/kg i.p. In the fourth column (N=8) the rats were implanted under light anesthesia with morphine pellet of 75 mg/pellet, 72hrs later the residual pellet was removed and two hrs. later Naloxone 1.0mg/kg was injected and the seven behavioral withdrawal expressions were measured respectfully by two research associates [13–15,18].
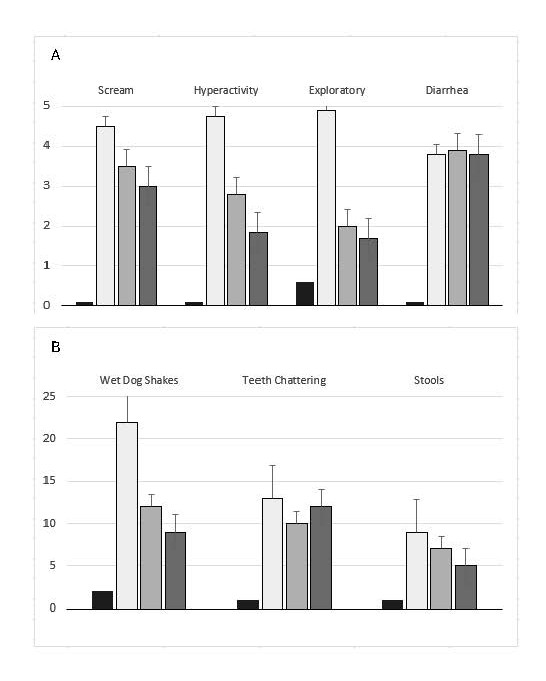
Figure 2. Summary of seven behavioral expressions before and after treatment with morphine pellet and after cortisol treatment. The upper four behavioral expressions (A) were rated on a scale from 1 (low activity) to 5 (high activity). The lower three behavioral expressions (B) represent the frequency of behaviors observed during a 10-minute period following naloxone administration (1.0 mg/kg, i.p). Each group consisted of N = 8 animals. Group 1 (Control): Received a placebo pellet. After 72 hours, animals were administered naloxone (1.0 mg/kg, i.p). Group 2 (Morphine Control): Implanted with a morphine pellet (75 mg/kg, s.c). After 72 hours, the residual morphine pellet was removed, and naloxone (1.0 mg/kg, i.p) was administered to induce withdrawal. Naloxone precipitated a significant increase (p<0.001) in all seven withdrawal-related behavioral expressions compared to the control group, indicating robust withdrawal symptoms. Group 3 (Cortisol Pre-Morphine Treatment): Treated with cortisol (2.0 mg/kg, i.p), followed one hour later by morphine pellet implantation (75 mg/kg, s.c). After 72 hours, the residual morphine pellet was removed, and naloxone (1.0 mg/kg, i.p) was administered. Group 4 (Cortisol Post-Morphine Treatment): Implanted with a morphine pellet (75 mg/kg, s.c). After 72 hours, the residual morphine pellet was removed, followed by cortisol administration (2.0 mg/kg, i.p). One hour later, animals were challenged with naloxone (1.0 mg/kg, i.p). In both cortisol-treated groups (Groups 3 and 4), all seven behavioral withdrawal measures were significantly attenuated (p<0.001) compared to the morphine control group. These results suggest that cortisol, whether administered before or after chronic morphine exposure, significantly reduces the intensity of naloxone-precipitated withdrawal behaviors.
Animals and experimental design
Four groups of male Sprague Dawley rats (N = 8 per group), each weighing approximately 200 g, were randomly assigned to one of four experimental groups: Group 1 – Control: Implanted with a placebo pellet. After 72 hours, the pellet was removed and rats received 1.0 mg/kg naloxone and behavioral assessment of withdrawal were performed. No withdrawal behaviors were observed (Figure 2). Group 2 – Morphine Control: Implanted with a morphine pellet for 72 hours. Following pellet removal, saline was administered and two hours later, 1.0 mg/kg naloxone was injected, and behavioral assessment of withdrawal were performed (Figure 2). Group 3 – Pre-Treatment Cortisol: Administered 2.0 mg/kg cortisol two hours prior to morphine pellet implantation. After 72 hours, the pellet was removed, and two hours later, 1.0 mg/kg naloxone was injected, and behavioral assessment of withdrawal were performed (Figure 2). Group 4 – Post-Treatment Cortisol: Implanted with a morphine pellet for 72 hours. After pellet removal, 2.0 mg/kg cortisol was administered, followed by 1.0 mg/kg naloxone and two hours later, behavioral assessment of withdrawal was performed (Figure 2). This design enabled evaluation of cortisol’s modulatory effects on preventing and treating morphine withdrawal symptoms. Seven behavioral withdrawal signs were assessed by two independent observers and scored from 0 (no difference from control) to 5 (maximum intensity). These behaviors, shown in Figure 2A, included the following behavioral expression 1. Scream to touch, 2. Hyperactivity, 3. Exploratory behavior, and 4. Diarrhea. Figure 2B presents three additional behavioral expressions: 5. Wet dog shakes, 6. Teeth chattering, and 7. Number of stools within 10 minutes post-naloxone administration respectfully. These assessments followed established protocols [13,14].
Result
Key findings
Group 1 (Placebo): No withdrawal behaviors were observed following naloxone administration. Group 2 (Morphine + Naloxone): Significant increases (p<0.001) in all seven withdrawal behaviors expression were observed, indicating morphine dependence. Group 3 (Pre-Treatment Cortisol): Significant attenuation of withdrawal behaviors, particularly scream to touch, hyperactivity, and exploratory activity, was observed following naloxone administration. Group 4 (Post-Treatment Cortisol): All seven withdrawal behaviors were significantly (p<0.001) reduced following cortisol treatment (2.0 mg/kg), demonstrating robust attenuation of morphine severity withdrawal symptoms. The results confirm that morphine induces physical dependence, as evidenced by naloxone-precipitated withdrawal behaviors, and that cortisol administered either before or after morphine exposure—significantly (p<0.001) reduces the severity of these symptoms.
Conclusion
This preclinical study demonstrates that a single dose of 2.0mg/kg cortisol, administered either prior to or after chronic morphine exposure, significantly attenuates behavioral withdrawal symptoms. The findings demonstrate that cortisol has therapeutic potential in preventing as well managing morphine dependence, relapse and toxification. Similar outcomes using cortisol were observed in other morphine-dependent animal models using escalating morphine dosing schedules over five days [13,14,19,22,23]. The mechanistic interpretation of these findings is discussed in the following section.
Discussion
Cortisol and drug abuse
Opioid dependence is an altered physiological state induced by repeated drug exposure, requiring continued use to prevent severe behavioral withdrawal symptoms. Withdrawal represents a stress response to drug cessation and activates both the hypothalamic–pituitary–adrenal (HPA) axis and the immune system [20,24,25]. This response reflects a rebound effect in which systems chronically suppressed by opioids become excessively activated upon withdrawal [26,27]. Rats experiencing withdrawal display increased neuronal activity across multiple brain regions, hyperlocomotion, body shakes (wet-dog shakes), teeth chattering, and diarrhea [13–16,24,28,29]. The severity of physical dependence correlates with withdrawal intensity, and modulating these symptoms may advance our understanding of opioid dependence mechanisms.
We propose that addiction induces a chronic stress state that engages the sympathetic-adrenomedullary system and the HPA axis [30]. Treating opioid-dependent subjects with the immunomodulator cortisol may attenuate behavioral withdrawal. Cortisol, synthesized in the adrenal cortex, is transported throughout the body and regulates a wide range of physiological processes [31]. Nearly all cells contain cortisol receptors, giving cortisol diverse effects depending on target tissue [32]. These include regulating metabolism and glycemic levels, modulating inflammation, influencing memory and cognition, maintaining blood pressure and electrolyte balance, and supporting fetal development [33–37]. Cortisol plays a dual role in immune function: it enhances certain immune cell activities but can also promote pro-inflammatory cytokine production [38,39]. It is critical for maintaining normal immunity and may protect the body from adverse consequences of drug dependence [40]. Repetitive opioid use disrupts the HPA axis, leading to cortisol deficiency associated with increased dependency and heightened relapse risk following drug cessation [41]. Opioids also produce broad endocrine effects and may induce adrenal insufficiency via chronic HPA suppression [42–44]. In addition, opioids alter the brain’s motivational circuitry, contributing to addiction vulnerability [41].
Evidence that the immune system (IS) participates in opiate addiction
The immune system is a highly intricate and dynamic network responsible for defending the body against a diverse array of threats [45–47]. The innate immune response serves as the first line of defense, providing immediate but nonspecific protection. Key components of this response include dendritic cells, macrophages, neutrophils, natural killer cells, and mast cells [45,48]. Dendritic cells function as intermediaries between the innate and adaptive immune responses by capturing and presenting antigens to T cells, thereby initiating adaptive immunity [49,50].
Critical early evidence for immune involvement in opioid effects came from the discovery of opioid receptors on immune cells [51,52] and dendrites [51–53]. Dendritic cells also produce cytokines, which act as biochemical messengers between the immune system and the brain [22]. Morphine influences neutrophil production and cytokine release, implicating cytokines in pain processing and suggesting their potential as targets for analgesic development [53].
Morphine activates central immune-signaling pathways that enhance mesolimbic–cortical dopamine, norepinephrine, and serotonin circuits involved in reward, tolerance, and withdrawal [8]. Chronic opioid use can impair immune competence and predispose individuals to health complications [54,55]. Hutchinson and Watkins [56] proposed that future addiction therapies should incorporate immunopharmacology, given the substantial contribution of CNS immune signaling to drug reward circuits [57].
Multiple studies demonstrate that an intact immune system is necessary for the expression of opioid withdrawal. Immune-ablated rats treated chronically with morphine do not show naloxone-precipitated withdrawal, whereas immune-reconstituted animals do [16,19,22,54,58]. Moreover, a single injection of cortisol in morphine-dependent rats significantly attenuates naloxone-induced withdrawal. These findings support a functional connection between the immune system and withdrawal expression and provide a rationale for cortisol-based immunotherapy in opioid dependence.
Rational to use cortisol (Cort)
Cortisol is an endogenous end product of the hypothalamic-pituitary-axis (HPA) produced by the adrenal glands. Cortisol is a steroid hormone, one of the glucocorticoids, whose release is stimulated by ACTH. It is made in the outer cortex of the adrenal glands and then released into the bloodstream and transported throughout the body. It is responsible for regulating a wide range of physiologic processes [31]. Almost every cell contains receptors for cortisol and thus cortisol has many different actions depending on the tissue it is acting upon [32]. These effects include controlling glycemic levels involved in regulating metabolism, acting as an anti-inflammatory, influencing memory function, controlling salt and water balance, influencing blood pressure, helping with the development of the fetus [33,36], controlling stress and influencing cognitive function [34,35,37]. Repetitive use of opiates induces effects on multiple levels of the endocrine system through mechanisms which are not fully elucidated [59]. Opiate use may induce endocrinopathies-adrenal insufficiency due to suppression of the HPA [42,44], as well as functions of the brain motivational system [41] that may hold clues to the nature of the motivational changes accompanying addiction and vulnerability to addiction. Opioid consumption exerts negative effects on the endocrine system. Opioid binds to receptors in the hypothalamus (and many other sites) that result in increase of prolactin as well alter the adrenal production of DHEA and opioid cession result in severe stress behavior [41].
Substance abuse causes stress-like responses [41]. Repetitive consumption of opiates dysregulates the hypothalamic-pituitary-axis (HPA) and elicits deficient cortisol activity that is associated with a risk for dependency and relapse after abrupt cessation of morphine [41]. Corticosteroids are nonspecific immunomodulators that modulate the immune system by affecting T-cells and B-cells and suppress cell-mediated and humoral-mediated immunity [60]. In addition, macrophages and monocytes appear to be greatly affected by cortisol. Activation of HPA arises from IL-1 stimulating hypothalamic corticotrophin releasing hormone (CRH) and subsequently activates the adrenocorticotrophic hormone (ACTH) and the adrenal cortex to release glucocorticoids. This increases plasma concentrations of glucocorticoids to modulate the immune system [40,61,62]. Corticosteroids are nonspecific immunomodulators because they affect macrophages and monocytes as well as T-cells and B-cells, and hence cell-mediated and humoral-mediated immune processes [60]. Cort treatment prior to repetitive morphine exposure and to morphine dependent rodent was able to reduce the naloxone-precipitated withdrawal syndrome [23]. Cortisol is anti-inflammatory acutely and pro-inflammatory in the long-term [57,63]. In humans, exogenous glucocorticoids are given to ameliorate inflammation and pain, which however, in the long term can cause immunosuppression. It is likely that administration of cortisol before repetitive morphine provides negative feedback to the HPA preventing further release of glucocorticoids and, subsequently, immune suppression. Morphine was reported to modulate the HPA axis [64] and cortisol administration may restore the HPA axis. Opioids activate the downstream pathway of the HPA [65,66] suggesting that glucocorticoids modulate the immune system. Thus, cortisol treatment may restore the HPA and reverse the effect of chronic opioid dependency and may explain why cortisol treatment attenuates the opiate withdrawal symptoms [23].
We postulate that addiction induces a chronic stress state, triggering sympathetic adrenomedullary and HPA systems [30]. Treating addicted individuals with the immunomodulator cortisol modulates the expression of behavioral withdrawal. Cortisol is synthesized in the adrenal cortex and transported through the bloodstream to regulate various physiological processes [31]. As nearly every cell contains cortisol receptors, its effects vary depending on the target tissue [32]. Cortisol regulates glycemic levels, metabolism, inflammation, memory function, salt and water balance, blood pressure, and cognitive function [33–37]. Cortisol plays a dual role in immune function, enhancing specific immune cell activity while promoting the production of pro-inflammatory cytokines [38,39,67]. It is essential for maintaining normal immunity and protecting the body against drug dependence [40]. Repetitive opioid use dysregulates the HPA axis, leading to cortisol deficiencies associated with increased dependency and relapse risk following abrupt drug cessation [41]. Opioid use also impacts multiple levels of the endocrine system through mechanisms not yet fully understood [59]. It may induce endocrinopathies such as adrenal insufficiency due to HPA suppression [42–44] and affect the brain's motivational system, providing insight into addiction vulnerability [41]. Corticosteroids, including cortisol, act as nonspecific immunomodulators, affecting T cells, B cells, macrophages, and monocytes, thereby suppressing cell-mediated and humoral immunity [60]. The HPA axis is activated through interleukin-1 (IL-1)-stimulated hypothalamic corticotropin-releasing hormone (CRH), which subsequently triggers ACTH release from the pituitary and glucocorticoid secretion from the adrenal cortex [40,61,62]. Studies have demonstrated that cortisol significantly (P<0.001) reduces naloxone-precipitated withdrawal symptoms [23]. Cortisol exhibits anti-inflammatory effects acutely but promotes inflammation over prolonged exposure [57,63]. Administering cortisol before repeated morphine use may provide negative feedback to the HPA, preventing excessive glucocorticoid release and subsequent immune modulation. Opioids influence the HPA axis downstream pathways [64], suggesting that glucocorticoids play a critical role in immune modulation [65,66]. Thus, cortisol treatment may restore HPA function, reverse chronic opioid dependency effects, and attenuate withdrawal symptoms.
Acknowledgement
The author would like to thank John Concha for figure production and the funding Supported by NIH NIDA RO1 grant #000803.
References
2. Vowles KE, McEntee ML, Julnes PS, Frohe T, Ney JP, van der Goes DN. Rates of opioid misuse, abuse, and addiction in chronic pain: a systematic review and data synthesis. Pain. 2015 Apr;156(4):569-576.
3. NIDA update 2018 and 2019.
4. NIDA note 2023 and 2025.
5. Hernandez JBR, Kim PY. Epidemiology Morbidity And Mortality. 2022 Oct 3. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–.
6. Fast Tract Action Committee. Oct 2018.
7. Breehl, N. The immune system: A new hope for opioid addiction?. United States: Labroots; 2018(Accessed 03/2022). Available from: https://www.labroots.com/trending/immunology/12679/immune-system-hope-opioid-addition
8. Coller JK, Hutchinson MR. Implications of central immune signaling caused by drugs of abuse: mechanisms, mediators and new therapeutic approaches for prediction and treatment of drug dependence. Pharmacol Ther. 2012 May;134(2):219–45.
9. Liang X, Liu R, Chen C, Ji F, Li T. Opioid System Modulates the Immune Function: A Review. Transl Perioper Pain Med. 2016;1(1):5–13.
10. Shen X, Kosten TR. Immunotherapy for drug abuse. CNS Neurol Disord Drug Targets. 2011 Dec;10(8):876–9.
11. Xu A, Kosten TR. Current status of immunotherapies for addiction. Ann N Y Acad Sci. 2021 Apr;1489(1):3-16.
12. Zalewska-Kaszubska J. Is immunotherapy an opportunity for effective treatment of drug addiction? Vaccine. 2015 Nov 27;33(48):6545-51.
13. Dafny N. Interferon modifies morphine withdrawal phenomena in rodents. Neuropharmacology. 1983a May;22(5):647–51.
14. Dafny N. Modification of morphine withdrawal by interferon. Life Sci. 1983b Jan 24;32(4):303–5.
15. Dafny N. Is interferon-α a neuromodulator?. Brain Res Brain Res Rev. 1998 Mar 1;26(1):1–5.
16. Dafny N. Immunotherapy as a treatment to confront the ongoing opioid epidemic- A review. J Cell Mol Immunol. 2022;1(1):20–27.
17. Dafny N, Lee JR, Dougherty PM. Immune response products alter CNS activity: interferon modulates central opioid functions. J Neurosci Res. 1988;19(1):130–9.
18. Dafny N, Dougherty PM, Pellis NR. The immune system and opiate withdrawal. Int J Immunopharmacol. 1989;11(4):371–5.
19. Dougherty PM, Dafny N. Cyclosporine affects central nervous system opioid activity via direct and indirect means. Brain Behav Immun. 1988 Sep;2(3):242–53.
20. Venkataraman S, Dafny N. Treatment to combat opiate dependence. Journal of Psychiatry Research Review & Report. 2020;2(1)1–11.
21. Wei E. Morphine analgesia, tolerance and physical dependence in the adrenalectomized rat. Br J Pharmacol. 1973 Apr;47(4):693–9.
22. Dougherty PM, Harper C, Dafny N. The effect of alpha-interferon, cyclosporine A, and radiation-induced immune suppression on morphine-induced hypothermia and tolerance. Life Sci. 1986 Dec 8;39(23):2191–7.
23. Montgomery SP, Dafny N. Cyclophosphamide and cortisol reduce the severity of morphine withdrawal. Int J Immunopharmacol. 1987;9(4):453–7.
24. Dafny N. Immunotherapy as a treatment to confront the ongoing opioid epidemic- A review. J Cell Mol Immunol. 2022;1(1):20–27.
25. Ihezie SA, Dafny N. Prevention of Opioid Addiction. Journal of Experimental Neurology. 2021 Dec 21;2(4):162–72.
26. Herz, A, and J Bläsig. “Behavioral Aspects of Precipitated Morphine Withdrawal in Rats.” Drug Addiction. New York: Stratton Intercontinental Medical Book Corp., 1974.
27. Jaffe JH. Drud addiction and drug abuse. In: Goodman LS, Gilman A, Editors. Pharmacological Basis of Therapeutics. Macmillan; 1975. pp. 284–324.
28. Bläsig J, Herz A, Reinhold K, Zieglgänsberger S. Development of physical dependence on morphine in respect to time and dosage and quantification of the precipitated withdrawal syndrome in rats. Psychopharmacologia. 1973 Oct 23;33(1):19–38.
29. Dafny N, Reyes-Vazquez C. Three different types of alpha-interferons alter naloxone-induced abstinence in morphine-addicted rats. Immunopharmacology. 1985 Feb;9(1):13–7.
30. King ME. The international encyclopedia of health communication. Hoboken, NJ: John Wiley and Sons, Inc; 2022. pp. 1–4.
31. Dafny N, Phillips MI, Taylor AN, Gilman S. Dose effects of cortisol on single unit activity in hypothalamus, reticular formation and hippocampus of freely behaving rats correlated with plasma steroid levels. Brain Res. 1973 Sep 14;59:257–72.
32. Andrews RC, Herlihy O, Livingstone DE, Andrew R, Walker BR. Abnormal cortisol metabolism and tissue sensitivity to cortisol in patients with glucose intolerance. J Clin Endocrinol Metab. 2002 Dec;87(12):5587–93.
33. Hellhammer DH, Wüst S, Kudielka BM. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology. 2009 Feb;34(2):163–171.
34. Joseph JJ, Wang X, Spanakis E, Seeman T, Wand G, Needham B, et al. Diurnal salivary cortisol, glycemia and insulin resistance: The multi-ethnic study of atherosclerosis. Psychoneuroendocrinology. 2015 Dec;62:327–35.
35. Kelly JJ, Mangos G, Williamson PM, Whitworth JA. Cortisol and hypertension. Clinical and Experimental Pharmacology and Physiology. 1998 Nov;25(S1):S51–6.
36. Kudielka BM, Hellhammer DH, Wüst S. Why do we respond so differently? Reviewing determinants of human salivary cortisol responses to challenge. Psychoneuroendocrinology. 2009 Jan;34(1):2–18.
37. Law R, Clow A. Stress, the cortisol awakening response and cognitive function. Int Rev Neurobiol. 2020;150:187–217.
38. Ince LM, Weber J, Scheiermann C. Control of Leukocyte Trafficking by Stress-Associated Hormones. Front Immunol. 2019 Jan 11;9:3143.
39. Reed RG, Raison CL. Stress and the immune system. InEnvironmental influences on the immune system 2016; Feb 4:pp. 97-126. Vienna: Springer Vienna.
40. Jefferies WM. Cortisol and immunity. Medical hypotheses. 1991 Mar 1;34(3):198–208.
41. Lovallo WR. Cortisol secretion patterns in addiction and addiction risk. Int J Psychophysiol. 2006 Mar;59(3):195–202.
42. Donegan D. Opioid induced adrenal insufficiency: what is new? Curr Opin Endocrinol Diabetes Obes. 2019 Jun;26(3):133–138.
43. Li T, Donegan D, Hooten WM, Bancos I. Clinical presentation and outcomes of opioid-induced adrenal insufficiency. Endocrine Practice. 2020 Nov 1;26(11):1291–7.
44. Saeed ZI, Bancos I, Donegan D. CURRENT KNOWLEDGE AND PRACTICES OF HEATH CARE PROFESSIONALS ON OPIOID-INDUCED ADRENAL INSUFFICIENCY. Endocr Pract. 2019 Oct;25(10):1012–21.
45. Alotiby A. Immunology of stress: A review article. J. Clin. Med. 2024 Oct 25;13(21):6394.
46. Re GF, Jia J, Xu Y, Zhang Z, Xie ZR, Kong D, et al. Dynamics and correlations in multiplex immune profiling reveal persistent immune inflammation in male drug users after withdrawal. Int Immunopharmacol. 2022 Jun;107:108696.
47. Mefford BM, Donaldson JC. Opioid and the Immune System. Pain Management 2000;45(3):HS–10.
48. Ben Achour S, Pascual O. Glia: the many ways to modulate synaptic plasticity. Neurochem Int. 2010 Nov;57(4):440–5.
49. Green JM, Sundman MH, Chou YH. Opioid-induced microglia reactivity modulates opioid reward, analgesia, and behavior. Neurosci Biobehav Rev. 2022 Apr;135:104544.
50. Steinman RM. Decisions about dendritic cells: past, present, and future. Annu Rev Immunol. 2012;30:1-22.
51. Watkins LR, Hutchinson MR, Johnston IN, Maier SF. Glia: novel counter-regulators of opioid analgesia. Trends Neurosci. 2005 Dec;28(12):661-9.
52. Zhang H, Largent-Milnes TM, Vanderah TW. Glial neuroimmune signaling in opioid reward. Brain Res Bull. 2020 Feb;155:102-111.
53. Clark JD, Shi X, Li X, Qiao Y, Liang D, Angst MS, et al. Morphine reduces local cytokine expression and neutrophil infiltration after incision. Mol Pain. 2007 Oct 2;3:28.
54. Pellis NR, Harper C, Dafny N. Suppression of the induction of delayed hypersensitivity in rats by repetitive morphine treatments. Exp Neurol. 1986 Jul;93(1):92–7.
55. Harricharan R, Abboussi O, Daniels WMU. Addiction: A dysregulation of satiety and inflammatory processes. Prog Brain Res. 2017;235:65–91.
56. Hutchinson MR, Watkins LR. Why is neuroimmunopharmacology crucial for the future of addiction research? Neuropharmacology. 2014 Jan;76 Pt B(0 0):218–27.
57. Hofford RS, Russo SJ, Kiraly DD. Neuroimmune mechanisms of psychostimulant and opioid use disorders. Eur J Neurosci. 2019 Aug;50(3):2562–2573.
58. Dafny N, Pellis NR. Evidence that opiate addiction is in part an immune response. Destruction of the immune system by irradiation-altered opiate withdrawal. Neuropharmacology. 1986 Aug;25(8):815–8.
59. Fountas A, Chai ST, Kourkouti C, Karavitaki N. Mechanisms of endocrinology: endocrinology of opioids. European J. of Endocrinology. 2018 Oct;179(4):R183–96.
60. Cupps TR, Fauci AS. Corticosteroid-mediated immunoregulation in man. Immunological reviews. 1982 Jan 1;65:133–55.
61. Al-Hashimi M, Scott SW, Thompson JP, Lambert DG. Opioids and immune modulation: more questions than answers. Br J Anaesth. 2013 Jul;111(1):80–8.
62. Eisenstein TK, Rahim RT, Feng P, Thingalaya NK, Meissler JJ. Effects of opioid tolerance and withdrawal on the immune system. J Neuroimmune Pharmacol. 2006 Sep;1(3):237–49.
63. Manetti L, Cavagnini F, Martino E, Ambrogio A. Effects of cocaine on the hypothalamic-pituitary-adrenal axis. J Endocrinol Invest. 2014 Aug;37(8):701-708.
64. Eisenstein TK. The Role of Opioid Receptors in Immune System Function. Front Immunol. 2019 Dec 20;10:2904.
65. Ninković J, Roy S. Role of the mu-opioid receptor in opioid modulation of immune function. Amino Acids. 2013 Jul;45(1):9–24.
66. Sacerdote P, Franchi S, Gerra G, Leccese V, Panerai AE, Somaini L. Buprenorphine and methadone maintenance treatment of heroin addicts preserves immune function. Brain Behav Immun. 2008 May;22(4):606–13.
67. Rogers TJ, Roy S. Editorial: The Role of Opioid Receptors in Immune System Function. Front Immunol. 2022 Jan 10;12:832292.
