Abstract
Study design: Prospective randomized pilot trial.
Background: Adhesive capsulitis (AC) occurs five times more often in people with diabetes. Exercises are usually recommended to manage AC. However, the recovery is slow and often incomplete, especially for patients with diabetes. Aerobic exercises improve hyperglycemia and insulin sensitivity. Currently, no research has formally assessed the benefits of incorporating an aerobic training program into the treatment plan of AC in patients with diabetes.
Purpose: This pilot trial compared the effect of a regular physiotherapy (PT) program (PT) to a regular PT program combined with a progressive walking program (PT+) in patients with and without diabetes who have AC.
Methods: Eight patients with (n=3) and without (n=5) diabetes (five men and three women, with a mean age of 57 years) were included. Patients were randomly allocated either to PT or PT+ groups. The functional performance of shoulder was the primary outcome as measured using the Functional Impairment Test-Hand and Neck/Shoulder/Arm (FIT-HaNSA) test. Secondary outcomes included shoulder pain and function; shoulder range of motion in flexion, abduction, and external rotation; muscle strength of shoulder flexors and abductors; and physical activity level. The primary outcome was evaluated at baseline and after six weeks. Secondary outcomes were evaluated at baseline, and after three, six and 12 weeks from enrollment.
Setting: Single centre study at a tertiary-care hospital.
Results: A total of 13 patients were contacted with study details, only eight patients accepted to participate, with a 62% recruitment rate. Adherence to research centre visits was (97%). Patients in both groups showed improvement in all outcome measures, with a mean change of PT= 38 ± 17, and PT+= 6 ± 33 seconds for FIT-HaNSA from baseline to six weeks follow-up. Future studies, with 80% power (a=0.05, ß=0.20) to detect a 20% between-group difference, would require a sample size of 89 participants per group.
Conclusion: This randomized pilot trial established that conducting a large-scale study to assess the effect of the physiotherapy program for managing AC is feasible. The current findings suggest that physiotherapy exercises may be effective in reducing pain and improving shoulder function and ROM in patients with and without diabetes who have AC. Researchers should be aware of the recruitment challenges and should work on minimizing performance and detection bias by blinding study personnel and outcome assessors.
Keywords
Adhesive capsulitis, Diabetes, Physiotherapy, Pilot trial
Abbreviations
AC: Adhesive Capsulitis; ROM: Range of Motion; PT: Physiotherapy; RCT: Randomized Clinical Trial; FITHaNSA: Functional Impairment Test - Hand and Neck/Shoulder/Arm tests; SPADI: Shoulder Pain and Disability Index; RAPA: Rapid Assessment of Physical Activity; BMI: Body Mass Index
Background
Adhesive capsulitis (AC), also known as ‘frozen shoulder’, is characterized by the development of dense adhesions and capsular thickening leading to a progressive and painful restriction of shoulder range of motion (ROM) and functional disability [1]. The onset is gradual, usually occurs between the ages of 40 and 60 years and is more common in females and diabetics [1].
Adhesive capsulitis has been described as a self-limiting condition that progresses through pain, frozen and thawing phases. However, Wong et al. examined the quality of the evidence that describes the theory of AC phases and reported a lack of evidence to support these theoretical phases of AC [2].
The condition can be classified as primary or secondary AC. Primary or idiopathic AC has no clear underlying cause [3]. However, secondary AC might develop following soft tissue injury, joint arthritis, or secondary to known systemic disease such as diabetes [4]. The prevalence of AC is reported to be 10-76% in type 1 and 7-30% in type 2 diabetes as compared to 0-10% in the general population [5]. Adhesive capsulitis was also reported to be associated with age in both types of diabetes [6] and with the duration in type 1 diabetes [5].
The usual approach for managing AC includes steroid injections, joint mobilization and exercises to restore function. Several systematic reviews reported that active exercises and joint mobilization can reduce pain, restore shoulder ROM and function in both short- and long-term follow up [7,8]. Only one recent systematic review has assessed the effectiveness of non-surgical intervention for managing AC in patients with diabetes and reported that low quality evidence suggests large effects of joint mobilization plus exercises on AC in diabetic patients [9].
Although some of the non-surgical interventions have been shown to be effective in managing AC, recovery is slow and often incomplete, especially for people with diabetes [10]. Patients with diabetes often develop longlasting shoulder stiffness and functional disability than patients without diabetes [10,11].
Aerobic exercises are reported to improve hyperglycemia and insulin sensitivity in skeletal muscle that can reduce diabetes related complications such as hypertension and hyperlipidemia [12]. None of the previous research has formally assessed the benefits of incorporating an aerobic training program into the treatment plan of AC in patients with diabetes.
The purpose of this pilot randomized trial was to compare the effect of a regular physiotherapy (PT) program to a regular PT combined with a walking program (PT+) in patients with and without diabetes who have AC. This pilot trial also evaluated the feasibility of recruitment, randomization, retention, assessment procedures, and implementation of the novel intervention. Data from this pilot trial was used to calculate an accurate sample size for a full-scale randomized clinical trial RCT.
Methods and Materials
Study design and setting
A prospective single-blinded parallel pilot RCT was conducted at a tertiary hospital. Local Research Ethics Board has approved the study (Project ID: 111647).
Inclusion and exclusion criteria
Inclusion criteria: 1) A confirmed diagnosis of AC. A diagnosis of AC was confirmed by the consultant shoulder surgeon (KF), who was blinded to treatment allocation, based on the following diagnostic criteria: shoulder pain for at least one month; inability to sleep on the affected side; and restriction of active and passive ROM in one or more planes [13]; 2) Patients aged 18 years or more; 3) A normal radiological appearance of the glenohumeral joint and 4) Ability to participate in the study and follow treatment instructions.
Exclusion criteria: Patients with previous shoulder surgery, significant shoulder injury within six-months, history of shoulder dislocation or arthritis, and patients with suspected rotator cuff tear were excluded from this study.
Outcome measures
Primary outcome measure: The primary outcome was the Functional Impairment Test - Hand and Neck/ Shoulder/Arm (FIT-HaNSA) tests. The FIT-HaNSA test measures the functional performance of the upper limb, while performing multi-level tasks. In the first task (waist-up), the patient lifts three one-kg containers one at a time, with the affected arm, between a shelf at waist level and a shelf 25 cm higher at speed of 60 beats per minute for five minutes or until patient is unable to continue. In the second task (eye-down), the patient returns the three containers back to the waist level shelf. In the third task (overhead work), using both arms, the patient repeatedly screws and unscrews bolts to simulate overhead work for five minutes or until patient feels unable to continue. The time of each task was determined using a stopwatch and the rhythmic speed was controlled using an auditory metronome (Soundbrenner, Berlin). All tasks were performed from a standing position. This test has been shown to be valid and reliable [14].
Secondary outcome measures: Secondary outcomes included shoulder range of motion (ROM) in flexion, abduction, and external rotation using a standard goniometer; shoulder pain and function using Shoulder Pain and Disability Index (SPADI) questionnaire; muscle strength of shoulder flexors and abductors using a dynamometer; and physical activity level using an accelerometer (Fitbit) and the Rapid Assessment of Physical Activity (RAPA) questionnaire. Secondary outcome measures were collected by a single physiotherapist at baseline, and at three, six, and 12 weeks.
Shoulder ROM
Shoulder ROM was measured using a standard goniometer with known concurrent validity and reliability (Intraclass Correlation Coefficients, ICCs > 0.94) [15]. Active flexion and abduction ROM were assessed by measuring the angle formed by the arm and thorax from sitting position. The axis of the goniometer was located at the acromion process; the movable bar was parallel to the humerus while keeping the stationary bar parallel to the trunk [1].
Active external rotation was assessed in sitting position with the arm adducted and the elbow at the side and flexed to 90 degrees. The axis of the goniometer was located at the olecranon process of the elbow and both the stationary and movable bars were parallel to the forearm [1].
Shoulder pain and function
Shoulder pain and function were assessed using SPADI questionnaire. This self-report questionnaire consists of two subscales: pain (five items) and function (eight items). The pain subscale is rated on scale from zero (no pain) to 10 (worst pain ever). The patient is asked to circle the number that best describes their pain and/ or disability. The subscale scores are calculated by adding the item scores for that subscale and dividing this number by the maximum score possible for the items that are deemed applicable by the subject. This number is then multiplied by 100. The two subscales are then added and the total out of 130 is then multiplied by 100. Higher scores indicate greater impairment or disability [16]. The SPADI has been shown to be a valid and reliable measure of shoulder pain and disability [16].
Muscle strength
Isometric muscle strength was assessed for shoulder flexors and abductors using the JTech Power Track handheld dynamometer (JTech; JTech Medical, Salt Lake City, UT, USA), with known concurrent validity [17] and reliability (ICCs 0.89-0.98) [18]. Patients were seated on a straight back chair to stabilize the trunk. Abductor strength was measured by placing the device on the lateral aspect of mid-humerus and flexor strength was measured by placing the device on the anterior aspect of the upper arm.
Assessment of physical activity level
Physical activity level was measured objectively using an accelerometer (Fitbit Zip) and subjectively use a selfreported questionnaire (RAPA). Physical activity level was objectively measured using the Fitbit Zip (Fitbit Inc, USA). This activity tracker contains a three-dimensional accelerometer and is designed to track steps, distance and calories burned. Fitbit Zip is small and discreet and can be worn in a pocket, on a belt or on a bra. Data from the Fitbit Zip syncs automatically to a computer or smartphone using free online application software. Participants were asked to wear the device during all waking hours and to sync their devices on a daily basis for six consecutive weeks. Step count and distance data were obtained from the Fitbit Zip and summarized into an activity tracking sheet. This device has been validated and found to be comparable to other accelerometers [19]. Physical activities were subjectively assessed using RAPA which consists of nine self-reported questions that assess physical activity levels with a response option of yes or no. The first seven questions assess weekly aerobic activity ranging from sedentary to vigorous levels with a total score of 1-7 points, where 1=rarely do any physical activity, and 7=20 minutes of vigorous activities 3+ days/ week. A respondent’s physical activity score is categorized into one of five levels of physical activity: sedentary, underactive, regular underactive (light activities), regular underactive, and regular active. The other two questions assess strength and flexibility training with a total score of three points; one point for strength training and two points for flexibility training. A full description of RAPA is published [20]. The RAPA questionnaire has been validated for use in clinical practice with older adults [20].
Procedures
Patients with and without diabetes, who have been diagnosed with AC were recruited from orthopedic clinics at our tertiary hospital via surgeon referrals. Eligible patients were then given a letter of information and were asked to sign a consent form. After signing the consent form, patients attended an orientation session and were provided with information about the study and the experimental design. Patient’s weight, height, age, gender, type and treatment of diabetes, affected shoulder side (right or left; dominant or non-dominant), and the duration of AC symptoms were collected during this session. Patients were then asked to complete two outcome questionnaires (SPADI and RAPA) and a Katz comorbidity scale [21].
Next, patients underwent blinded randomization into one of the two groups: regular PT program or regular PT with a progressive walking program (PT+). The randomization was stratified by intervention (walking program) and diabetes status using a computergenerated random number table. Patients were allocated into groups using sequentially numbered, opaque, and sealed envelopes issued by the blinded assistant research.
The initial intention of this study was to refer all patients to physical therapy facilities according to their preferences. However, because some patients were recently completed their PT treatment and some others were unable to start a formal PT treatment due to the long wait list at physiotherapy clinics, those patients were provided with a home exercise program from the research team. This program included a group of shoulder exercises that are proven to improve shoulder clinical outcomes in patients with AC. A detailed exercise program is described online [22].
In the PT+ group, patients were instructed to walk at their own pace for 30-45 min, five days per week for six consecutive weeks. They recorded their walking date/time on a diary form provided by the research team. Patients in the PT+ group were not restricted from walking more than 45 minutes a day, as long as they did not feel tired or uncomfortable. Patients in both groups were provided with a Fitbit Zip accelerometer to accurately estimate their physical activity level.
The primary outcome measures were evaluated by a single research team member at baseline and after six weeks. Secondary outcomes were evaluated at baseline, at three and six weeks, and again at 12 weeks after enrollment (Figure 1).
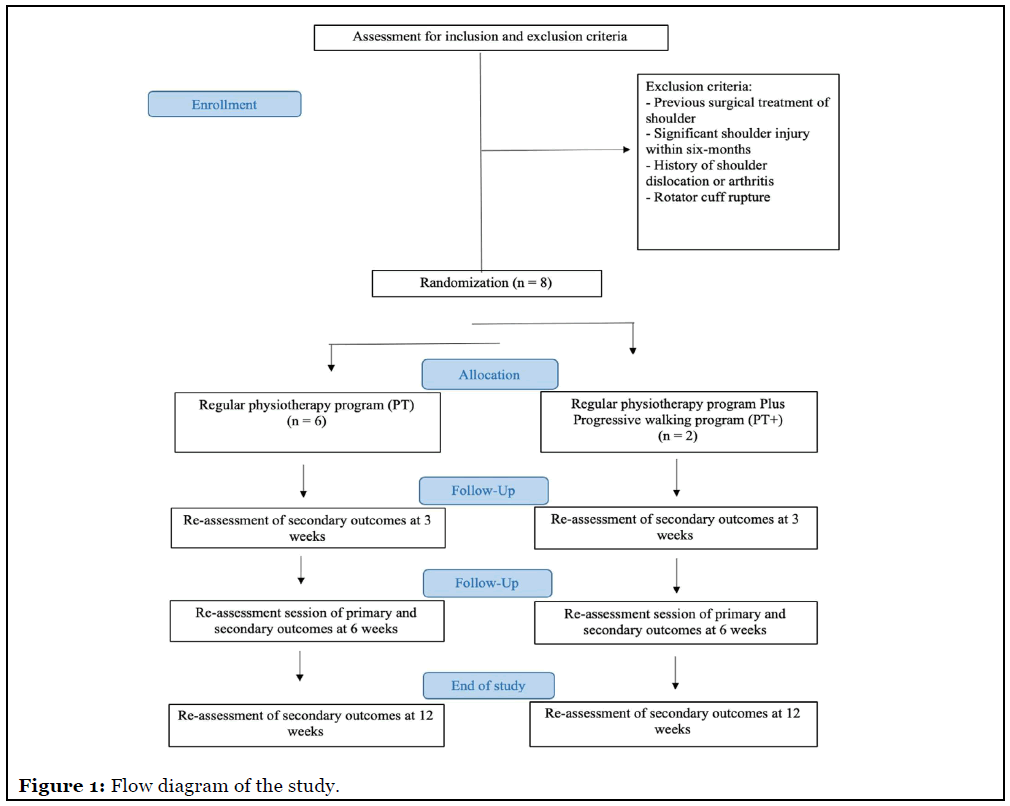
Statistical Analysis
Statistical analysis was performed using SPSS, version 21 (SPSS Inc., Chicago, IL, USA). The analysis of this pilot study is mainly descriptive. Estimates of means and standard deviations for continuous outcomes measures, and an estimate of the proportion for categorical outcome measures were calculated. The recruitment rate was calculated by dividing the total number of patients who consented to participate by the total number of patients contacted with study details. The attendance rate was calculated by dividing the actual number of visits by the total number of all patients’ visits.
Results
Thirteen consecutive patients with AC were referred by the orthopedic surgeon (KF) between September 2018 and November 2019. Of the 13 patients contacted with study details, only eight patients, including three patients with diabetes, agreed to participate in this study, with a 62% recruitment rate (8/13). Adherence with scheduled research centre visits was excellent with a 97% attendance rate (31/32). Only one patient missed a last follow-up visit which included completing SPADI and RAPA questionnaires and measures of active ROM and muscle strength.
This study included five male and three female patients with mean age of 57 years. Table 1 presents the clinical characteristics of patients for both PT and PT+ groups. Six patients were allocated in the PT group, including three patients with diabetes, and two patients were allocated in the PT+ group. Patients in the PT group were overweight with a Body Mass Index (BMI) of 26 and reported more comorbidities (Table 1).
| Variable | Group allocation | All patients | |
|---|---|---|---|
| PT | PT + | ||
| Number of patients | 6 | 2 | 8 |
| Age: mean ± SD (range) years | 56 ± 8 (43-65) | 61 ± 4 (59- 64) | 57±8 (43-65) |
| Sex (M:F) | 4:2 | 1:1 | 5:3 |
| Affected side (n) | |||
| Right | 1 | 1 | 2 |
| Left | 5 | 5 | |
| Both | 1 | 1 | |
| Duration of AC (months) | 17 ± 27 (2-72) | 6 ± 1 (5-7) | 14 ± 23 (2-72) |
| BMI | 26 ± 4 (18-30) | 21 ± 4 (18-23) | 24 ± 5 (18-30) |
| Diabetes (yes / no) | 3:3 | 0:2 | 3:5 |
| Diabetes duration (years) | 5 ± 7 (6-17) | -- | -- |
| Heart disease (n) | 1 | 1 | 2 |
| Hypertension (n) | 3 | -- | 3 |
| Stomach ulcers (n) | 1 | -- | 1 |
| Kidney disease (n) | 1 | -- | 1 |
| Depression (n) | 1 | -- | 1 |
| Osteoarthritis (n) | 3 | -- | 3 |
| Back pain (n) | 3 | 1 | 4 |
PT: Regular Physiotherapy Group, PT+: Regular Physiotherapy plus walking program, n: Number, SD: Standard
Deviation, M: Male, F: Female, AC: Adhesive Capsulitis, BMI: Body Mass Index.
Table 1: Clinical characteristics of patients (n=8).
Three patients (two in PT+) were enrolled in a formal physiotherapy program that included ROM, stretching and strengthening exercises (two patients received one session/month; one patient received 12 sessions: two sessions/week), and five patients were provided a home exercise program by the research team (the five patients completed a formal PT program before the beginning of this study which included PT modalities and exercises).
Change over time in outcome measures
All patients showed improvement over the six weeks follow-up time in the FIT - HaNSA outcome measure (from 155 ± 89 to 180 ± 88). This improvement was more pronounced in the PT group as illustrated in Table 2. Similarly, all patients had improvements in SPADI (pain, function, total), active ROM (flexion, abduction, external rotation), and muscle strength (flexors and abductors) over the 12-week follow-up period. The improvement of these outcome measures was more pronounced in the PT group when compared to PT+ group (Table 2).
| Outcome | Baseline (Mean ± SD) | At three weeks (Mean ± SD) | At six weeks (Mean ± SD) | At 12 weeks (Mean ± SD) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PT (n=6) | PT+ (n=2) | All patients | PT (n=2) | PT+ (n=2) | All patients | PT (n=2) | PT+ (n=2) | All patients | PT (n=6) | PT+ (n=1) | All patients | |
| FIT-HaNSA (Seconds) | ||||||||||||
| Task 1 | 151±89 | 300±0 | 189±102 | - | - | - | 163±71 | 243±81 | 183±77 | - | - | - |
| Task 2 | 88±56 | 237±89 | 124±90 | - | - | - | 124±97 | 212±125 | 146±103 | - | - | - |
| Task 3 | 125±60 | 177±4 | 151±74 | - | - | - | 189±87 | 276±34 | 211±85 | - | - | - |
| Tasks average | 121±68 | 238±47 | 155±89 | 159±85 | 244±80 | 180±88 | ||||||
| SPADI (Pain %) | 69±16 | 61±4 | 67±14 | 60±26 | 64±6 | 61±22 | 54±25 | 45±10 | 52±22 | 38±30 | 64 | 42±29 |
| SPADI(Function %) | 56±19 | 61±4 | 57±16 | 50±28 | 54±6 | 51±24 | 40±28 | 35±8 | 38±24 | 23±29 | 73 | 30±32 |
| SPADI (Total %) | 61±16 | 62±4 | 61±13 | 54±27 | 58±6 | 55±23 | 45±26 | 39±1 | 44±22 | 30±28 | 69 | 35±29 |
| RAPA | 6±2 | 5±2 | 8±1 | 6±2 | 6±1 | 6±2 | 6±2 | 7±1 | 6±2 | 6±2 | 8 | 6±2 |
| AROM (Flexion-degrees) | 102±31 | 125±14 | 107±29 | 104±19 | 130±14 | 111±21 | 107±13 | 128±11 | 112±15 | 116±14 | 100 | 114±14 |
| AROM(Abduction- degrees) | 68±10 | 95±7 | 75±15 | 78±8 | 87±25 | 81±12 | 82±19 | 99±1 | 86±18 | 95±24 | 70 | 91±24 |
| AROM(External rotation-degrees) | 24±6 | 39±13 | 28±10 | 33±14 | 47±9 | 37±14 | 35±8 | 47±2 | 38±9 | 45±11 | 20 | 41±14 |
| Muscle strength (Shoulder flexors- kg) | 10±4 | 11±3 | 10±4 | 10±5 | 12±5 | 11±5 | 11±5 | 11±3 | 11±4 | 13±5 | 8 | 12±5 |
| Muscle strength (Shoulder abductors- kg) | 8±4 | 12±1 | 9±4 | 9±6 | 11±3 | 10±5 | 11±4 | 11±1 | 10±4 | 12±5 | 5 | 11±5 |
FIT-HaNSA: Functional Impairment Test - Hand and Neck/ Shoulder/Arm, PT: Regular Physiotherapy group, PT+: Regular Physiotherapy plus walking program, SD: Standard Deviation, SPADI: Shoulder Pain and Disability Index, RAPA: Rapid Assessment of Physical Activity questionnaire, AROM: Active Range of Motion
Table 2:Descriptive analysis of outcome measures for PT and PT+ groups.
The physical activity level of both groups remained the same throughout the study (RAPA=6) (Table 2). When comparing groups, PT+ group was more active at the six and 12-week follow-up times (RAPA=7, 8, respectively) and showed higher step count and longer travelled distance over the six-week use of the Fitbit activity tracker (Table 3).
| Week/ Variable | Fitbit (steps) (Mean ± SD) | Fitbit (distance -km) (Mean ± SD) | Walking program (n=2) Time (minutes) | |||||
|---|---|---|---|---|---|---|---|---|
| PT | PT+ | All | PT | PT+ | All | N1 | N2 | |
| Week 1 (PT: n= 6; PT+: n = 2) | 4861±2674 | 8199 | 4670±3168 | 3.6±2.2 | 5.6 | 3.5±2.5 | 40 | 55 |
| Week 2 (PT: n= 6; PT+: n = 2) | 5476±2247 | 4919±3773 | 5337±2389 | 4.3±1.7 | 5.5±0.1 | 4.6±1.5 | 40 | 45 |
| Week 4 (PT: n= 6; PT+: n = 2) | 4208±1963 | 7302±1514 | 4981±2266 | 3.5±2.3 | 5.2±1.1 | 3.9±2.1 | 40 | 29 |
| Week 4 (PT: n= 5; PT+: n = 2) | 5695±2057 | 7854±382 | 9850±10319 | 4.2±2.0 | 6.0±0.3 | 4.0±2.2 | 60 | 42 |
| Week 5 (PT: n= 5; PT+: n = 2) | 4501±1781 | 7224±249 | 5279±1972 | 3.5±1.4 | 5.1±0.2 | 3.9±1.4 | 0 | 30 |
| Week 6 (PT: n= 4; PT+: n = 2) | 4556±3342 | 7846±53 | 5652±3096 | 3.8±3.1 | 6.1±0.8 | 4.6±2.7 | 0 | 50 |
| Total | 4883±2344 | 7224±1194 | 5962±3868 | 3.8±2.1 | 5.6±0.5 | 4.1±2.1 | 180 | 251 |
PT: Regular Physiotherapy group, PT+: Regular Physiotherapy plus walking program, SD: Standard Deviation.
Table 3: Description of groups physical activity level.
A comparison between patients with and without diabetes
Table 4 and 5 present a comparison between patients with and without diabetes for all outcome measures. In general, patients with diabetes were younger (53 ± 11 years) and had AC for a longer period of time (29 ± 38 years). In addition, patients with diabetes had worse baseline outcome measures and their improvement at six weeks was less pronounced when compared to patients without diabetes. However, patients with diabetes showed higher improvement at 12-week follow-up period compared to patients without diabetes in response to physiotherapy exercise program (Table 4).
| Variable | Baseline (Mean ± SD) | At three weeks (Mean ± SD) | At six weeks (Mean ± SD) | At 12 weeks (Mean ± SD) | ||||
|---|---|---|---|---|---|---|---|---|
| Diabetes (n=3) | No diabetes (n=5) | Diabetes (n=3) | No diabetes (n=5) | Diabetes (n=3) | No diabetes (n=5) | Diabetes (n=3) | No diabetes (n=4) | |
| Clinical characteristics: | ||||||||
| Age | 53 ± 11 | 61 ± 4 | -- | -- | -- | -- | -- | -- |
| Sex (M:F) | 2:1 | 3:2 | -- | -- | -- | -- | -- | -- |
| BMI | 24 ± 6 | 25 ± 4 | -- | -- | -- | -- | -- | -- |
| AC duration | 29 ± 38 | 6 ± 2 | -- | -- | -- | -- | -- | -- |
| FIT-HaNSA (seconds) | ||||||||
| Task 1 | 112 ± 31 | 234 ± 103 | -- | -- | 136 ± 16 | 211 ± 87 | -- | -- |
| Task 2 | 79 ± 47 | 152 ± 103 | -- | -- | 92 ± 58 | 178 ± 116 | -- | -- |
| Task 3 | 97 ± 35 | 183 ± 74 | -- | -- | 184 ± 101 | 226 ± 81 | -- | -- |
| SPADI (Pain) | 77 ± 10 | 61 ± 13 | 71 ± 11 | 56 ± 26 | 67 ± 6 | 43 ± 23 | 31 ± 26 | 50 ± 32 |
| SPADI (Function) |
67 ± 1 | 52 ± 19 | 61 ± 5 | 45 ± 29 | 47 ± 16 | 33 ± 28 | 12 ± 10 | 44 ± 38 |
| SPADI (Total) | 71 ± 4 | 55 ± 14 | 65 ± 4 | 49 ± 28 | 55 ± 12 | 37 ± 25 | 19 ± 16 | 48 ± 33 |
| RAPA | 5 ± 2 | 6 ± 2 | 6 ± 3 | 6 ± 2 | 6 ± 2 | 6 ± 2 | 7 ± 2 | 6 ± 2 |
| AROM (Flexion) |
83 ± 38 | 122 ± 8 | 92 ± 20 | 122 ± 11 | 97 ± 3 | 121 ± 11 | 118 ± 16 | 110 ± 14 |
| AROM (Abduction) |
65 ± 5 | 81 ± 17 | 77 ± 7 | 83 ± 15 | 73 ± 3 | 94 ± 19 | 100 ± 26 | 85 ± 23 |
| AROM (External rotation) |
23 ± 7 | 31 ± 11 | 27 ± 6 | 43 ± 14 | 30 ± 10 | 43 ± 4 | 45 ± 17 | 39 ± 13 |
| Muscle strength (Shoulder flexors) | 10 ± 1 | 10 ± 5 | 10 ± 4 | 11 ± 5 | 11 ± 5 | 11 ± 4 | 14 ± 4 | 11 ± 6 |
| Muscle strength (Shoulder abductors) | 6 ± 2 | 10 ± 4 | 6 ± 4 | 12 ± 5 | 10 ± 4 | 10 ± 4 | 12 ± 3 | 11 ± 7 |
M: Male, F: Female, AC: Adhesive Capsulitis, FIT-HaNSA: Functional Impairment Test - Hand and Neck/ Shoulder/ Arm, SD: Standard Deviation, SPADI: Shoulder Pain and Disability Index, RAPA: Rapid Assessment of Physical Activity questionnaire, AROM: Active Range of Motion
Table 4: A comparison of outcome measures between patients with and without diabetes.
| Week/ Variable | Fitbit (steps) (Mean ± SD) | Fitbit (distance -km) (Mean ± SD) | ||
|---|---|---|---|---|
| Diabetes | No-diabetes | Diabetes | No-diabetes | |
| Week 1 (diabetes: n= 3; no diabetes: n= 5) | 5320 ± 3547 | 4280 ± 3280 | 4.0 ± 3.0 | 3.1 ± 2.4 |
| Week 2 (diabetes: n= 3; no diabetes: n= 5) | 5577 ± 3191 | 5193 ± 2198 | 4.0 ± 2.4 | 5.0 ± 0.8 |
| Week 3 (diabetes: n= 3; no diabetes: n= 5) | 3645 ± 2874 | 5783 ± 1647 | 3.4 ± 3.5 | 4.2 ± 1.1 |
| Week 4 (diabetes: n= 3; no diabetes: n= 4) | 4708 ± 2267 | 6435 ± 1825 | 3.2 ± 2.6 | 4.9 ± 1.4 |
| Week 5 (diabetes: n= 3; no diabetes: n= 4) | 4830 ± 2365 | 5616 ± 1922 | 3.8 ± 1.8 | 4.0 ± 1.3 |
| Week 6 (diabetes: n= 3; no diabetes: n= 3) | 4595 ± 4092 | 6710 ± 1967 | 3.9 ± 3.8 | 5.2 ± 1.6 |
| Total | 4779 ± 3056 | 5670 ± 2140 | 3.7 ± 2.9 | 4.4 ± 1.4 |
SD: Standard Deviation
Table 5: A comparison of physical activity level of patients with and without diabetes.
The level of physical activity for patients with diabetes was less at baseline (RAPA=5) but increased over time and became higher than patients without diabetes at week 12 (RAPA=7). However, patients without diabetes showed higher step count and longer travelled distance as compared to patients with diabetes over the six-week use of the Fitbit activity tracker as presented in Table 5.
Sample size
Sample size calculations were based on the mean FITHaNSA scores and the pooled Standard Deviation (SD) at the six-week follow-up visit. We calculated the sample size for future studies using Zhong (2009) formula [23]. The clinically acceptable margin was determined by calculating a 20% clinically important difference in the follow-up mean score. This resulted in 77 participant/ group and a total of 154. Considering a drop-out rate of 15%, the total sample size required is 178 (89 per group).
Discussion
This prospective randomized pilot trial determined that conducting a large-scale study to assess the effect of physiotherapy program for managing AC is feasible. However, the effect of adding a progressive walking program to PT exercises was not tested for patients with diabetes due to the stratified randomization with this small sample; all patients with diabetes (n=3) were randomized to the regular PT program group.
Participant recruitment was satisfactory and there were no dropouts. For future studies, a sample size of 178 (89 participants per group), to detect 20% difference between-groups is required. Seven patients would need to be recruited each month over 26-month period to successfully complete the trial in a single Centre. A multicenter approach may be more pragmatic and efficient.
This study demonstrated that clinical outcomes including shoulder performance, shoulder pain and function, active ROM, and muscle strength improved in patients with AC over a 12-week follow-up period. The improvement was more pronounced in the PT group as compared to PT+ group; however, we consider these results unstable due to the very low sample size of PT+ group. However, patients in PT+ group were more physically active as indicated by RAPA and Fitbit results. This can be due to the nature of our study in which the research team encouraged patients in the PT+ group to perform regular walking activity. Provision of the Fitbit might in itself have acted as a motivator for improving physical activity; since this is not a standard practice.
Although patients with diabetes had worse outcome measures at baseline, they improved overtime and showed greater recovery at week 12 when compared to patients without diabetes. At the 12-week followup visit, two patients without diabetes reported severe pain and inability to move their arm after performing intensive housekeeping activities. This may explain the inferior recovery in the nondiabetic patient group. However, the level of physical activity was lower in patients with diabetes at baseline as indicated by the Fitbit results although the RAPA score was higher in patients with diabetes. The sample size was too small to detect treatment differences and to make definitive conclusions. However, the preliminary findings of this study, including the excellent adherence rate to research centre visits and the acceptable recruitment rate, show that the novel approach taken in this pilot trial is worthy of investigation in future randomized trials.
It was difficult to recruit the required number of patients for this pilot trial. Although our recruitment rate was acceptable (62%), not all patients agreed to take part in the trial and few (n=13) were referred to our centre. Patients had several reasons for refusing to participate in the study including lengthy travel distances to the research centre and insufficient time to commit to the study. The low recruitment rate in this trial may be due to the fact that patients with early stages of AC are usually seen by primary care centres who prescribe patient medications and refer them to physiotherapy facilities. The infrequent referrals limited us from recruiting enough patients to this trial and in the future, large trials on patients with AC may be best conducted at a primary care clinical setting.
The research team leader (SA) was not part of the clinical team and this also may have affected recruitment of subjects for the study. In spite of the research leader regularly communicating with other clinical team members such as physiotherapists and administrative people, the recruitment was lower than expected. As this is a critical issue, improving communication between researchers and clinical team members may be important.
In this trial, the rate of recruitment (62%) was acceptable and the rate of adherence (97%) was excellent, with only one patient lost to a final follow-up session. Patients were required to participate in many assessment sessions, an initial assessment session and in three more assessment sessions (at three weeks, six weeks, and 12 weeks), and to attend physiotherapy treatment sessions or perform the home exercise program. Both of these programs involved time and effort from patients. This suggests that with the current methodology, a future study is feasible and performing advanced statistical analysis with larger sample sizes seems achievable. However, it is important to take into account that AC is a disabling condition and patients are usually eager to try something to get their shoulder feeling better. If it was less disabling, the adherence to the program would likely be lower.
Due to the random and concealed allocation of patients in this trial, selection bias was minimized. In addition, patients in this trial were blinded to treatment groups, PT vs. PT+, and were randomly referred to physiotherapy facilities that might minimized performance bias. However, since some patients were given a home exercise program by the research leader, there may be a source of performance bias. Further, the research leader performed all the assessments and was aware of group allocations which is considered a source of detection bias. The attrition rate was minimal during the baseline, three weeks, and six weeks assessment sessions, however, at 12-week assessment session, one patient was lost to follow-up with an attrition rate of 13%. Lastly, based on our initial registered protocol, all the mentioned primary and secondary outcomes were collected and reported in this trial which minimized reporting bias. However, there was an intention to collect data at 24-week followup session, but because patients were unwilling to attend this session, no data was collected.
The main aim of all AC physiotherapy programs is to relieve pain, improve function, increase shoulder ROM and to improve the patient’s quality of life. In the current literature, there is no consensus on the most appropriate treatment for AC in patients with and without diabetes [9,24]. Several studies have shown that physiotherapy interventions (mainly exercises) reduce pain, restore shoulder function, and improve ROM in patients with and without diabetes who have AC [25,26], which concur with the results of the current trial. However, the recovery might be similar [27] or inferior [10] to patients without diabetes contradicting the results of this pilot trial. Because of the very low sample size of patients with diabetes in this pilot trial, comparing its results to other studies could be misleading.
Lastly, no previous research has investigated the effect of incorporating specific shoulder exercises with aerobic training program to investigate shoulder recovery in patients with diabetes who have AC. Aerobic exercises that improve hyperglycemia and insulin sensitivity in skeletal musculature may have an impact on the pathophysiology of AC. Because none of our patients with diabetes were randomized to the group that include regular walking program, examining this effect was not possible. Future studies are required to examine the effect of adding walking program to the shoulder specific exercises for managing AC and to assess if walking is the ideal aerobic exercise for this population or other aerobic programs such as swimming or biking.
Strengths and Limitations
The strengths of this study are: (1) All assessment sessions were performed by the research leader which reduced assessment variations among patients; (2) This trial reduced selection bias by having a computerized randomization process; (3) In this trial, the surgeon and the research leader were trained and experienced in the field of shoulder rehabilitation which contributed to the successful conduct of this trial. However, there are some limitations to this pilot that need to be recognized: (1) Having a small sample size has reduced the power of this trial; (2) The inability to blind the outcome assessor has introduced detection bias to this study; (3) Since no patient with diabetes were randomized to the walking program, examining the effect of incorporating aerobic program to specific shoulder exercise for managing AC was not possible.
Clinical Impacts and Future Research Directions
This study suggests that physiotherapeutic exercises might be effective in reducing pain and improving shoulder function in patient with and without diabetes who have AC.
• Based on the findings of the current pilot trial, we have calculated the sample size for future randomized trials; for a trial to have 80% power ((α= 0.05, β=0.20), and to detect 20% difference between-groups, it would require a minimum of 89 participants per group and 178 participants in-total.
• Rigorous randomized controlled trials are needed to define the optimal combination of physiotherapy interventions for managing AC in patients with and without diabetes.
• Future trials should be well-designed to minimize biases and should be reported using CONSORT criteria [28].
• Better reporting of standardized outcomes is needed including reliable and responsive measures of physical activity level (RAPA) and FIT-HaNSA. Standardized measurement instruments would improve the quality of existing research and contribute to the ability to conduct meta-analysis in future.
• The research team members have identified some barriers to patients’ recruitment. Future trials should be aware of the possible challenges of conducting a large-scale randomized trial.
Conclusion
This randomized pilot trial established that conducting a large-scale study to assess the effect of physiotherapy program for managing AC is feasible. However, the effect of adding progressive walking program to PT exercises was not tested for patients with diabetes due to the stratified randomization with this small sample; all patients with diabetes were randomized to the regular PT program group. The current findings suggest that physiotherapy exercises may be effective in reducing pain and improving shoulder function and ROM in patients with and without diabetes who have AC. Researchers should be aware of the recruitment challenges and should work on minimizing performance and detection bias by blinding study personnel and outcome assessors.
Trial Registration
ClinicalTrials.gov, ID number: NCT03462420. Registered 30 January 2018, https://register. clinicaltrials.gov/prs/app/action/LoginUser?ts=2&cx=- jg9qo3
Ethics Approval and Consent to Participate
The University of Western Ontario Research Ethics Board for Health Sciences Research Involving Human Subjects (HSREB). Informed consent was obtained from all participants. Project ID: 111647.
Competing Interests
The authors declare that there is no conflict of interest
Acknowledgements and Funding
The authors would like to thank all patients who participated in the study. Dr. Joy C MacDermid was supported by a CIHR Chair in Gender, Work and Health and the Dr. James Roth Research Chair in Musculoskeletal Measurement and Knowledge Translation during the conduct of this study. CIHR FRN: SCA-145102.
References
2. Wong CK, Levine WN, Deo K, Kesting RS, Mercer EA, Schram GA, et al. Natural history of frozen shoulder: fact or fiction? A systematic review. Physiotherapy. 2017 Mar 1;103(1):40-7.
3. Codman EA. The shoulder. Kreiger Publishing Company Malabar FL; 1934.
4. Bunker TD. Frozen shoulder : unravelling the enigma. Annals of the Royal College of Surgeons of England. 1997 May;79(3):210.
5. Zreik NH, Malik RA, Charalambous CP. Adhesive capsulitis of the shoulder and diabetes: a meta-analysis of prevalence. Muscles Ligaments Tendons Journal. 2016 Jan;6(1):26.
6. Arkkila PET, Gautier J-F. Musculoskeletal disorders in diabetes mellitus: an update. Best Practice & Research Clinical Rheumatology. 2003 Dec 1;17(6):945-70.
7. Blanchard V, Barr S, Cerisola FL. The effectiveness of corticosteroid injections compared with physiotherapeutic interventions for adhesive capsulitis: a systematic review. Physiotherapy. 2010 Jun 1;96(2):95-107.
8. Jain TK, Sharma NK. The effectiveness of physiotherapeutic interventions in treatment of frozen shoulder / adhesive capsulitis : A systematic review. Journal of Back and Musculoskeletal Rehabilitation. 2014 Jan 1;27(3):247-73.
9. Alsubheen S, Nazari G, Bobos P, MacDermid JC, Overend TJ, Faber K. Effectiveness of nonsurgical interventions for managing adhesive capsulitis in patients with diabetes: A systematic review. Archives of Physical Medicine and Rehabilitation. 2019 Feb 1;100(2):350-65.
10. Vastamäki H, Ristolainen L, Vastamäki M. Range of motion of diabetic frozen shoulder recovers to the contralateral level. Journal of International Medical Research. 2016 Dec;44(6):1191-9.
11. Rill BK, Fleckenstein CM, Levy MS, Nagesh V, Hasan SS. Predictors of outcome after nonoperative and operative treatment of adhesive capsulitis. The American Journal of Sports Medicine. 2011 Mar;39(3):567-74.
12. Marwick TH, Hordern MD, Miller T, Chyun DA, Bertoni AG, Blumenthal RS, et al. Exercise training for type 2 diabetes mellitus. Circulation. 2009 Jun 30;119(25):3244-62.
13. Bulgen DY, Hazleman BL, Roberts S. Frozen shoulder : long-term prospective study. Annals of the rheumatic diseases. 1984 Jun 1;43(3):361-4.
14. MacDermid JC, Ghobrial M, Quirion KB, St- Amour M, Tsui T, Humphreys D, et al. Validation of a new test that assesses functional performance of the upper extremity and neck (FIT-HaNSA) in patients with shoulder pathology. BMC Musculoskeletal Disorders. 2007 Dec;8(1):1-0.
15. Kolber MJ, Hanney WJ. The reliability and concurrent validity of shoulder mobility measurements using a digital inclinometer and goniometer: a technical report. International Journal of Sports Physical Therapy. 2012 Jun;7(3):306.
16. MacDermid JC, Solomon P, Prkachin K. The Shoulder Pain and Disability Index demonstrates factor, construct and longitudinal validity. BMC musculoskeletal disorders. 2006 Dec 1;7(1):12.
17. Roy J-S, MacDermid JC, Orton B, Tran T, Faber KJ, Drosdowech D, et al. The concurrent validity of a hand-held versus a stationary dynamometer in testing isometric shoulder strength. Journal of Hand Therapy. 2009 Oct 1;22(4):320-7.
18. Dollings H, Sandford F, O’conaire E, Lewis JS. Shoulder strength testing: the intra-and intertester reliability of routine clinical tests, using the PowerTrackTM II Commander. Shoulder & Elbow. 2012 Apr;4(2):131-40.
19. Tully MA, McBride C, Heron L, Hunter RF. The validation of Fitbit ZipTM physical activity monitor as a measure of free-living physical activity. BMC Research Notes. 2014 Dec;7(1):1-5.
20. Topolski TD, LoGerfo J, Patrick DL, Williams B, Walwick J, Patrick MAJMB. Peer reviewed: the Rapid Assessment of Physical Activity (RAPA) among older adults. Preventing Chronic Disease. 2006 Oct;3(4).
21. Sangha O, Stucki G, Liang MH, Fossel AH, Katz JN. The Self-Administered Comorbidity Questionnaire: a new method to assess comorbidity for clinical and health services research. Arthritis Care & Research: Official Journal of the American College of Rheumatology. 2003 Apr 15;49(2):156-63.
22. Department of Orthopaedics and Sports Medicine. Home Exercises for the Stiff or Frozen Shoulder [Internet]. 2020;Available from: https://orthop.washington.edu/ patient-care/articles/shoulder/home-exercises-for-thestiff- or-frozen-shoulder.html
23. Zhong B. How to calculate sample size in randomized controlled trial? J Thorac Dis 2009;1(1):51–4.
24. Yip M, Francis A-M, Roberts T, Rokito A, Zuckerman JD, Virk MS. Treatment of adhesive capsulitis of the shoulder: a critical analysis review. JBJS Reviews. 2018 Jun 1;6(6):e5.
25. Ekim AA, Inal EE, Gönüllü E, Hamarat H, Yorulmaz G, Mumcu G, et al. Continuous passive motion in adhesive capsulitis patients with diabetes mellitus: A randomized controlled trial. Journal of Back and Musculoskeletal Rehabilitation. 2016 Jan 1;29(4):779-86.
26. Muthukrishnan R, Rashid AA, Al-Alkharji F. The effectiveness of extracorporeal shockwave therapy for frozen shoulder in patients with diabetes: randomized control trial. Journal of Physical Therapy Science. 2019;31(7):493–7.
27. Düzgün I, Baltaci G, Atay OA. Manual therapy is an effective treatment for frozen shoulder in diabetics: an observational study. Joint Diseases and Related Surgery. 2012; 23 (2):094-9.
28. Moher D, Hopewell S, Schulz KF, Montori V, Gøtzsche PC, Devereaux PJ, et al. CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. International Journal of Surgery. 2012 Jan 1;10(1):28-55.
