Abstract
Background: Transcatheter aortic valve replacement (TAVR) is currently the most commonly utilized management strategy for severe aortic stenosis. The short-term outcomes of TAVR in patients who experienced a recent acute myocardial infarction (AMI) remain unclear.
Methods: Using data from the National Readmissions Database years 2016 to 2020, we identified patients who underwent TAVR and had a recent AMI (either ST-elevation [STEMI] or non-ST-elevation myocardial infarction [NSTEMI]) and underwent percutaneous coronary intervention (PCI) within 90 days prior to the TAVR. The primary outcome assessed was in-hospital mortality. A multivariable logistic regression model adjusting for patient and hospital level characteristics was conducted.
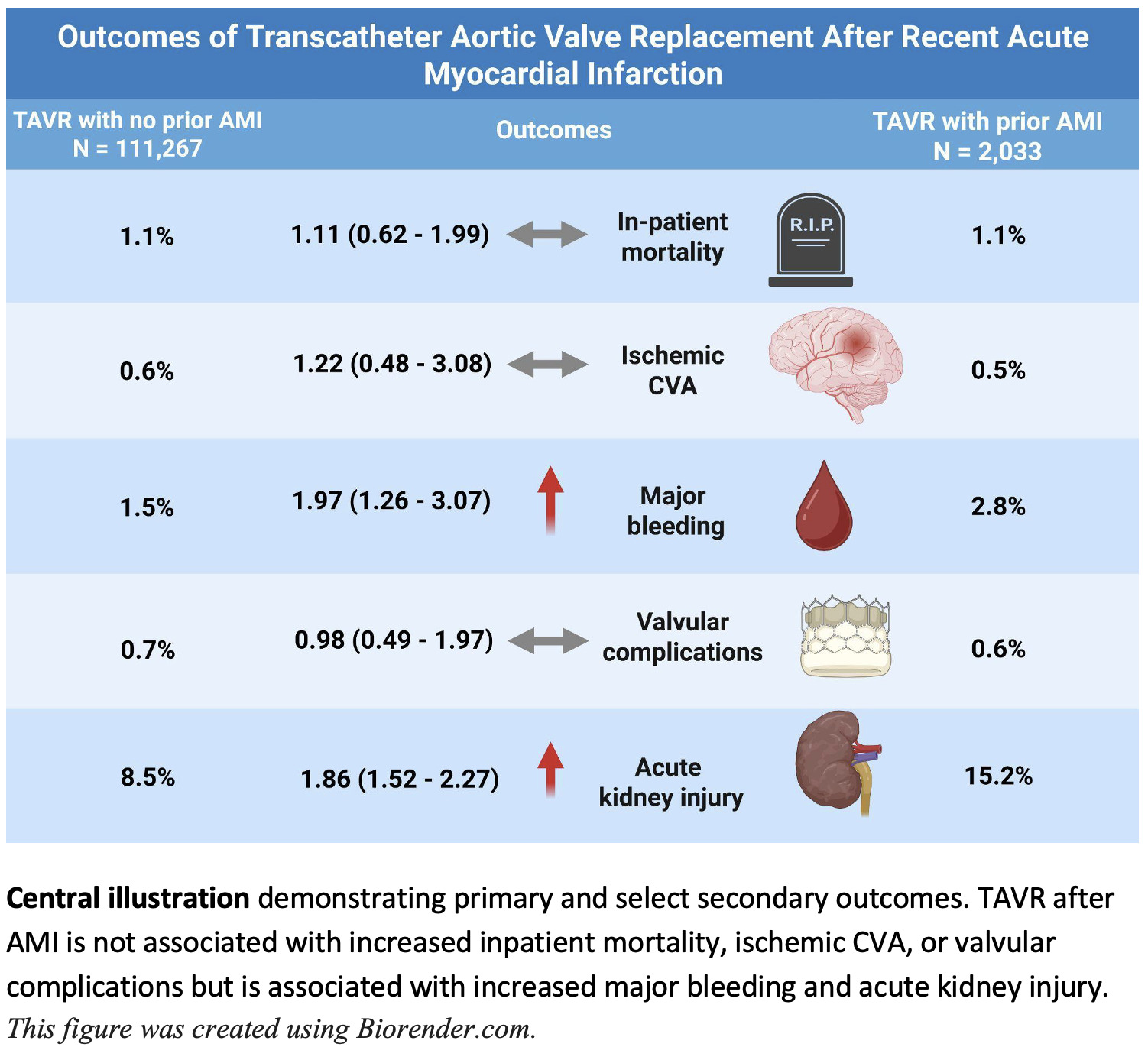
Results: Among 113,300 patients who underwent TAVR, 2,033 (1.8%) had an AMI within 90 days prior to the TAVR. Compared with those who did not have a recent AMI, there was no significant difference in in-hospital mortality (adjusted odds ratio [aOR] 1.11, 95% confidence interval [CI] 0.62 - 1.99, P=0.72), stroke (aOR 1.22, CI 0.48 - 3.08, P=0.68), and valvular complications (aOR 0.98, CI 0.49 - 1.97, P=0.95). However, there was a higher incidence of major bleeding (aOR 1.97, CI 1.26 - 3.07, P=0.003) and acute kidney injury (aOR 1.86, CI 1.52 - 2.27, P<0.001). There was no difference in the outcomes based on the initial presentation (STEMI vs NSTEMI).
Conclusions: Among patients who are undergoing TAVR and had an AMI within 90 days, TAVR appears to be safe, and not associated with higher short-term mortality. However, these patients had a higher incidence of AKI and major bleeding.
Keywords
TAVR, Acute MI, STEMI, NSTEMI, Outcomes research, Aortic stenosis, Structural heart disease
Introduction
Transcatheter aortic valve replacement (TAVR) has become the most common therapeutic approach for severe aortic stenosis (AS), irrespective of surgical risk. TAVR is associated with lower mortality rates, stroke, and major bleeding compared with surgical aortic valve replacement [1,2]. Approximately 50–75% of AS patients referred for aortic valve replacement (AVR) have coexisting coronary artery disease (CAD) [1,3]. Concomitant CAD worsens AS by increasing myocardial ischemia and left ventricular pressure [4]. Furthermore, patients with concomitant obstructive CAD and severe AS have worse outcomes, including lower survival and worse morbidity after valve replacement [5]. Previous studies have shown that coexistent CAD does not increase the risk of mortality, readmission, or symptoms of patients undergoing TAVR [6]. However, data examining the outcomes of TAVR among patients who had a recent acute myocardial infarction (AMI) remain scarce. Current guidelines recommend delaying elective surgeries for at least 4-6 weeks after an AMI to reduce perioperative risk, but they do not specifically address the timing of TAVR after a recent AMI [7,8]. Using a nationally representative database, we sought to assess the short-term outcomes among patients who had any AMI within 90 days prior to TAVR.
Methods
Data sources and study population
This is an observational study using the National Readmission Database (NRD). Data was collected between the years 2016–2020. The NRD is a large, public, all-payer, readmission database that provided by the Agency of Healthcare Research and Quality. The NRD includes more than 50% of hospitalizations in the United States. Discharge weights for each record are employed to ensure that the results are generalizable.
We included adults, defined as greater than 18 years of age, who underwent hospitalization for TAVR using the appropriate ICD-10 codes. We then categorized the patients to those who had any AMI (defined as ST-elevation [STEMI] or non-ST-elevation myocardial infarction [NSTEMI]) and underwent percutaneous coronary intervention (PCI) within 90 days prior to the TAVR versus patients without an AMI and PCI. The ICD-10 codes used are listed in Supplemental Table 1. Since the NRD does not cross over in the following calendar year, we included hospitalizations for TAVR procedures between April and December to allow for a retrospective period of 3 months for any hospitalizations for AMI prior to TAVR.
The following patient and hospital level characteristics were identified: age, sex, hypertension, diabetes mellitus, smoking, peripheral vascular disease, chronic kidney disease, heart failure, primary insurance, median income for patient’s ZIP code, hospital bed size, hospital region, and teaching status.
Outcomes
The primary outcome was all-cause in-hospital mortality. Secondary outcomes included ischemic cerebrovascular accident (CVA), major bleeding (defined as gastrointestinal, intracranial, pulmonary, and other bleeding), valvular complications (defined as valvular leak, valve displacement, valve infection, valve breakdown, and unspecified valvular complications), acute kidney injury (AKI), length of stay, and total charges.
Statistical analysis
Categorical variables were summarized as frequencies and percentages (chi-square tests used to compare) while continuous variables were described as means and standard deviations (student t-test used to compare). Logistic regression was used to compare dichotomous and categorical variables while linear regression was used to compare continuous variables. Multilevel multivariate models were used for both logistic and linear regressions. The models were adjusted for age, sex, hypertension, diabetes mellitus, smoking, peripheral vascular disease, history of CAD (defined as history of AMI, history of PCI, or history of coronary artery bypass surgery), heart failure, primary insurance, median income for patient’s ZIP code, hospital bed size, teaching status. These variables were included after running the individual baseline characteristics against the primary outcome and the variables that were statistically significant (P<0.05) were included. Two subgroup analyses were performed: i) based on the timing of AMI: within 30 days and within 31–90 days before the TAVR; and ii) based on the initial AMI presentation (i.e., STEMI versus NSTEMI). A sensitivity analysis was performed after excluding patients who underwent transapical TAVR. A p value <0.05 was used as the threshold for statistical significance. Furthermore, p values were two sided. Summary estimates were reported as odds ratio (OR) and corresponding 95% confidence interval. According to the HCUP data use agreement, any variable with <11 observations was not reported. Analyses were conducted using STATA 18 (STATA Corp, College Station, Texas 77845 USA).
Results
Among 113,300 patients who underwent TAVR during the study period, 2,033 (1.8%) had an AMI and PCI performed within the prior 90 days. Of which, 952 patients had an AMI within 30 days before TAVR, and 1,081 patients had an AMI within 31–90 days (Figure 1). The median time between the AMI and TAVR was 31 days. For patients who had the AMI within 30 days before the procedure, the median time between the AMI and TAVR was 13 days. In contrast, the average time for patients with a TAVR between 31–90 days after the AMI was 51 days. Regarding demographic data, patients with recent AMI had a lower population of women (36.3% vs 43.2% in those without recent AMI). Most patients were insured through Medicare, and most procedures were performed in large, metropolitan, teaching hospitals. The baseline characteristics are summarized in Table 1.
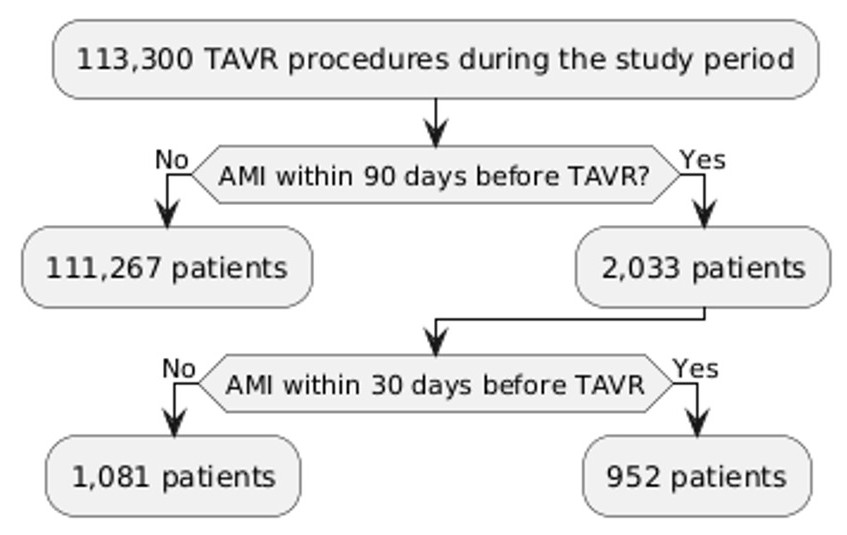
Figure 1. Flow chart.
|
TAVR with no prior AMI |
TAVR with prior AMI |
P value |
|
|
Age, mean and SD |
78.2 (±0.1) |
78.2 (±0.4) |
0.94 |
|
Female (%) |
48,059 (43.2) |
732 (35.9) |
<0.001 |
|
Primary insurance (%) Medicare Medicaid Private insurance Self-pay No charge/other |
98,409 (88.5) 1,477 (1.3) 8,329 (7.4) 351 (0.3) 2,578 (2.3) |
1,812 (89.1) 26 (1.3) 133 (6.5) 7 (0.3) 56 (2.8) |
0.78
|
|
Median household income for patient's zip code (%) 0–25th percentile 26th–50th percentile 51st–75th percentile 76th–100th percentile |
30,935 (28.1) 29,707 (27) 26,358 (24) |
612 (30.5) 472 (23.6) 427 (21.3) |
0.003
|
|
Hospital bed size (%) Small Medium Large |
5,849 (5.3) 25,594 (23) 79,824 (71.7) |
133 (6.5) 494 (24.3) 1,407 (69.2) |
0.21
|
|
Hospital location and teaching status (%) Metropolitan non-teaching Metropolitan teaching Non-metropolitan hospital |
11,342 (10.2) 98,829 (88.8) 1,096 (1) |
240 (11.8) 1,773 (87.2) 20 (1) |
0.41
|
|
Hypertension (%) |
21,738 (19.5) |
240 (11.8) |
<0.001 |
|
Diabetes mellitus (%) |
17,898 (16.1) |
475 (23.4) |
<0.001 |
|
Tobacco smoking (%) |
5,739 (5.2) |
118 (5.8) |
0.35 |
|
Peripheral vascular disease (%) |
1,359 (12.2) |
282 (13.9) |
0.13 |
|
CKD (%) |
33,372 (29.9) |
681 (33.5) |
0.03 |
|
Heart failure (%) |
81,308 (73.1) |
1,725 (84.8) |
<0.001 |
|
Atrial fibrillation (%) |
40,157 (36.1) |
681 (33.5) |
0.09 |
Outcomes
There was no difference in in-hospital mortality between patients with or without recent AMI (1.1% in both groups, adjusted odds ratio [aOR] 1.11, 95% CI: 0.62–1.99, P=0.72). There was no significant difference in ischemic CVA (aOR 1.22, CI 0.48–3.08, P=0.68), and valvular complications (aOR 0.98, CI 0.49–1.97, P=0.95). Major bleeding was higher among patients with recent AMI (aOR 1.97, CI 1.26–3.07, P=0.003) as well as the incidence of AKI (aOR 1.86, CI 1.52–2.27, P<0.001) (Figure 2). There were no patients with a recent AMI group who had conversion to open surgery, so the comparison was not feasible. TAVR for patients with a recent AMI was associated with longer hospital stay (adjusted mean difference [aMD] 0.96 days, CI 0.48 to 1.44, P<0.001) and higher total charges (aMD $15,489, CI 6,818–24,160, P<0.001). The primary and secondary outcomes are summarized in Table 2.
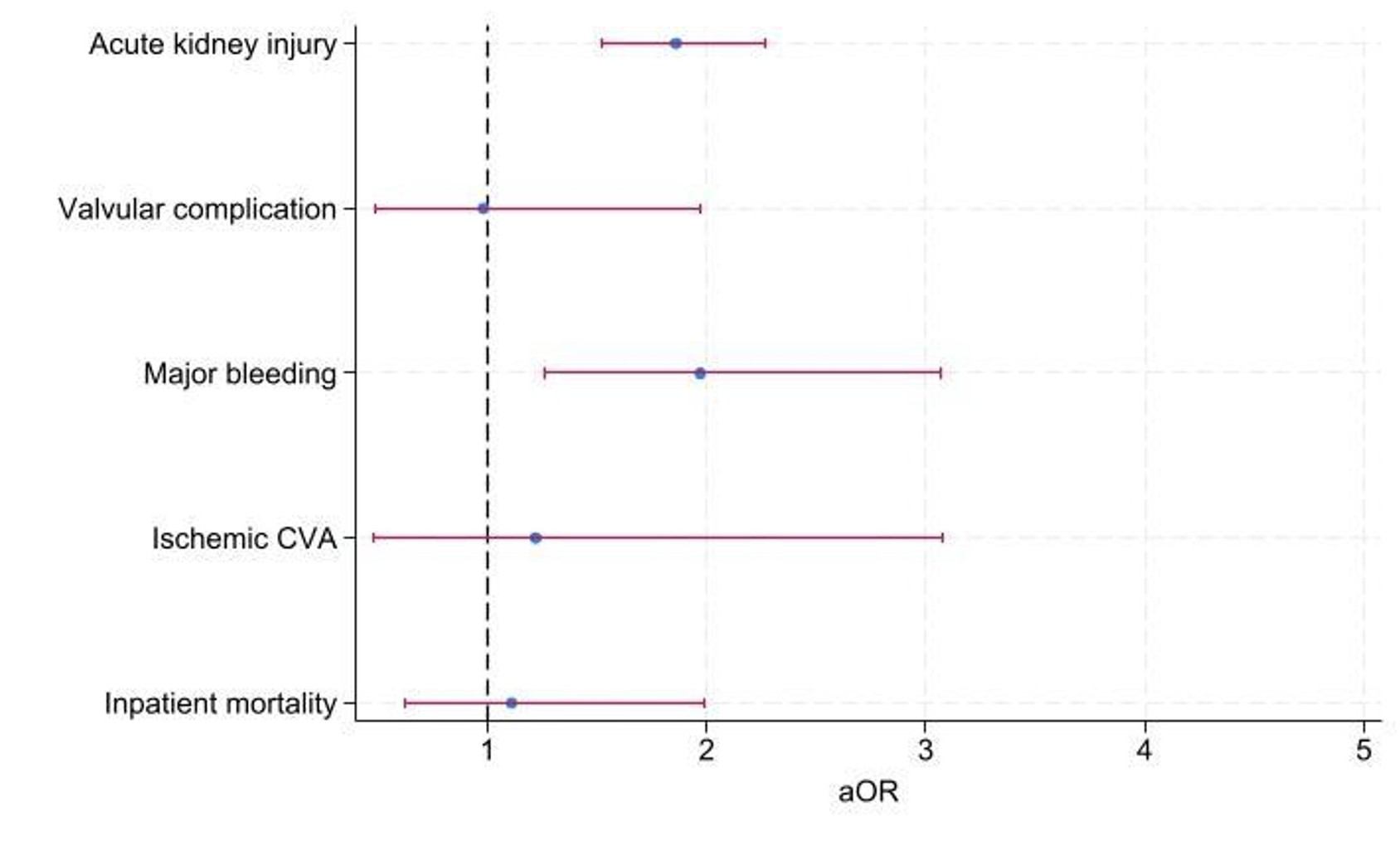
Figure 2. Forest plot of the primary and select secondary outcomes.
|
TAVR with no prior AMI N=111,267 |
TAVR with prior AMI N=2,033 |
aOR/aMD (95% CI) |
P-value |
|
|
Inpatient mortality (%) |
1,189 (1.1) |
22 (1.1) |
1.11 (0.62–1.99) |
0.72 |
|
Ischemic CVA (%) |
622 (0.6) |
11 (0.5) |
1.22 (0.48–3.08) |
0.68 |
|
Major bleeding (%) |
1,625 (1.5) |
56 (2.8) |
1.97 (1.26–3.07) |
0.003 |
|
Valvular complications (%) |
738 (0.7) |
12 (0.6) |
0.98 (0.49–1.97) |
0.95 |
|
Acute kidney injury (%) |
9,480 (8.5) |
309 (15.2) |
1.86 (1.52–2.27) |
<0.001 |
|
LOS in days, mean (SD) |
3.4 (0.1) |
4.4 (0.4) |
0.96 (0.48–1.44) |
<0.001 |
|
Total charges in US $, mean (SD) |
214,981 (±8,875) |
229,900 (±11,057) |
15,489 (6,818–24,160) |
<0.001 |
Subgroup analysis
Compared to those who had TAVR within 31–90 days after AMI, those who had TAVR within 30 days did not have a higher incidence of in-hospital mortality (aOR 0.4, 95% CI: 0.12–1.39, P=0.15), ischemic CVA (aOR 0.99, CI 0.12–8.24, P=0.99), or major bleeding (aOR 1.87, CI 0.76–4.57, P=0.16). However, earlier TAVR was associated with less valvular complications (aOR 0.2, CI: 0.05–0.78, P=0.02), increased AKI (aOR 1.72, CI: 1.18 - 2.5, P=0.005), and length of stay (aMD 1.22 days, 95% CI: 0.12–2.32, P=0.02) but not with a difference in total charges (aMD $16,521, CI: -281 to 33,324, P=0.05). The findings of the subgroup analysis were reported in Table 3. There was no difference in the outcomes based on the initial presentation (i.e., STEMI vs NSTEMI) (Supplemental Table 2 A/B). Similarly, there was no difference in the outcomes in the sensitivity analysis excluding patients who underwent transapical TAVR (Supplemental Table 3).
|
|
TAVR within 31-90 days of an AMI N=1,081 |
TAVR within 30 days of an AMI N=952 |
aOR/aMD (95% CI) |
P-value |
|
Inpatient mortality (%) |
15 (1.3) |
NR |
0.4 (0.12–1.39) |
0.15 |
|
Ischemic CVA (%) |
NR |
NR |
0.99 (0.12–8.24) |
0.99 |
|
Major bleeding (%) |
21 (1.9) |
35 (3.6) |
1.87 (0.76–4.57) |
0.16 |
|
Valvular complications (%) |
NR |
NR |
0.2 (0.05–0.78) |
0.02 |
|
Acute kidney injury (%) |
127 (11.7) |
181 (19) |
1.72 (1.18–2.5) |
0.005 |
|
LOS in days, mean (SD) |
3.7 (0.7) |
5.2 (0.5) |
1.22 (0.12–2.32) |
0.02 |
|
Total charges in US $, mean (SD) |
220,535 (31,870) |
240,520 (11,822) |
16,521 (-281–33,324) |
0.05 |
Discussion
In this nationwide observational analysis of TAVR procedures performed between 2016–2020, we found that a small proportion of TAVR procedures were performed among patients who had a recent AMI (median 31 days). Among patients with a recent AMI, we found that TAVR is generally safe and not associated with a higher incidence of in-hospital mortality. However, there was an association with a higher incidence of major bleeding and AKI. There was no difference in the outcomes whether the procedure was performed within 30 days versus 31–90 days. Additionally, there was no difference in outcomes based on the initial presentation (STEMI vs NSTEMI).
CAD is prevalent among patients with severe AS, with approximately half of those undergoing TAVR having CAD [9]. In our study, we found no significant difference in mortality compared to patients undergoing TAVR without prior AMI. We also found no significant differences in the outcomes between TAVR performed within 30 days versus 31-90 days. These findings suggest that performing TAVR within a few weeks after primary PCI may be reasonable, especially considering that delaying TAVR among symptomatic patients is associated with worse outcomes [10]. The incidence of major bleeding was higher among the recent AMI group, which might be related to the dual antiplatelet therapy. Additionally, patients with recent MI may have more comorbid conditions, such as chronic kidney disease, which are predictors of early severe bleeding post-TAVR [11].
Previous studies have shown that TAVR patients with CAD have comparable outcomes to those without CAD [6,12]. However, the timing of TAVR after AMI remains a knowledge gap. This study provides reassurance that the outcomes of TAVR up to 3 months after AMI are generally safe.
A major strength of this study rests in the use of the NRD, the largest all-payer readmission database in the United States, which allows for assessing outcomes in a real-world setting. However, the study has some limitations. Given the observational nature of the analysis, there is a potential for unmeasured confounding. To address this, we employed multilevel multivariate models in all analyses. Additionally, there have been some advancements in TAVR technology including newer devices and techniques since the time frame of this study. Last, the NRD is an administrative database and has inherent limitations. First, the database lacks other relevant clinical and echocardiographic details. Given the administrative nature of the database, there is a risk of coding errors. Furthermore, it does not track the same patient or hospital across multiple years, and each year represents a separate sample, which may limit longitudinal follow-up. Additionally, because participation varies across states and hospitals, the dataset may not fully capture all U.S. hospitalizations, potentially affecting the generalizability of the findings. Despite these limitations, this study is, to our knowledge, the largest to address this knowledge gap. Future studies using dedicated clinical registries, such as the Transcatheter Valve Therapy Registry, may provide more granular clinical, procedural, and echocardiographic data to further evaluate the relationship between recent AMI and TAVR.
Conclusions
In this nationwide observational analysis of TAVR procedures performed between 2016–2020, we found that a small proportion of TAVR procedures were performed among patients who had a recent AMI. Among patients with a recent AMI, we found that TAVR is generally safe and not associated with a higher incidence of in-hospital mortality, but a higher incidence of major bleeding and AKI. These findings could help inform decision making for TAVR among patients with a recent AMI.
Disclosure
None of the authors have any conflict of interest.
Funding
None.
References
2. Writing Committee Members; Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP 3rd, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Thorac Cardiovasc Surg. 2021 Aug;162(2):e183–353.
3. Di Gioia G, Bartunek J, Tesorio T, Vukcevic V, Aleksandric S, Dobric M, et al. Pathophysiology, Diagnosis, and Treatment of Patients with Concomitant Severe Aortic Stenosis and Coronary Artery Disease: A Closer Look to the Unresolved Perplexity. J Clin Med. 2021 Apr 11;10(8):1617.
4. Michail M, Davies JE, Cameron JD, Parker KH, Brown AJ. Pathophysiological coronary and microcirculatory flow alterations in aortic stenosis. Nat Rev Cardiol. 2018 Jul;15(7):420–31.
5. Beach JM, Mihaljevic T, Svensson LG, Rajeswaran J, Marwick T, Griffin B, et al. Coronary artery disease and outcomes of aortic valve replacement for severe aortic stenosis. J Am Coll Cardiol. 2013 Feb 26;61(8):837–48.
6. Kurmani S, Modi B, Mukherjee A, Adlam D, Banning A, Ladwiniec A, et al. Coronary artery disease and outcomes following transcatheter aortic valve implantation. Open Heart. 2024 Mar 29;11(1):e002620.
7. Thompson A, Fleischmann KE, Smilowitz NR, de Las Fuentes L, Mukherjee D, Aggarwal NR, et al. 2024 AHA/ACC/ACS/ASNC/HRS/SCA/SCCT/SCMR/SVM Guideline for Perioperative Cardiovascular Management for Noncardiac Surgery: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024 Nov 5;150(19):e351–442.
8. Halvorsen S, Mehilli J, Cassese S, Hall TS, Abdelhamid M, Barbato E, et al. 2022 ESC Guidelines on cardiovascular assessment and management of patients undergoing non-cardiac surgery. Eur Heart J. 2022 Oct 14;43(39):3826–924.
9. Faroux L, Guimaraes L, Wintzer-Wehekind J, Junquera L, Ferreira-Neto AN, Del Val D, et al. Coronary Artery Disease and Transcatheter Aortic Valve Replacement: JACC State-of-the-Art Review. J Am Coll Cardiol. 2019 Jul 23;74(3):362–72.
10. Roule V, Rebouh I, Lemaitre A, Sabatier R, Blanchart K, Briet C, et al. Impact of wait times on late postprocedural mortality after successful transcatheter aortic valve replacement. Sci Rep. 2022 Apr 8;12(1):5967.
11. Sun Y, Liu X, Chen Z, Fan J, Jiang J, He Y, et al. Meta-analysis of Predictors of Early Severe Bleeding in Patients Who Underwent Transcatheter Aortic Valve Implantation. Am J Cardiol. 2017 Aug 15;120(4):655–61.
12. Klinkhammer B. Transcatheter Aortic Valve Replacement After Coronary Artery Bypass Graft Is Associated With Increased Pacemaker Implantation but Not Reduced Overall Survival. Cardiol Res. 2018 Feb;9(1):40–5.