Abstract
Background: The aim of this study was to evaluate the long-term effects of continuous subcutaneous insulin infusion (CSII) therapy on the improvement of metabolic control and beta-cell function in patients with type 2 diabetes mellitus.
Methods: A single-center retrospective observational study was carried out in patients with T2DM who required CSII therapy due to suboptimal glycemic control. T2DM patients treated with the DANA-R Diabe care insulin pump (SOOIL Development Co., Ltd.) was followed for 6 years. Glucose control measures (hemoglobin A1c, total daily insulin dose) and beta-cell function (c-peptide index, insulinogenic index) were analyzed and clinical outcome data (body mass index, systolic/diastolic blood pressure, hemoglobin, serum protein, serum albumin, serum creatinine, total cholesterol, triglyceride, HDL, and LDL) were assessed after 2,4, and 6-years use of CSII. Statistical significances were calculated by Student’s paired t-test.
Results: Fifty-seven T2DM patients with mean age of 58.2±1.1 years were included in the study with 6 years (75.1±5.0 months) of follow-up. We found a statistically significant improvement in glycated hemoglobin (7.7±0.2, 6.3±0.1, 6.3±0.1, and 6.5±0.1%, p<0.0001), total daily insulin dose (48.6±3.1, 39.7±2.9, 34.2±2.0, and 34.4±2.2 U/day, p<0.0001), c-peptidogenic index (0.28±0.03. 0.42±0.07, 0.32±0.04, and 0.38±0.07, p<0.05), and insulinogenic index (3.28±0.53, 8.62±1.09, 6.47±0.74, and 5.84±0.96, p<0.001) after 2, 4, and 6-years use of CSII, respectively. Additionally, there were differences in clinical outcomes such as serum protein, albumin, total cholesterol, triacylglycerol, HDL, and LDL after 6 years.
Conclusions: The use of long-term CSII therapy for patients with T2DM led to a statistically significant and sustained improvement in glycemic control and beta-cell function.
Keywords
Long-term follow-up, T2DM, Glycemic control, Beta-cell function, Insulin pump
Introduction
Diabetes mellitus is a group of metabolic diseases characterized by hyperglycemia resulting from defects in insulin secretion, insulin action, or both. If left untreated, chronic hyperglycemia leads to long-term microvascular (e.g. nephropathy, retinopathy, and neuropathy) and macrovascular complications (e.g., coronary heart disease, peripheral arterial disease, and cerebrovascular disease). According to the Centers for Disease Control and Prevention (CDC), about 8.3% of the population in the United States, including children and adults, have diabetes, and its prevalence is likely to be increased to nearly 10 % by 2050. Type 1 diabetes mellitus (T1DM), which accounts for 5% to 10% of all diabetes cases, is an autoimmune disease characterized by the destruction of the pancreatic beta cells and insulin deficiency. Although T1DM can develop at any age, it usually occurs in children and young adults. Individuals with T1DM require daily insulin administration by injection or an insulin pump in order to survive, maintain glycemic control and normal body weight, and promote normal development in children. Type 2 diabetes mellitus (T2DM) is a progressive metabolic disorder characterized by insulin resistance and beta-cell dysfunction. T2DM accounts for 90% to 95% of all diabetes cases in adults in the United States and results from insulin resistance as well as impaired insulin secretion by the beta cells in the pancreas. As the disease progresses and the need for insulin rises, the pancreas can eventually lose its ability to produce insulin, necessitating insulin therapy [1].
Despite lifestyle changes and pharmacological interventions, many patients eventually require insulin therapy. Tight glycemic control is achieved with a combination of physiological basal and mealtime (prandial) insulins that mimic normal pancreatic function (i.e., peak less basal insulin secretion, rapid release of insulin in response to meals, and rapid clearance of the prandial insulin peak). These insulin therapies can be administered to patients by external continuous subcutaneous insulin infusion (CSII) via insulin infusion pump, which is intended to deliver insulin in a manner that most closely mimics the body’s physiologic release of insulin. CSII offers an alternative to multiple daily injections, aiming for more physiological insulin delivery and improved glucose control. CSII therapies divide insulin doses into basal, prandial and corrective elements, which can be adjusted independently to achieve optimal results. The use of CSII may improve treatment adherence, dosing accuracy, and lifestyle flexibility; however, it can also be technically demanding, costly, and requires a high level of engagement [2–6].
Randomized controlled trials (RCTs) of insulin pump therapy versus Multiple Daily Injections (MDI) have usually been conducted in volunteers with type 1 diabetes without specific clinical problems and not in those with persistent poor control on MDI. Meta-analyses and long-term observational studies show HbA1c improvements of 0.5–1.0% over several years [7,8]. Concerning the occurrence of hypoglycemia associated with the use of insulin-pumps, long-term CSII reduces the risk of severe hypoglycemia events, particularly in patients with a history of hypoglycemia unawareness or frequent episodes. Participants using CSII had a sustained reduction in hypoglycemia-related complications over 17 years and 75% reduction in severe hypoglycemia risk [9,10]. Further side effects of the insulin-pump therapy are associated with the insulin-injection as well as very rarely occurring ketoacidosis. A subgroup analysis of the DCCT study shows a minimal increased rate of ketoacidosis using an insulin-pump and CSII is associated with reduced risk of microvascular complications (retinopathy, nephropathy, and neuropathy) due to better long-term glycemic control [9]. Based on 22 studies involving individuals with type?1 diabetes, the meta-analysis found that total daily insulin dose was consistently lower with CSII versus MDI, and this was associated with improved glycemic control and reduced HbA1c without an increase in hypoglycemic events [11].
However, several observational short-term studies have shown a substantial reduction in HbA1c level on switching from MDI to CSII in Type 2 diabetes. The subjects in these observational reports have often been people who have had problems controlling their diabetes on MDI, and who have thus had higher HbA1c levels and/or insulin requirements at baseline, compared with the volunteers with type 2 diabetes in some of the RCTs of MDI vs CSII. This hints that the most appropriate evaluation and subsequently the clinical usage of CSII in Type 2 diabetes should be, in the first instance, in people deliberately selected as having failed to achieve adequate glycemic control on MDI [12].
Previous short-term studies have demonstrated the efficacy of CSII in type 2 diabetes; however, long-term data of T2DM, especially beyond 2–3 years, remain limited. This study aims to evaluate the impact of long-term CSII therapy on glycemic control, insulin requirements, beta-cell function, and general metabolic health in T2DM patients over a 6-year period.
Materials and Methods
Study design and participants
This was an analytical, retrospective observational study designed to evaluate the long-term effect of the use of CSII on clinical outcomes and metabolic control. Type 2 diabetic patients, aged 19 years and older, started CSII therapy using the DANA-R Diabetes care insulin pump (SOOIL Development Co., Ltd., Korea) at Diabetes Center at Konkuk University Chungju Hospital and subsequently monitored for at least five years were included in the study. Patients were included based on poor glycemic control with prior insulin regimens such as multiple daily injection therapy; submission of a written informed consent form with signatures and dates in accordance with Good Clinical Practice (GCP) and local laws. Exclusion criteria included uncontrollable hyperglycemia (HbA1c≥15% one year following insulin pump therapy); frequent hypoglycemia; myocardial infarction, stroke, or transient ischemic attack within the 3 months prior to CSII therapy; impaired liver or kidney function or cancer within the past 5 years; antidiabetic medication, anti-obesity medication, steroid, or thyroid hormone therapy; perimenopausal women with inadequate contraception, pregnant women, or breast-feeding mothers; alcohol or drug addicts; participants in other clinical trials. All study protocols were approved by the Institutional Review Board of Konkuk University Chungju Hospital.
Assessments
Baseline characteristics included age, gender, weight, BMI, systolic and diastolic blood pressure (BP), disease duration, and duration of insulin pump therapy. The beneficial effects of long-term insulin pump therapy were assessed by comparing glycemic control (HbA1c and total daily insulin dose) and beta-cell function (c-peptidogenic index and insulinogenic index) with the levels recorded at the initiation of CSII. Additionally, clinical and biochemical outcomes—including BMI, BP, hemoglobin, serum creatinine, total protein, albumin, total cholesterol, triglycerides, HDL, and LDL—were assessed. Blood samples were collected after an overnight fast (≥12 h) and 30 min after the ingestion (PP30 m) of a standard formulated mixed meal (500 kcal; 52.9% carbohydrate, 30.4% fat, and 16.7% protein). Insulin pump use was discontinued for at least 9 hours overnight prior to sampling. The indices were calculated as follows:
- c-peptidogenic index: (c-peptide at PP30 m – fasting c-peptide) / (plasma glucose at PP30 m – fasting plasma glucose)
- Insulinogenic index: (insulin at PP30 m – fasting insulin) / (plasma glucose at PP30m – fasting plasma glucose)
Statistical analysis
The appropriate sample size for parametric statistics was determined using G*Power to ensure a normal distribution. With an effect size of 0.5, α error of 0.05, power of 0.95 and one group set, a minimum of 45 participants were required. Normally distributed variables are expressed as mean ± standard error and changes in variables by therapies were compared using Student’s paired t-test. A two-sided p value of <0.05 was considered significant. Statistical analyses were performed using IBM SPSS Statistics version 29.0.1.0 (171) for Windows.
Results
Baseline characteristics
During the six years (75.1±0.5 months) of observational period, 57 subjects (mean age 58.2±1.1 years; 35.1% female) with a mean diabetes duration of 11.8±1.2 met the inclusion and exclusion criteria. The demographic characteristics of the participants at baseline are summarized in Table 1.
|
|
Mean ± SE or frequency |
Median |
Min, Max |
|
Age (years) |
58.2±1.1 |
58 |
40, 75 |
|
Female |
20 (35.1%) |
|
|
|
Disease duration (years) |
11.8±1.2 |
10 |
0.1, 35 |
|
Weight |
65.2±1.6 |
65 |
39, 94 |
|
Height |
162.5±0.9 |
162 |
147, 180 |
Effectiveness in glycemic control and β-cell function
Following treatment with CSII, HbA1c significantly decreased from 7.7±0.2% at baseline to 6.3±0.1% at 2 years and remaining stable at 6.4±0.1% at 4 years and 6.5±0.1% at 6 year (p<0.0001 for all timepoints; Table 2, Figure 1). And total daily insulin requirements also significantly decreased from 48.6±3.1 U/day to 34.2±2.2 U/day at 6 years (p<0.0001; Figure 2). Furthermore, CSII significantly increased c-peptidogenic index, a surrogate marker for β-cell function, from 0.28±0.03 to 0.38±0.07 at 6 years (p<0.05; Figure 2), indicating the partial preservation of endogenous insulin secretion. The insulinogenic index also increased from 3.28±0.53 to 5.84±0.96 at 6 years (p<0.001; Figure 3), demonstrating an enhanced early-phase insulin response. Overall, insulin pump therapy significantly improved glycemic control, TDD, and c-peptidogenic and insulinogenic index as compared to the levels observed at the initiation of insulin pump therapy.
|
|
Baseline |
Insulin pump therapy for |
|
|||||||
|
2 years |
4-years |
6-years |
|
|||||||
|
|
HbA1c (%) |
7.7±0.2 |
6.3±0.1d |
6.4±0.1d |
6.5±0.1d |
|||||
|
|
TDD (U/day) |
48.6±3.1 |
39.7±2.9b |
34.2±2.0 d |
34.2±2.2 d |
|||||
|
|
c-peptidogenic index |
0.28±0.03 |
0.42±0.07a |
0.32±0.04a |
0.38±0.07a |
|||||
|
|
Insulinogenic index |
3.28±0.53 |
8.62±1.09 d |
6.47±0.74 d |
5.84±0.96c |
|||||
|
|
All values are presented as mean ± standard error. HbA1c: Glycosylated Hemoglobin; TDD: Total Daily Insulin Dose. ap<0.05, bp<0.01, cp<0.001 or dp<0.0001 vs Baseline, analyzed using paired t-test |
|||||||||
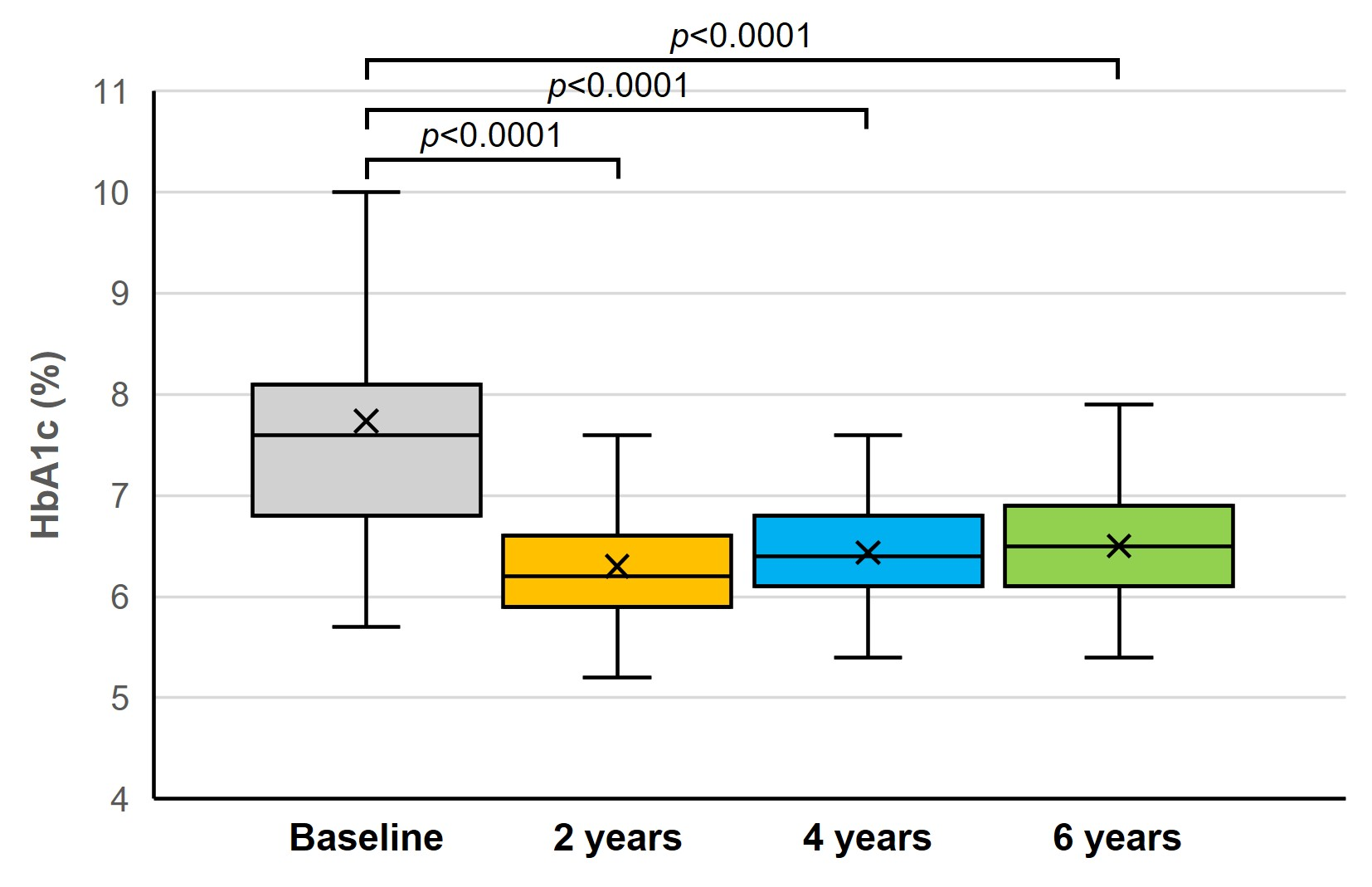
Figure 1. The effect of long-term insulin pump therapy on HbA1c of T2DM.
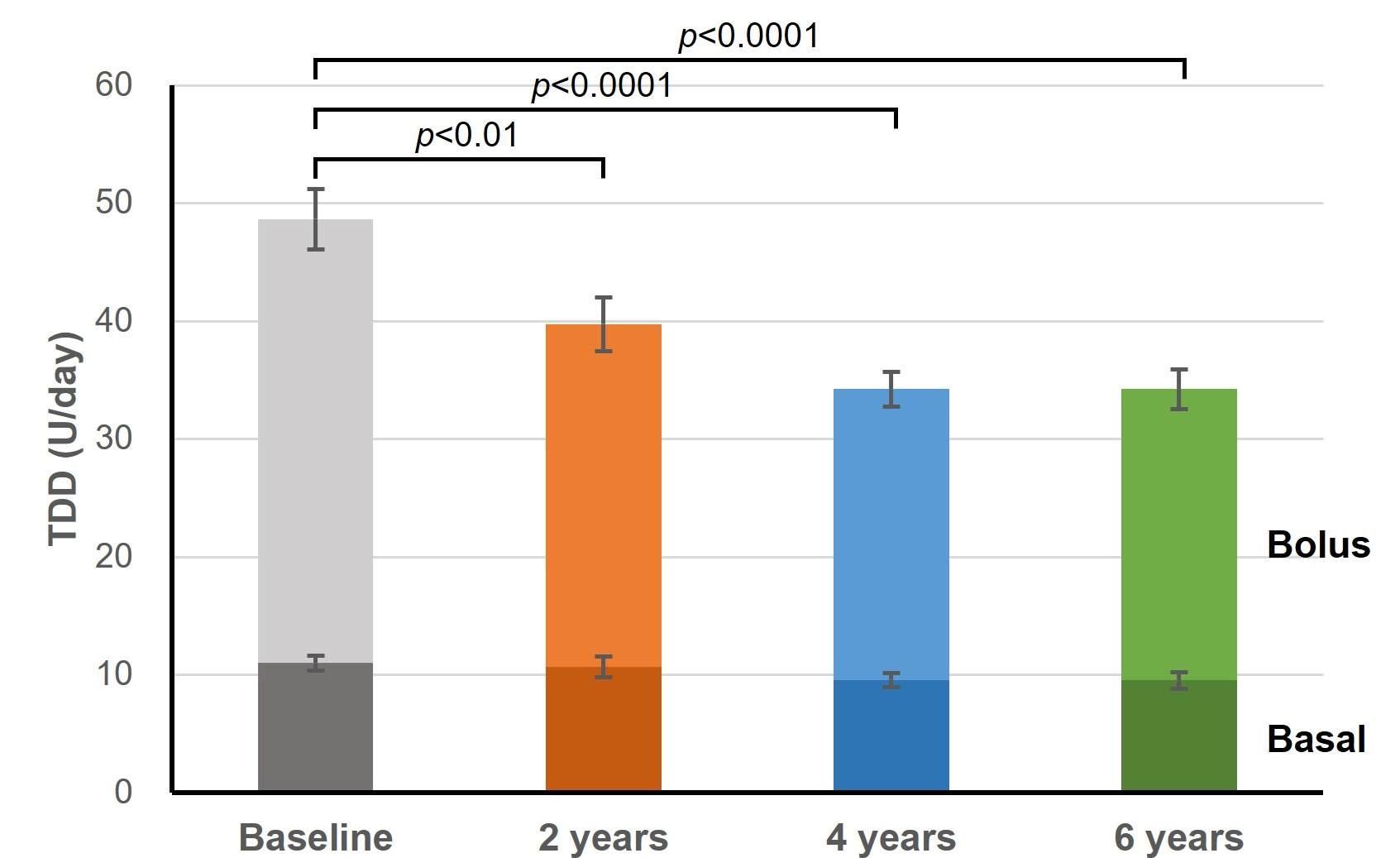
Figure 2. The effect of long-term insulin pump therapy on total daily insulin dose of T2DM.
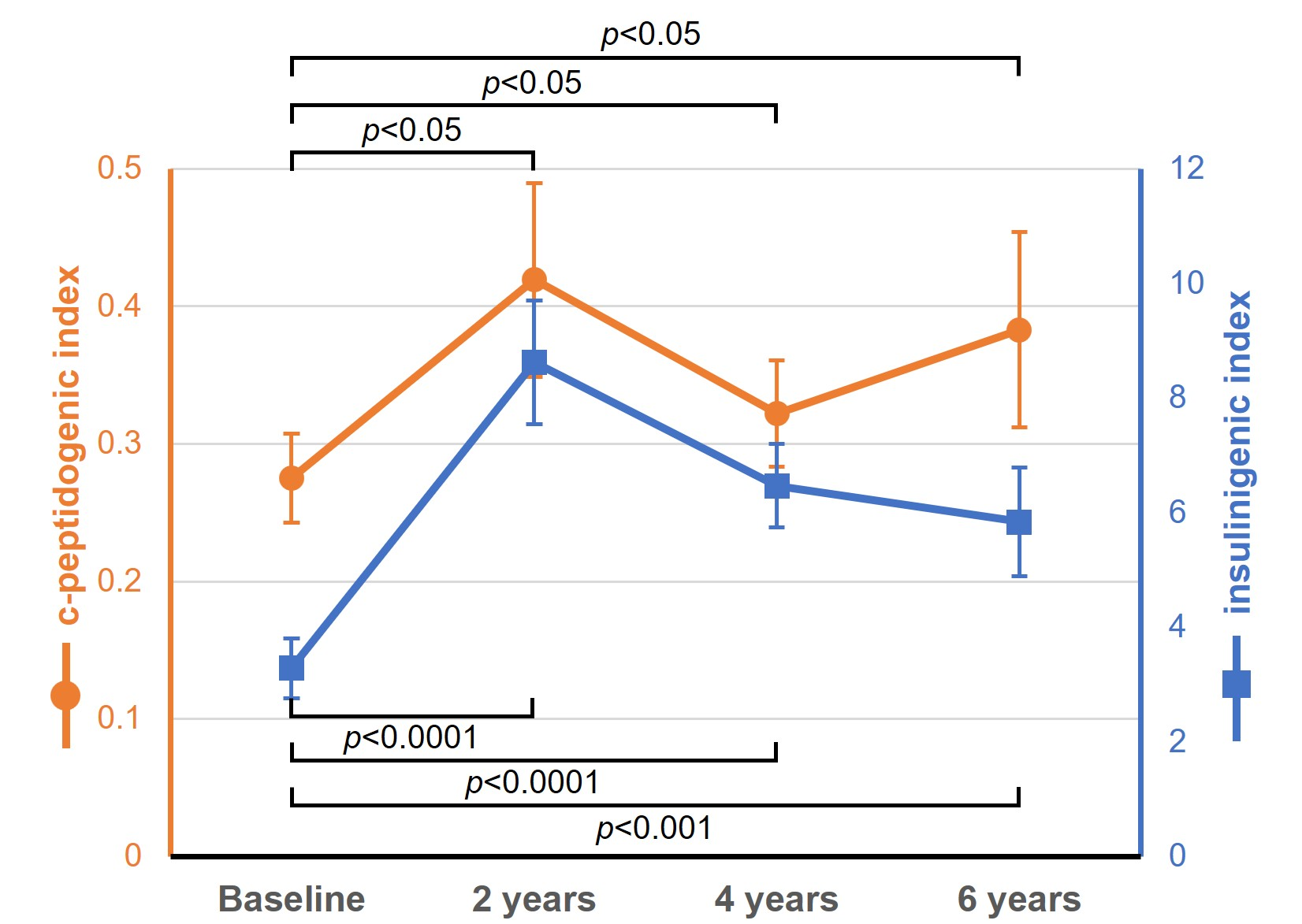
Figure 3. The effect of long-term insulin pump therapy on beta-cell function of T2DM.
Effect on clinical outcomes
Diastolic blood pressure decreased significantly from 75.9±0.9 to 69.1±10.6 mmHg (p<0.0001) (Table 3), while systolic pressure slightly increased (Figure 4a). Serum protein and albumin levels significantly increased (p<0.0001; Figure 4b), suggesting improved nutritional status. Lipid profiles showed improvements in HDL (from 43.7±1.3 to 50.4±1.8 mg/dL, p<0.001) and triglycerides (from 150.8±13.8 to 121.9±8.5 mg/dL, p<0.05), with modest increases in LDL and total cholesterol (Figure 4c). On the other hand, no significant change from baseline in BMI or serum creatinine were observed throughout the observation period (Figure 4d).
|
|
Baseline |
Insulin pump therapy for |
||
|
2-years |
4-years |
6-years |
||
|
Systolic BP (mmHg) |
125.8±1.4 |
121.9±1.4 |
122.9±1.7 |
131.0±2.5 |
|
Diastolic BP (mmHg) |
75.9±0.9 |
74.0±0.7 |
73.7±0.8 |
69.1±1.4d |
|
Serum protein (g/dl) |
6.63±0.06 |
7.05±0.05d |
7.09±0.06d |
7.11±0.06d |
|
Serum albumin (g/dl) |
3.96±0.04 |
4.24±0.03d |
4.25±0.04d |
4.20±0.03d |
|
Total cholesterol (mg/dl) |
163.9±4.4 |
176.8±5.9a |
169.2±3.7 |
186.5±5.5d |
|
Triglyceride (mg/dl) |
150.2±13.8 |
122.8±10.6a |
113.9±9.0b |
122.0±8.5a |
|
HDL (mg/dl) |
43.7±1.3 |
48.4±1.7c |
51.8±1.7 d |
50.4±1.8c |
|
LDL (mg/dl) |
94.6±4.0 |
107.5±5.2a |
97.2±3.4 |
108.5±4.5b |
|
BMI (kg/m2) |
24.6±0.5 |
25.4±0.5 |
25.4±0.5 |
25.0±0.5 |
|
Serum creatinine (mg/dl) |
0.80±0.03 |
0.84±0.03 |
0.84±0.03 |
0.97±0.03 |
|
Hemoglobin (g/dl) |
13.8±1.4 |
13.8±1.5 |
13.9±1.4 |
13.9±1.3 |
|
PP0m (mg/dL) |
133.8±5.7 |
121.1±3.2a |
129.0±3.1a |
135.3±4.3 |
|
PP30m (mg/dL) |
198.6±7.4 |
197.2±5.6 |
217.9±4.9a |
217.9±5.7* |
|
PP120m (mg/dL) |
275.1±11.2 |
263.5±10.8 |
288.5±9.3 |
293.8±10.4 |
|
All values are presented as mean ± standard error. BMI: Body Mass Index; BP: Blood Pressure; HDL: High Density Lipoprotein; LDL: Low Density Lipoprotein; PP: Postprandial. ap<0.05, bp<0.01, cp<0.001 or dp<0.0001 vs Baseline, analyzed using paired t-test. |
||||
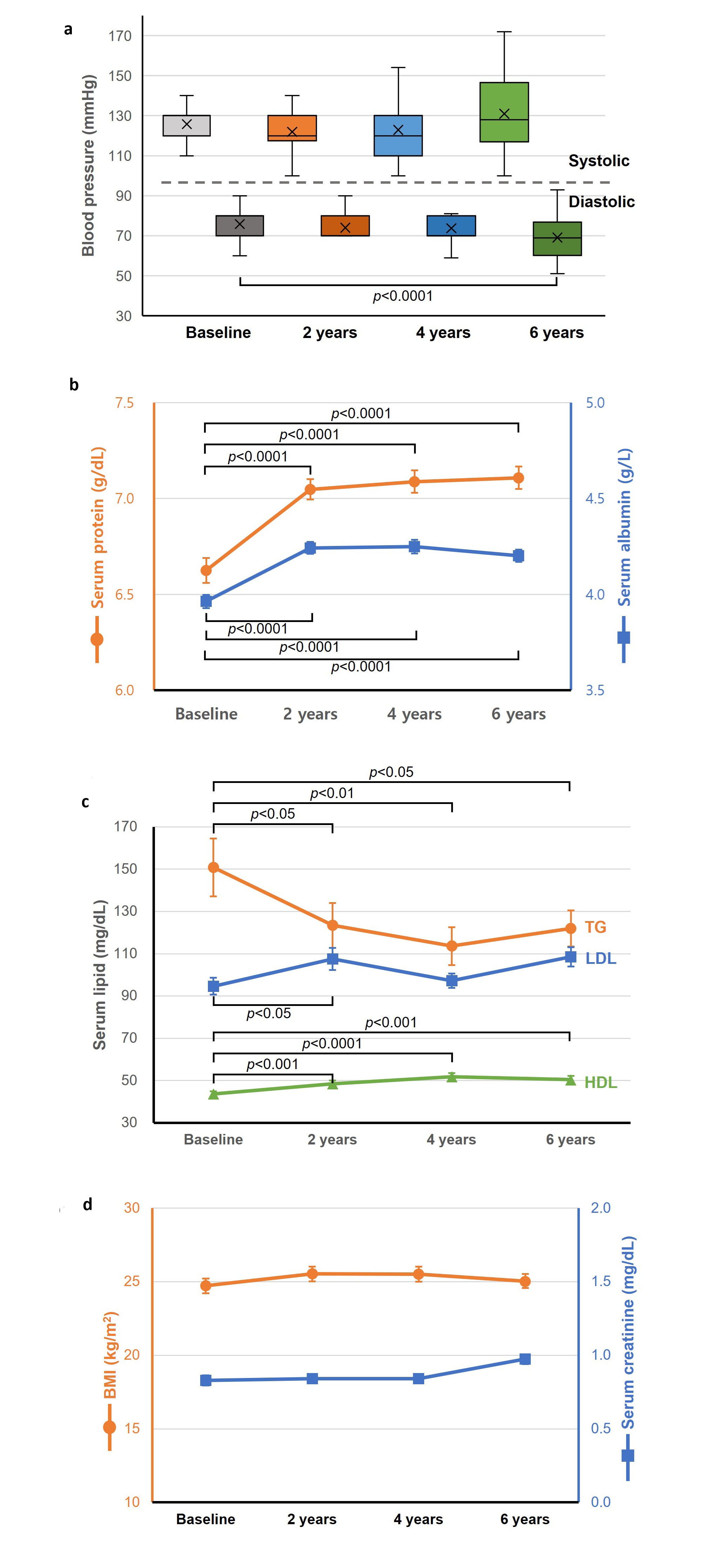
Figure 4. Comparison of clinical outcomes between baseline and post-insulin pump therapy (a) blood pressure, (b) Serum proteins, (c) Serum lipids, (d) BMI and creatinine.
Discussion
While the CSII therapy for T1DM has been shown to improve glycemic control without increasing the risk of hypoglycemia, but with weight loss and lower insulin requirements [13,14], the long-term efficacy of insulin pump therapy remained unexplored in T2DM. Therefore, our study aimed to investigate the efficacy of insulin pump therapy in patients with long-standing, inadequately controlled T2DM. In the present study, we observed that long-term CSII therapy improved glycemic control significantly, as evidenced by a reduction in HbA1c 7.7±0.2% at baseline to 6.5±0.1%, p<0.0001, Figure 1), reaching good glycemic control. In alignment with the reduction of TDD reported in previous research [15], we also observed that TDD decreased significantly compared to the baseline level (p<0.0001, Figure 2).
As we hypothesized, the long-term CSII therapy increased β-cell function assessed through c-peptidogenic index by 1.4 times (p<0.05; Figure 3a) compared with the baseline level. In addition, early-phase insulin response, as assessed by insulinogenic index, significantly increased following long term CSII therapy compared to the baseline levels (p<0.001; Figures 3b). These findings suggest that resolving glucotoxicity by lowering hyperglycemia may enhance both insulin sensitivity and β-cell function. Glucotoxicity is closely associated with insulin resistance and the deterioration of β-cell function, induced by increased apoptosis and reactive oxygen species in β-cells [16–18]. Indeed, previous reports have indicated that lowering plasma glucose levels with insulin therapy improves insulin sensitivity [19]. However, there are limited reports on the improvement of β-cell function through medical therapy, particularly in patients with long-standing T2D. Therefore, our present study is noteworthy in that long-term insulin pump therapy improved β-cell function with a reduction of TDD in patients with long-standing T2D who failed previously to achieve optimal glycemia.
There were several reasons why our patients with long-standing T2D might not achieve optimal glycemic control despite conventional therapy. These reasons include insufficient self-monitoring of blood glucose to accurately titrate insulin doses, irregularity of meal times or sleep patterns, lack of exercise, and inadequate nutrition. These factors can only be corrected through life style modifications, which should be accompanied in order to promote the efficacy of conventional therapy. However, given the association of chronic hyperglycemia with micro- and macrovascular diabetes complications, we reasoned that additional glucose-lowering interventions should be considered for our patients when their life style modifications were not inadequate. In that sense, long-term insulin pump therapy for T2DM was effective in reducing hyperglycemia in the present study.
This 6-year follow-up study demonstrates that CSII is effective in providing sustained glycemic control and improving beta-cell function in patients with T2DM. These results suggest that CSII may be a viable long-term therapeutic strategy for insulin-requiring T2DM patients, especially those with suboptimal control on conventional regimens. Notably, the reduction in insulin requirements despite maintained glycemic control suggests improved insulin sensitivity or beta-cell preservation. Improvements in HDL, triglycerides, and protein markers indicate broader metabolic and nutritional benefits. The major strengths of this study are its long follow-up duration and comprehensive metabolic profiling. However, limitations include its retrospective design, lack of a control group, and single-center nature.
Conclusion
CSII therapy over a 6-year period led to sustained improvements in glycemic control, reduced insulin requirements, and enhanced beta-cell function in T2DM patients. Additionally, positive effects on lipid and nutritional parameters were observed. CSII may be considered as a long-term therapeutic option in selected patients with T2DM.
Conflicts of Interest
Soobong Choi is a stock holder of the Soil Development CO, Ltd.
Data Availability
Soobong Choi had full access to all the data of the quantitative study, wrote and submitted the article for publication. The datasets used and/or analyzed during the current study are available from Soobong Choi on reasonable request.
References
2. Golden SH, Sapir T. Methods for Insulin Delivery and Glucose Monitoring in Diabetes: Summary of a Comparative Effectiveness Review. J Manag Care Pharm. 2012 Aug;18(6 Suppl): S1–17.
3. Raskin P, Bode BW, Marks JB, Hirsch IB, Weinstein RL, McGill JB, et al. Continuous subcutaneous insulin infusion and multiple daily injection therapy are equally effective in type 2 diabetes: a randomized, parallel-group, 24-week study. Diabetes Care. 2003 Sep;26(9):2598–603.
4. Reznik Y, Cohen O. Insulin pump for type 2 diabetes: use and misuse of continuous subcutaneous insulin infusion in type 2 diabetes. Diabetes Care. 2013 Aug;36 Suppl 2(Suppl 2): S219–25.
5. Heinemann L. Overcoming obstacles: new management options. Eur J Endocrinol. 2004 Oct;151 Suppl 2: T23-7; discussion T29–30.
6. Pickup JC, Sutton AJ. Severe hypoglycemia and glycaemic control in Type 1 diabetes: meta-analysis of multiple daily insulin injections compared with continuous subcutaneous insulin infusion. Diabet Med. 2008 Jul;25(7):765–74.
7. Weissberg-Benchell J, Antisdel-Lomaglio J, Seshadri R. Insulin pump therapy: a meta-analysis. Diabetes Care. 2003 Apr;26(4):1079–87.
8. Siebenhofer A, Jeitler K, Berghold A, Horvath K, Pieber TR. Severe hypoglycemia and glycaemic control in Type 1 diabetes: meta-analysis of multiple daily insulin injections compared with continuous subcutaneous insulin infusion. Diabet Med. 2009 Mar;26(3):311–2
9. Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ, et al. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005 Dec 22;353(25):2643–53.
10. Pickup J, Mattock M, Kerry S. Glycaemic control with continuous subcutaneous insulin infusion compared with intensive insulin injections in patients with type 1 diabetes: meta-analysis of randomized controlled trials. BMJ. 2002 Mar 23;324(7339):705.
11. Jeitler K, Horvath K, Berghold A, Gratzer TW, Neeser K, Pieber TR, et al. Continuous subcutaneous insulin infusion versus multiple daily insulin injections in patients with diabetes mellitus: systematic review and meta-analysis. Diabetologia. 2008 Jun;51(6):941–51.
12. Pickup JC. Banting Memorial Lecture 2014* Technology and diabetes care: appropriate and personalized. Diabet Med. 2015 Jan;32(1):3–13.
13. Borus JS, Laffel L. Adherence challenges in the management of type 1 diabetes in adolescents: prevention and intervention. Curr Opin Pediatr. 2010 Aug;22(4):405–11.
14. Korkmaz Ö, Demir G, Çetin H, Mecidov İ, Atik Altınok Y, Özen S, et al. Effectiveness of Continuous Subcutaneous Insulin Infusion Pump Therapy During Five Years of Treatment on Metabolic Control in Children and Adolescents with Type 1 Diabetes Mellitus. J Clin Res Pediatr Endocrinol. 2018 Jun 1;10(2):147–52.
15. Rosenstock J, Jelaska A, Frappin G, Salsali A, Kim G, Woerle HJ, et al. Improved glucose control with weight loss, lower insulin doses, and no increased hypoglycemia with empagliflozin added to titrated multiple daily injections of insulin in obese inadequately controlled type 2 diabetes. Diabetes Care. 2014 Jul;37(7):1815–23.
16. Wajchenberg BL. beta-cell failure in diabetes and preservation by clinical treatment. Endocr Rev. 2007 Apr;28(2):187–218.
17. Rossetti L, Giaccari A, DeFronzo RA. Glucose toxicity. Diabetes Care. 1990 Jun;13(6):610–30.
18. Shannon C, Merovci A, Xiong J, Tripathy D, Lorenzo F, McClain D, et al. Effect of Chronic Hyperglycemia on Glucose Metabolism in Subjects With Normal Glucose Tolerance. Diabetes. 2018 Dec;67(12):2507–17.
19. Garvey WT, Olefsky JM, Griffin J, Hamman RF, Kolterman OG. The effect of insulin treatment on insulin secretion and insulin action in type II diabetes mellitus. Diabetes. 1985 Mar;34(3):222–34.
