Abstract
Background: Aneurysmal bone cyst (ABC) is a benign but locally aggressive bone lesion. USP6 gene rearrangements have been proposed as a key diagnostic marker, detectable via fluorescence in situ hybridization (FISH). However, its clinical utility and correlation with recurrence remains unclear. This study evaluates USP6 FISH as a diagnostic tool and investigates whether USP6 positivity is associated with an increased recurrence risk.
Materials & methods: This retrospective study analyzed 87 patients with primary ABC at Klinikum rechts der Isar, Technical University of Munich (1991–2021). Histopathological and radiological criteria were used for diagnosis, with USP6 FISH testing performed on available specimens. Statistical analyses, including Kaplan–Meier recurrence estimates and Cox regression were conducted to assess the association between USP6 status and recurrence.
Conclusion: USP6 FISH is a highly specific but moderately sensitive tool for diagnosing primary ABC. While a positive USP6 result supports the diagnosis, clinical and histopathological correlation remains essential. Importantly, USP6 positivity showed no significant correlation with recurrence, suggesting that surgical factors play a larger role. These findings confirm the diagnostic relevance of USP6 FISH but indicate that recurrence risk should be assessed independently of molecular status.
Keywords
Aneurysmatic bone cyst, UPS-6, Recurrence aneurysmatic bone cyst
Introduction
The aneurysmal bone cyst (ABC) is a benign expansive giant cell rare bone tumour with an incidence rate of 0,14–0,32 pro 100.000 [1]. Commonly these lesions occur in the 1st 2 decades of life and are located mainly in long bones in half of the cases [1,2], as well as rarely in the spine and skull [1,3]. It is typically found within the metaphysis of long bones, with a typical fluid-fluid level cyst visible on MRI [4].
The lesion can radiologically be described as an eccentrically located multilobulated bone cyst with fluid-fluid levels separated with septae and a surrounding reactive thin bone shell [4,5]. Due to tits radiological aspect Jaffè and Lichtenstein coined in 1942 the “aneurysmal bone cyst” term [6]. Through epidemiological analysis show that males are more often affected than females independently from the age [3].
In a multicentric study on lesions with ABC morphology in children 70% of the cases were considered primary lesions, while in the other 30% a different underlying entity could be detected [7]. The most common primary bone lesions associated with ABC-like changes are giant cell tumor and chondroblastoma. The majority of the tumors with ABC-like chances demonstrate a higher presence of fluid-fluid levels in radiological exams [3].
Multiple thesis were published about the underlying pathophysiology of the ABC, from vascular genesis, to traumatic or reactive; only in the early 2000s a genetic pathophysiological background has been proposed.
If we retrace history:1969 Dabska and Buraczewski proposed a natural development of the ABC within 4 phases: the initial phase described as osteolysis, the growth phase with the progressive destruction of the bone, the stabilisation phase with formation of the typical osseus septa and the healing phase [8]. Afterwards in 1990 Campanacci, Lichtenstein and Ratcliffe proposed a traumatic aetiology as a response to local haemorrhage or fracture, describing the ABC as a reactive lesion rather than a true neoplasm [9].
Oliveira et al. could demonstrate in his histopathological study a gene rearrangement localized to t (16;17). This translocation is placed in the ubiquitin-specific protease 6 (USP-6) oncogene region, an important protein which is been regulated from the cadherin-11 (CDH11) promoter [10,11].
Interestingly no translocation were found in lesions with ABC-like changes, which contributes to the assumption that here the morphological features are rather from a reactive change. That is one of the reasons why our study decided to exclude all the lesions ABC-like changes to assure a more uniform data set.
This discovery is considered a milestone in the etiology of ABCs indicating that this lesion is a true neoplastic bone tumor. After 2004 others have confirmed this etiology. The upregulation of USP-6 induces a metalloproteinase inducing matrix production by activation of a nuclear factor k-light chain enhancer of activated B cells, which is the first step in the tumorgenesis [12,13]. The chromosomal aberration (16;17) (q22; p13) results in a fusion gene and overexpression of USP-6, a gene that was first discovered in 1992 as a potential oncogene in Ewing Sarcoma [13,15].
This translocation is hardly expressed in healthy patients except in testis and ovaries but overall, it is specific for tumor cells. In fact, Oliveira et al. demonstrated that in 69% of the patients in his study a positive fluorescence in situ hybridization (FISH) for this translocation could be detected [13,14,16].
Currently, the international accepted therapy for ABCs is curettage and grafting with autologous, allogeneic bone or cement and with or without adjuvant therapy [17]. The current adjuvant therapies, that are being used and described are cryotherapy, sclerotherapy, radionuclide ablation arterial embolization [18–20].
A reduced incidence of recurrence thanks to the use of the mentioned adjuvants is not yet proven [21,22].
Adjuvant therapies for ABCs are an important area of clinical practice. At our institution, standardized protocols are implemented for local adjuvant treatment following intralesional tumor resection. Commonly employed techniques include the use of a high-speed burr for intralesional tumor debulking, as well as chemical adjuvants such as hydrogen peroxide (H2O2), with or without the addition of 95% ethanol, for local tumor cell inactivation.
The recurrence of ABCs characterize predominantly lesions with a high cellular component, in fact a high osteid fraction was proven to be a good prognostic factor [23]. Recurrence incidence was importantly higher in specimens with a mitotic index of ³7 per 50 fields, presence of immature lace pattern and fibro myxoid-like areas [24,25]. On the clinical side: age <12 years and open phases are been associated with an increased risk of recurrence [21,26,27]. Following the WHO classification and guidelines an association with UPS-6 break apart and recurrence has not been proven yet. The analysis of the recurrence rate was the aim of this study.
Material and Methods
This retrospective study included 211 patients with a diagnosis of an aneurysmatic bone cyst treated at Klinikum rechts der Isar from 1991 to 2021. The local institutional review and ethics board (Klinikum rechts der Isar, Technical University of Munich) approved this study in 2020 (No. 48/20S).
All diagnosis were validated through the pathology department of the TUM Institution and Sarcoma Centre. Histology was obtained through CT scan-guided or open biopsy performed in our institution and discussed at our interdisciplinary sarcoma board following the WHO guidelines. Exclusion criteria were: insufficient clinical data, insufficient histopathologic material for FISH analysis. Lesions with ABC-like changes were also excluded to allow a more uniform patient collective. In total 87 patients were included.
All patients were operated at Klinikum rechts der Isar (Munich, Germany) with a curettage and grafting with bone or cement, in some case an osteosynthesis were performed to insure stability.
The operation technic will not be discussed and evaluated in this study.
Aftercare for all patients was performed every 26 weeks for the first 5 years and on annual check thereafter, according to the common practice of the German Musculoskeletal Tumour Society. Time between the operation and local recurrence were calculated (TR). All locations were included.
Histopathological evaluation
Tissue microarray (TMA) construction
For USP6 FISH analysis, tissue micro arrays (TMA) were constructed using formalin-fixed and paraffin-embedded tissue samples. Of each sample representative areas of interest were selected and cores with a diameter of 1.5 mm were extracted.
Fluorescence in situ hybridization (FISH)
FISH was performed using 2 µm paraffin embedded tissue sections, which were deparaffinized in xylol, dehydrated and then washed with Aqua dest. The tissue sections were treated with Heat Pretreatment Solution Citric for 15 min at 98°C, rinsed with Aqua dest (3 x 1 min) and air-dried. Next followed digestion with pepsin for 10 min at 37°C. After washing and dehydration the USP6-probes (ZytoLight ® SPEC USP6 Dual Color Break Apart Probe; ZytoVision GmbH, Germany) were applied, followed by denaturation for 10 min at 75°C and hybridization over night at 37°C. The slides were washed with TBS buffer and mounted with Vectashield with 4′,6-diamidino-2-phenylindole (DAPI).
For a positive FISH result, at least 15% of the lesional cells should show a clear spilt signal.
Statistical evaluation
Data were processed and analyzed using StatPlus Pro 2020 (AnalystSoft). Statistical reporting followed STROBE guidelines.
Descriptive statistic were performed using figures and tables. We calculated hazard ratios (HRs) to assess the relative risk of recurrence associated with UPS-6. Additionally, box plots were constructed to visually represent the distribution and variability of local recurrence across the study groups.
Statistical significance was set at p<0.05. Long Rank test were calculated, long with the 95% confidence interval. Cox regression analysis was performed to estimate the risk association between UPS-6 positivity and time to recurrence.
Results
The initial cohort of ABC diagnosis at Klinikum rechts der Isar (Munich) included 211 patients. In total 124 had to be excluded in the further analysis due to the following reasons: 9 patients were excluded due to mismatch in the sampling coding or missing reports. 16 patients were diagnosed with different entities showing ABC-like changes while 11 were classified as ABC but the diagnosis was later reviewed and another diagnosis was confirmed. To ensure good tissue quality for the further FISH analysis 83 cases with material earlier than 2010 were excluded and further 5 cases without sufficient tissue samples. In 25 performed FISH Analysis the USP-6 coloration wasn’t evaluable, so in conclusion a total of 62 patients could be enrolled.
The clinical characteristics are summarized in Table 1. The mean age was 19 years; the youngest patient was 4 years old, the oldest 62 years old.
|
|
n |
Percentage |
Mean |
Min |
Max |
|
Age |
87 |
- |
18 years |
4 years |
62 years |
|
females |
|
|
18 years |
4 years |
54 years |
|
males |
|
|
21 years |
9 years |
62 years |
|
Sex |
|
|
|
|
|
|
Male |
42 |
48,% |
- |
- |
- |
|
Female |
45 |
52% |
- |
- |
- |
|
Site |
|
|
|
|
|
|
Femur |
22 |
25% |
- |
- |
- |
|
Tibia |
19 |
22% |
- |
- |
- |
|
Foot |
12 |
14% |
- |
- |
- |
|
Hip |
16 |
18% |
- |
- |
- |
|
Spine |
2 |
2% |
- |
- |
- |
|
Humerus |
8 |
9% |
- |
- |
- |
|
Radius/Ulna |
5 |
6% |
- |
- |
- |
|
Hand |
3 |
3% |
- |
- |
|
|
USP-6 |
|
|
|
|
|
|
At diagnosis |
28 |
32% |
- |
- |
- |
|
Performed retrospectively |
59 |
68% |
- |
- |
- |
The main age among the female patients was 18 years (4–54 years). On the other hand, in the male cohort a mean age of 21 years was calculated, with a minimal age of 9 and a maximal age of 62.
All localizations were evaluated: in 76% of the patients the lower extremities were affected (25% femur, 19% Tibia, 18% hip and 12% in the foot). 2% were localized in the spine. The rest was localized in the upper extremity (8% humerus, 5% in radius and ulna, and 3% in the hand).
32% (28 patients) of the USP-6 analysis were performed at time of diagnosis. To ensure a critical analysis 59 (68%) new FISH with a USP-6 analysis were performed retrospectively and analyzed by two pathologists (one resident and one senior fellow).
We systematically analyzed the association between follow-ups and histopathological features to enhance our understanding of disease progression. Time of recurrence (TR) were calculated between the time of the operation and the time of the histopathological biopsy to confirm the recurrence. Every histopathological report was checked to confirm the recurrence. Median Time of recurrence was 18.5 months (4–210 months) and median time of follow-up of all the cohort was 208 months (24–422 months).
We divided the patients in two cohorts to analyse the incidence of recurrence between the ABC with a positive USP-6 FISH and a negative one. In total only 7 patients showed a recurrence (8 %). The patients with a positive USP6 break apart showed a median TR of 11 months and a median follow up of 215 months (4–407 months); on the other side, patients with a negative USP6 FISH showed a median TR of 38 months and a median follow up of 89 months (4–167 months) (Figure 1).
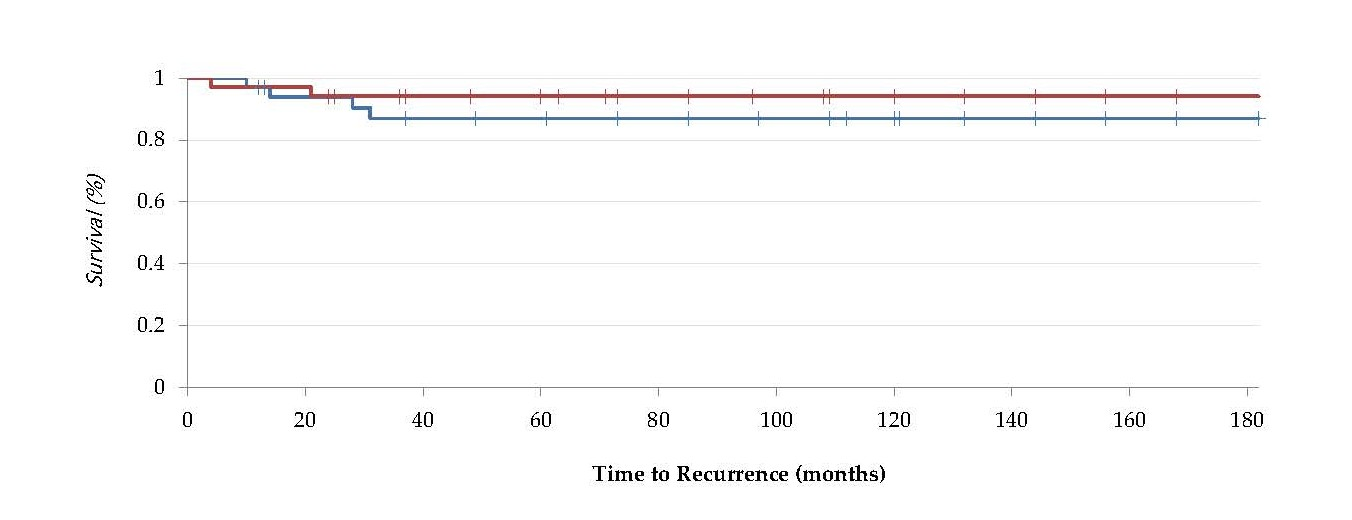
Figure 1. Box Plot analysis of the correlation between a positive USP-6 FISH (on the right) or a negative UPS-6 FISH (on the left) with TR (Time to Recurrence) expressed in months. No distinction in recurrence were made: in the negative cohort (n=30) the 5 patients that showed recurrence were included as well the 2 patients in the positive cohort (n=32).
From all 87 included patient, only 7 (8%) developed a recurrence in the analyzed time range. Of these 7 cases 5 were USP6 negative while 2 cases showed a positive USP-6 break apart. All the patients were treated with -curettage and plombage by our orthopedic surgeons’ team.
To assess the potential association between USP6 positivity and recurrence risk, we performed a log-rank test and calculated the hazard ratio. The long Rank Test showed a value of 0.774 (p-value of 0.37): given this data and the limited patient count we could not find any statistical relevant correlation.
Analyzing the Hazard Ratio between the two groups we calculated a risk value for a positive USP-6 FISH to encounter a recurrence of 2,10 (LCL 0,424-UCL 10,47 (95%), this higher risk could be explained due to the limited amount of recurrence in our cohort (Figure 2).
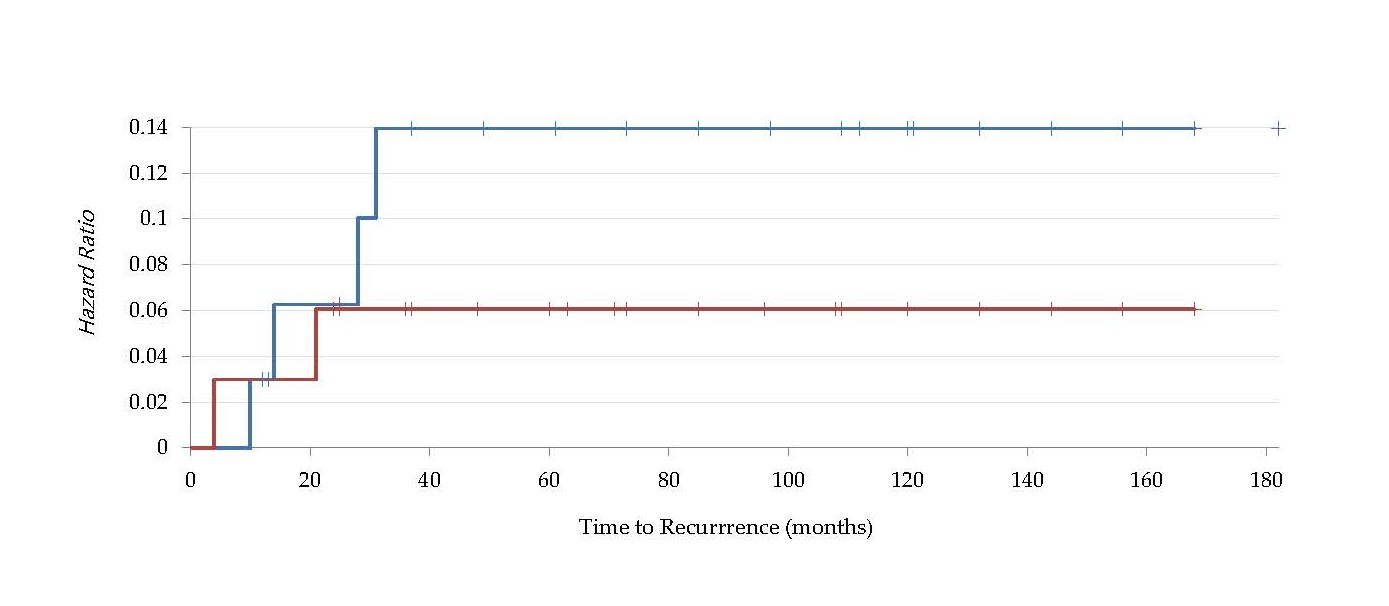
Figure 2. Kaplan-Meier Curve and Cumulative Hazard Ratio Curve for positive USP-6 specimens (red line) and negative USP-6 specimens (blue line).
Fish results
Due to the above stated exclusion criteria, 87 patients (41%) could be included in the FISH analysis. In 28 cases (32%) from these 87 patients USP-6 testing was performed directly within the initial diagnostic process. Furthermore 58 cases (67%) had available representative tissue samples from 2010 to 2021 to perform retrospective USP6-FISH evaluation. In one additional case USP-6 testing was performed at diagnosis but could not be evaluated initially due to technical issues, so here the USP-6 FISH was also repeated retrospectively.
38% of the FISH performed FISH analysis were evaluable (33 specimens), unfortunately 29% (25 specimens) were not evaluable, mostly due to poor signal quality of the probes and strong background reaction of the adjacent stroma. The representative pictures of the FISH examinations, which were performed at diagnosis, were also reevaluated. In total, in 62 specimens (71%) FISH results could be evaluated.
A cut-off value of 15% of observed break apart in the lesional spindle cells was required to be considered a UPS-6 test positive (Figure 3). The characteristics of the UPS-6 analysis are summarized in Table 2.
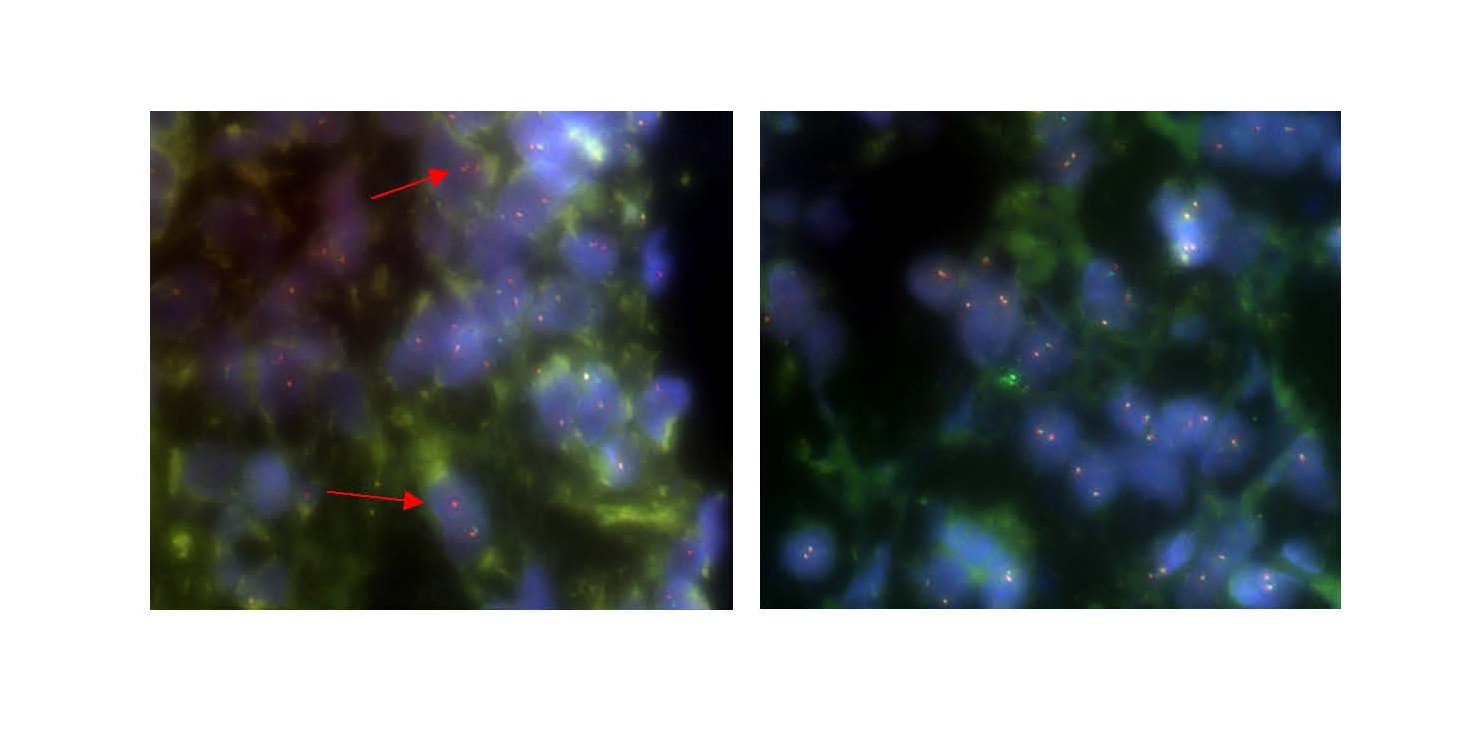
Figure 3. Photos of one positive FISH for USP-6 analysis (on the left) and one negative FISH for USP-6 analysis (on the right).
|
USP-6 |
n |
Breakapart |
|
Not evaluable |
25 (29%) |
- |
|
Evaluable |
62 (72%) |
|
|
Performed at diagnosis |
28 |
|
|
Performed retrospectively |
33 |
|
|
Performed at diagnosis and retrospectively |
1 |
|
|
Positive breakaparts |
|
32 (52%) |
|
Negative breakaparts |
|
30 (48%) |
Among the 62 evaluable samples, USP6 gene rearrangement was detected in 32 cases (52%), whereas 30 cases (48%) showed no evidence of a USP6 break apart. These findings highlight a substantial proportion of USP6 rearrangements among the tested cohort, supporting the utility of FISH as a diagnostic adjunct in the assessment of lesions with ABC morphology.
Discussion
ABC is now recognized as a clonal neoplasm, distinguished by recurrent USP6 gene rearrangements in some of primary cases [28]. The discovery of these translocations (most often t (16;17) (q22;p13) fusing CDH11 to USP6) cemented the neoplastic nature of ABC, overturning its former designation as a purely reactive lesion [1,2]. In the literature it is described that approximately 60–75% of primary ABCs harbor a USP6 rearrangement, whereas such alterations are absent in “secondary” ABC-like changes arising within other tumors [12,28].
A study by Li et al. evaluated the efficacy of USP6 break-apart FISH in differentiating primary ABCs from other morphologically similar lesions. In their analysis, they found that USP6 rearrangements were present in 53% (9 out of 17) of ABC cases. Importantly, none of the lesions with ABC-like changes, giant cell tumors (GCTs), or telangiectatic osteosarcomas (TOS) tested positive for USP6 rearrangements. This resulted in a specificity of 100% for USP6 break-apart FISH in diagnosing primary ABCs [29].
These findings confirm that the presence of a USP6 break-apart signal detected by FISH strongly supports the diagnosis of ABC and that this method demonstrates high specificity in this diagnostic context. In our series, roughly half of the tumors tested demonstrated the USP6 break-apart, a little lower incidence rate than the expected frequency in ABC. Notably, the remaining USP6-negative cases could still be confidently diagnosed as ABC on clinicopathological grounds, highlighting that a negative result does not exclude the disease. In fact, a recent study of jaw ABC found USP6 translocations in only 40% (4/10) of cases, underscoring that a substantial subset of ABCs will not show a USP6 break apart. The limited sensitivity of the assay on the order of 50–70% in practice means that USP6 FISH is best utilized as an adjunct rather than a stand-alone test [14].
Clinical and histopathological correlation remains paramount in diagnosing ABC, with molecular testing reserved for challenging cases. In routine practice, the characteristic radiographic appearance (expansile, lytic, septated lesion) with the histology (blood-filled cystic spaces with fibrous septa containing osteoclast-type giant cells and proliferating spindle cells) are often sufficient. We employed FISH primarily to confirm the diagnosis in equivocal cases or to distinguish primary ABC from lesions with ABC-like changes with a different underlying tumor. This approach aligns with other centers’ experience that the USP6 gene rearrangement test is a valuable confirmatory tool but not superior to careful clinicopathological evaluation. As Obermeier et al. noted, the correct diagnosis of ABC “can often only be made in combination with clinic, radiology, and histopathology,” with markers like USP6 serving to facilitate or confirm the diagnosis when needed [14]. Similarly, Novák et al. emphasize that while most ABCs can be recognized by their clinical and morphological presentation, molecular identification of USP6 fusions is necessary in ambiguous cases to distinguish true ABC from ABC-like changes in other bone lesions [14]. Additionally, the evaluation of USP6 FISH is challenging with only a few cells showing the break apart. Furthermore, fluorescence signals are often weak and background reaction from the adjacent stroma makes the evaluation even more difficult so that many cases cannot be evaluated sufficiently.
Beyond its diagnostic utility, the USP6 oncogene itself appears to drive the pathogenesis of ABC. Overexpression of USP6 (also known as TRE17) in translocation-positive ABCs has been implicated in the tumor’s locally aggressive behavior [30]. Functional studies have demonstrated that ectopic USP6 expression can induce a cascade of osteolytic and pro-inflammatory effects characteristic of ABC: for example, USP6 upregulates matrix metalloproteinases (MMP-9 and MMP-10) via NF-κB activation, consistent with the prominent proteolytic activity, bone destruction, and cystic degeneration seen in these lesions [28]. Concurrently, USP6 overexpression disrupts normal bone formation by blocking osteoblastic maturation. Chou and colleagues showed that TRE17/USP6 potently inhibits osteoblast differentiation in a ubiquitin-protease-dependent manner, partly by downregulating osteogenic signals (such as bone morphogenetic protein-4) and upregulating BMP antagonists [31]. The resulting attenuation of osteoblast activity is thought to contribute to the progressive bone loss and osteolysis observed in ABC. These insights into the molecular mechanism support the notion that USP6-rearranged ABCs behave as true neoplasms, with the USP6 fusion driving the cyst’s expansion through enhanced bone resorption and impaired bone repair. Importantly, however, this neoplastic behavior remains benign—USP6 activation confers local aggressiveness but not malignant potential [32].
One practical concern in ABC management is the risk of recurrence, which is reported in a significant fraction of cases after curettage. Recurrence rates of 20–30% are often cited, especially for incompletely excised lesions or those in anatomically challenging locations [32,33]. In contrast to the recurrence rates reported in the literature, the recurrence rate observed in our cohort is notably low (8%), which we attribute to our standardized surgical protocol involving double curettage combined with high-frequency burring and adjuvant treatment with hydrogen peroxide (H2O2), with or without the addition of 95% ethanol. We investigated whether the USP6 status of an ABC might correlate with its propensity to recur, given that USP6-positive lesions represent clonal neoplasms. Our data indicate no statistically significant correlation between USP6 positivity and recurrence. In our cohort, USP6-positive and USP6-negative ABCs showed virtually identical recurrence patterns. The median time to recurrence was shorter in USP6-positive cases (11 months) than in USP6-negative cases (38 months), but this difference was not significant (p- value: 0.37). Kaplan–Meier analysis confirmed overlapping recurrence-free survival curves for the two groups (log-rank p=0.77). The hazard ratio for recurrence in USP6-positive ABCs was ~2.10 (95% CI 0.424–10.47). Analyzing the HR the presence of the USP6 fusion could confer a measurable prognostic advantage or disadvantage in terms of recurrence risk, but the low recurrence rate precludes meaningful statistical comparison; therefore, no conclusions regarding the prognostic relevance of USP-6 status can be drawn from this cohort. This finding suggests that USP6 rearrangement status could be a driver of recurrence and that USP6-negative ABCs (which may represent either false-negative tests or ABCs driven by alternative genetic events) behave maybe less aggressively than USP6-positive ones. It is likely that clinical factors—such as the completeness of surgical removal and the lesion’s location—remain the primary determinants of ABC recurrence, but any intrinsic molecular difference should be further investigated. Indeed, ABC is well known to recur if residual tumor lining remains after surgery, regardless of USP6 status. Our results align with the clinical observation that all ABCs require adequate local control, and that even the “translocation-negative” cases should be treated and followed with the same vigilance as translocation-positive ones.
In conclusion, USP6 FISH emerges as a useful diagnostic adjunct for ABC that can confirm the lesion’s neoplastic nature, but it is not superior to standard clinical and histopathological assessment. A positive USP6 FISH greatly strengthens an ABC diagnosis and can help distinguish ABC from histologic mimics, yet the diagnosis should ultimately be grounded on the classical clinicoradiological and microscopic features. Taken together, these findings underscore that the value of USP6 testing in ABC lies in diagnosis rather than prognosis. While ongoing research into USP6’s role may eventually enable targeted therapies, at present the management of ABC should continue to rely on thorough surgical curettage and clinicopathological judgment, with molecular confirmation reserved for cases where diagnostic certainty is needed. The integration of USP6 FISH into the diagnostic toolkit thus enhances our confidence in diagnosing ABC without superseding the tried-and-true correlation of clinical presentation and histopathology in guiding patient care.
Conclusion
The findings of this study reinforce the diagnostic value of USP6 FISH in confirming aneurysmal bone cyst as a neoplastic entity. However, while USP6 rearrangement testing is highly specific, its limited sensitivity (~50–70%) highlights its role as a diagnostic adjunct rather than a standalone tool. Clinical, radiologic, and histopathological correlation remain indispensable in establishing a definitive diagnosis. Additionally, our analysis shows no statistically significant correlation between USP6 positivity and recurrence, indicating that USP6 status does not influence disease prognosis. Instead, recurrence risk is likely driven by clinical factors such as surgical completeness and anatomical location. Although recurrence did not occur significantly later in patients with negative USP6 status, this lack of statistical significance may be due to the overall low recurrence rate in our cohort. Therefore, the absence of a detectable effect should be interpreted with caution. Further multicenter studies with larger sample sizes would be desirable to validate our findings and better assess potential prognostic factors. Overall, USP6 FISH enhances diagnostic certainty but does not replace the fundamental role of clinicopathologic assessment in managing ABC. Future research should investigate the molecular mechanisms underlying USP6-driven pathogenesis and explore potential targeted therapies for ABC.
The retrospective design of our study inherently limits the control over data quality and completeness, introducing potential biases in data collection and interpretation. Second, the relatively small sample size may reduce the statistical power to detect significant differences or associations, particularly in subgroup analyses. These limitations must be considered when interpreting the findings, and they underline the need for prospective, multicenter studies with larger patient cohorts to confirm our results and further investigate prognostic factors.
Author Contributions
S.C.: Project administration, Conceptualization, Writing, Original Draft, Data Curation; F.S.: Original Draft, Data Curation, Writing, Review & Editing, Formal analysis; S.B. Review & Editing; U.L.: Review & Editing, R.v.E.-R.: Supervision, Validation; C.M.: Methodology, Supervision, Validation; C.K.: Methodology, Supervision, Validation, Review & Editing. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Ethics Committee of Klinikum rechts der Isar, Technical University of Munich (N°48/20S).
Informed Consent Statement
Not applicable. The paper is a retrospective study.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding authors (Dott. S. Consalvo, Felix Schicktanz). The data are not publicly available due to restrictions e.g., their containing information that could compromise the privacy of research participants.
Conflicts of Interest
The authors declare no conflict of interest.
Funding
Department of Orthopedics and Sports Orthopedic, Klinikum rechts der Isar, Technical University of Munich.
References
2. Leithner A, Machacek F, Haas OA, Lang S, Ritschl P, Radl R, et al. Aneurysmal bone cyst: a hereditary disease? J Pediatr Orthop B. 2004 May 1;13(3):214–7.
3. Zehetgruber H, Bittner B, Gruber D, Krepler P, Trieb K, Kotz R, et al. Prevalence of aneurysmal and solitary bone cysts in young patients. Clin Orthop®. 2005 Oct 1; 439:136–43.
4. Tsukamoto S, Errani C, Facchini F, Papagelopoulos P, Mavrogenis AF. Fluid-fluid levels in musculoskeletal tumor imaging. Curr Med Imaging Rev. 2021 Feb 1;17(2):157–65.
5. Van Dyck P, Vanhoenacker FM, Vogel J, Venstermans C, Kroon HM, Gielen J, et al. Prevalence, extension and characteristics of fluid-fluid levels in bone and soft tissue tumors. Eur Radiol. 2006 Dec;16(12):2644–51.
6. Jaffe HL, Lichtenstein L. Solitary unicameral bone cyst: with emphasis on the roentgen picture, the pathologic appearance and the pathogenesis. Arch Surg. 1942 Jun 1;44(6):1004–25.
7. Cottalorda J, Kohler R, de Gauzy JS, Chotel F, Mazda K, Lefort G, et al. Epidemiology of aneurysmal bone cyst in children: a multicenter study and literature review. J Pediatr Orthop B. 2004 Nov 1;13(6):389–94.
8. Dabska M, Buraczewski J. Aneurysmal bone cyst. Pathology, clinical course and radiologic appearances. Cancer. 1969 Feb;23(2):371–89.
9. Campanacci M. Bone and soft tissue tumors: clinical features, imaging, pathology and treatment. Spr Sci & Busi Me; 2013 Jun 29.
10. Oliveira AM, Hsi BL, Weremowicz S, Rosenberg AE, Cin PD, Joseph N, et al. USP6 (Tre2) fusion oncogenes in aneurysmal bone cyst. Cancer Res. 2004 Mar 15;64(6):1920–3.
11. Oliveira AM, Chou MM. USP6-induced neoplasms: the biologic spectrum of aneurysmal bone cyst and nodular fasciitis. Hum Pathol. 2014 Jan 1;45(1):1–11.
12. Ye Y, Pringle LM, Lau AW, Riquelme DN, Wang H, Jiang T, et al. TRE17/USP6 oncogene translocated in aneurysmal bone cyst induces matrix metalloproteinase production via activation of NF-κB. Oncogene. 2010 Jun;29(25):3619–29.
13. Oliveira AM, Chou MM. The TRE17/USP6 oncogene: a riddle wrapped in a mystery inside an enigma. Front Biosci Schol. 2012 Jan 1;4(1):321–34.
14. Obermeier KT, Schmöckel E, Otto S, Haidari S. Frequency of translocation of USP‐6 in the aneurysmal bone cyst of the jaw. J Oral Pathol Med. 2023 Feb;52(2):145–9.
15. Panoutsakopoulos G, Pandis N, Kyriazoglou I, Gustafson P, Mertens F, Mandahl N. Recurrent t (16;17) (q22; p13) in aneurysmal bone cysts. Genes Chromosomes Cancer. 1999 Nov;26(3):265–6.
16. Oliveira AM, Perez-Atayde AR, Inwards CY, Medeiros F, Derr V, Hsi BL, et al. USP6 and CDH11 oncogenes identify the neoplastic cell in primary aneurysmal bone cysts and are absent in so-called secondary aneurysmal bone cysts. Am J Pathol. 2004 Nov;165(5):1773–80.
17. Ozaki T, Hillmann A, Lindner N, Winkelmann W. Cementation of primary aneurysmal bone cysts. Clin Orthop Relat Res. 1997 Apr;(337):240–8.
18. Peeters SP, Van der Geest IC, de Rooy JW, Veth RP, Schreuder HW. Aneurysmal bone cyst: the role of cryosurgery as local adjuvant treatment. J Surg Oncol. 2009 Dec 15;100(8):719–24.
19. Gettleman BS, Padilla AN, Kumar S, Wren TAL, Miller J, Pawel BR, et al. Use of Surgical Adjuvants Does Not Decrease Recurrence of Aneurysmal Bone Cysts in Surgical Intervention with Pediatric Patients. J Pediatr Orthop. 2024 Jan 1;44(1): e79–e83.
20. Varshney MK, Rastogi S, Khan SA, Trikha V. Is sclerotherapy better than intralesional excision for treating aneurysmal bone cysts? Clin Orthop Relat Res. 2010 Jun;468(6):1649–59.
21. Bush CH, Adler Z, Drane WE, Tamurian R, Scarborough MT, Gibbs CP. Percutaneous radionuclide ablation of axial aneurysmal bone cysts. AJR Am J Roentgenol. 2010 Jan;194(1): W84–90.
22. Rossi G, Mavrogenis AF, Rimondi E, Ciccarese F, Tranfaglia C, Angelelli B, et al. Selective arterial embolisation for bone tumours: experience of 454 cases. Radiol Med. 2011 Aug;116(5):793–808.
23. Docquier PL, Delloye C, Galant C. Histology can be predictive of the clinical course of a primary aneurysmal bone cyst. Arch Orthop Trauma Surg. 2010 Apr;130(4):481–7.
24. de Silva MV, Raby N, Reid R. Fibromyxoid areas and immature osteoid are associated with recurrence of primary aneurysmal bone cysts. Histopathology. 2003 Aug;43(2):180–8.
25. Brindley GW, Greene JF Jr, Frankel LS. Case reports: malignant transformation of aneurysmal bone cysts. Clin Orthop Relat Res. 2005 Sep; 438:282–7.
26. Lin WC, Wu HT, Wei CJ, Chang CY. Aneurysmal bone cyst arising from fibrous dysplasia of the frontal bone (2004:2b). Eur Radiol. 2004 May;14(5):930–2.
27. Başarir K, Pişkin A, Güçlü B, Yildiz Y, Sağlik Y. Aneurysmal bone cyst recurrence in children: a review of 56 patients. J Pediatr Orthop. 2007 Dec;27(8):938–43.
28. Lau AW, Pringle LM, Quick L, Riquelme DN, Ye Y, Oliveira AM, et al. TRE17/ubiquitin-specific protease 6 (USP6) oncogene translocated in aneurysmal bone cyst blocks osteoblastic maturation via an autocrine mechanism involving bone morphogenetic protein dysregulation. J Biol Chem. 2010 Nov 19;285(47):37111–20.
29. Li L, Bui MM, Zhang M, Sun X, Han G, Zhang T, et al. Validation of Fluorescence in situ Hybridization Testing of USP6 Gene Rearrangement for Diagnosis of Primary Aneurysmal Bone Cyst. Ann Clin Lab Sci. 2019 Sep;49(5):590–7.
30. Legrand M, Jourdan ML, Tallet A, Collin C, Audard V, Larousserie F, et al. Novel partners of USP6 gene in a spectrum of bone and soft tissue lesions. Virchows Arch. 2021 Jul;479(1):147–156.
31. Balko J, Golas W, Kaspar L, Krskova L, Strnadova M, Kotis J, et al. Novel and unusual USP6 fusion partners in aneurysmal bone cyst and their role in pathogenesis and histopathological evaluation of this disease. J Clin Pathol. 2025 May 25;78(6):399–403.
32. Hiemcke-Jiwa LS, van Gorp JM, Fisher C, Creytens D, van Diest PJ, Flucke U. USP6-Associated Neoplasms: A Rapidly Expanding Family of Lesions. Int J Surg Pathol. 2020 Dec;28(8):816–25.
33. Bavan L, Eastley N, Stevenson J, Mifsud M, Bayliss L, Mahmoud S, et al. Aneurysmal bone cysts: A UK wide tumor center experience. J Surg Oncol. 2024 Mar;129(3):601–8.
