Abstract
Introduction and Objective: Semi-compliant (SC) balloons are essential in PCI for lesion preparation before stent deployment. The Optima SC PTCA balloon catheter, a next-generation device, was evaluated in this post-marketing, multicenter study to assess its real-world performance and safety.
Methods: This was a retrospective, observational study conducted across four interventional cardiology centers in India. A total of 183 adult patients with coronary artery disease who underwent PCI with Optima SC balloon pre-dilatation were included. Procedural and device success, adverse events, and operator-assessed device performance were evaluated. Patients were followed up to 3 months, as the time frame was adequate to establish procedural safety and performance of this PTCA catheter.
Results: Device and procedural success were achieved in 179 out of 183 patients (97.8%); failure in 2.2% was reported in complex lesions. No major adverse cardiac events (MACE), serious adverse events (SAE), or device-related complications were reported during hospital stay, though two clinical events (angina and target lesion failure) were reported in two patients during follow-up. Operator assessment rated performance satisfactory or better in >90% of cases across all parameters.
Conclusion: The Optima SC semi-compliant PTCA balloon catheter demonstrated a high procedural success rate and an excellent short-term safety profile in a real-world clinical setting. These findings support its use as a reliable and effective tool for coronary lesion pre-dilatation during PCI.
Keywords
Percutaneous coronary intervention, Coronary artery disease, Lesion preparation, Semi-compliant balloon, Balloon catheter, Optima SC, Device safety
Introduction
Percutaneous transluminal coronary angioplasty (PTCA), a cornerstone of percutaneous coronary intervention (PCI), remains a vital therapeutic strategy in the management of coronary artery disease (CAD). Since its inception more than four decades ago, PTCA has significantly evolved, offering a minimally invasive solution to re-establish myocardial perfusion in patients with obstructive atherosclerotic lesions [1,2]. Despite the widespread adoption and success of PCI, the procedure is not without risk. Post-procedural complications such as coronary artery dissection, perforation, target lesion failure (TLF), and the need for target vessel revascularization (TVR) remain relevant concerns [3,4]. Therefore, continued innovation in angioplasty tools, particularly balloon catheter systems, plays a critical role in enhancing procedural safety and long-term outcomes.
Balloon catheters serve as the primary interventional tools in coronary revascularization, employed during both pre-dilatation and post-dilatation phases of PCI [5]. The performance of these devices is highly dependent on their physical and mechanical properties, including crossing profile, inflation pressure, trackability, and lesion conformity [6]. The choice between semi-compliant and non-compliant balloons is dictated by lesion morphology and procedural goals. Semi-compliant (SC) balloons, in particular, offer a favorable balance between flexibility and pressure responsiveness, making them ideal for controlled vessel expansion and optimal stent deployment.
The Optima SC-Semi Compliant PTCA balloon catheter (Translumina Therapeutics Private Limited) represents a next-generation semi-compliant balloon catheter, engineered with advanced technologies to improve deliverability, inflation control, and procedural success. The Optimal SC-Semi Compliant PTCA Balloon Catheter features a low crossing profile, high burst pressure tolerance, and enhanced folding characteristics for precise and reproducible inflation. These attributes are designed to facilitate safe navigation and deliver consistent lesion preparation prior to stenting.
In light of these innovations, a post-market clinical follow-up study was conducted to evaluate the real-world safety and performance of the Optima SC-Semi Compliant PTCA balloon catheter in patients undergoing PCI. The findings aim to contribute to the growing body of evidence supporting the use of advanced SC balloon systems in contemporary interventional cardiology practice.
Methods
Study design
This study was designed as a retrospective, observational, multicenter, single-arm post-marketing clinical follow-up investigation to evaluate the safety and performance of the Optima SC semi-compliant PTCA balloon catheter. The study was conducted at four interventional cardiology centers across India between January and June 2024. The study protocol was reviewed and approved by the respective Institutional Ethics Committees (IEC) of all participating sites. Given the retrospective nature of the analysis, a waiver of informed consent was granted by the respective IECs. The study was conducted in compliance with the principles outlined in the Declaration of Helsinki, the International Conference on Harmonisation Good Clinical Practice (ICH-GCP) guidelines, and applicable national regulatory requirements.
Study population
A total of 183 adult patients (aged ≥18 years) who underwent percutaneous coronary intervention with the use of the Optima SC semi-compliant PTCA balloon catheter were included in the analysis. All patients had documented coronary artery disease with stenotic lesions in native coronary arteries and had received balloon pre-dilatation with the investigational devices as part of their PCI procedure. Patients who required additional plaque modification before SC balloon use and those with incomplete medical records were excluded.
Study device
The Optima SC semi-compliant PTCA balloon catheter is a CE certified, double-lumen coronary balloon dilatation catheter designed on a rapid exchange delivery platform with a balloon near the distal tip that can be inflated to dilate the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion, coronary artery occlusion, and balloon dilation of a stent after implantation. The Optima SC semi-compliant PTCA balloon catheter features a dual-lumen design for inflation and advancement, incorporating Trac+™ patented pre-molded tip technology and a balloon made of Grilamid® and Pebax® materials over the inner lumen. The catheter’s proximal part is made of a stainless-steel hypotube with two radiopaque marker bands for positioning. The proximal end has a luer hub for inflating the balloon, connected to a strain relief tube. The distal part of the Optima SC balloon benefits of a hydrophilic coating to facilitate access to the lesion site. The catheter is available in balloon diameters ranging from 1.50 mm to 5.00 mm and lengths from 6 mm to 50 mm, with an overall working length of 140 cm.
Study endpoints
The primary endpoint of the study was procedural success, defined as the successful delivery of the catheter to and across the target lesion, successful balloon inflation and deflation, and withdrawal of the device without complications, along with the final achievement of thrombolysis in myocardial infarction (TIMI) grade 3 flow in the treated vessel. Secondary endpoints included the incidence of device- or procedure-related complications, device success rate (defined as residual stenosis <50% with successful balloon withdrawal), and clinical success at up to 3 months post-procedure, determined by freedom from patient-oriented composite endpoints such as all-cause death, myocardial infarction, target lesion revascularization (TLR), or target vessel revascularization (TVR).
Data collection
All clinical data were abstracted retrospectively from patient medical records and documented in standardized case report forms (CRFs) by trained personnel. Adverse events (AEs) and serious adverse events (SAEs), as well as in-hospital and follow-up outcomes, were recorded for analysis. Data was reviewed for consistency and completeness before inclusion in the final dataset.
Statistical analysis
Statistical analysis was performed using SPSS version 24.0 (IBM Corp., Armonk, NY, USA). Continuous variables were presented as mean ± standard deviation (SD), median, and range, depending on data distribution. Categorical variables were summarized as counts and percentages. Descriptive statistics were used to report baseline characteristics and device performance metrics. Statistical significance was defined as a p-value <0.05 for all tests.
Results
Baseline characteristics
A total of 179 patients were included in the final analysis. The baseline demographic and clinical characteristics are summarized in Table 1. The cohort comprised predominantly male patients (n=131, 73.18%) with a mean age of 59.67 ± 10.94 years. Common cardiovascular risk factors included hypertension in 72 patients (40.22%), diabetes mellitus in 55 (30.73%), smoking history in 32 (17.88%), and dyslipidemia in 44 (26.35%). A family history of coronary artery disease was reported in 27 patients (15.61%), and severe left ventricular systolic dysfunction (LVEF<30%) was observed in 13 patients (7.26%).
|
Baseline Characteristics |
n=179 |
|
Age (year), Mean ± SD |
59.67 ± 10.94 |
|
Male, n (%) |
131 (73.18) |
|
Hypertension, n (%) |
72 (40.22) |
|
Diabetes, n (%) |
55 (30.73) |
|
Smoking, n (%) |
32 (17.88) |
|
Dyslipidemia, n (%) |
44 (26.35) |
|
Severely reduced LVEF, n (%) |
13 (7.26) |
|
History of CAD, n (%) |
27 (15.61) |
|
Heart Rate (bpm), Mean ± SD |
81 ± 17.64 |
|
Diastolic Blood Pressure, Mean ± SD |
81.49 ± 11.15 |
|
Systolic Blood Pressure, Mean ± SD |
131.92 ± 20.95 |
|
Clinical Characteristics |
|
|
STEMI, n (%) |
89 (49.72) |
|
NSTEMI, n (%) |
17 (9.49) |
|
Unstable Angina, n (%) |
66 (36.87) |
|
Chronic Stable Angina, n (%) |
7 (3.91) |
|
Single Vessel Disease, n (%) |
145 (81.00) |
|
Double/Triple Vessel Disease, n (%) |
34 (18.99) |
|
Baseline TIMI Flow |
|
|
Grade I, n (%) |
27 (15.08) |
|
Grade II, n (%) |
120 (67.04) |
|
Grade III, n (%) |
32 (17.88) |
|
CAD: Coronary Artery Disease; STEMI: ST-Elevated Myocardial Infarction; NSTEMI: Non-ST-Elevated Myocardial Infarction; TIMI: Thrombolysis in Myocardial Infarction |
|
In terms of clinical presentation, the most frequent diagnosis was ST-elevation myocardial infarction (STEMI), observed in 89 patients (49.72%), followed by unstable angina in 66 (36.87%), non-ST elevation myocardial infarction (NSTEMI) in 17 (9.49%), and chronic stable angina in 7 (3.91%). Notably, no cases of total arterial occlusion were recorded; however, thrombus presence within the target vessel was documented in 19.1% of patients. Lesion classification based on ACC/AHA criteria is detailed in Table 2.
Procedural characteristics
The majority of patients (n = 145, 81.00%) had single-vessel coronary artery disease, while 34 patients (18.99%) exhibited multi-vessel involvement (double or triple vessel disease). The left anterior descending artery (LAD) was the most commonly affected vessel. Pre-procedural angiographic characteristics, including lesion location and morphology, are outlined in Table 2.
|
Procedural Angiographic Characteristics |
n=179 |
|
Pre-Procedural % Stenosis, Mean ± SD |
88.45 ± 8.85 |
|
Lesion length (mm), Mean ± SD |
25.61 ± 13.19 |
|
ACC/AHA Lesion Classification |
|
|
Type A, n (%) |
20 (11.17) |
|
Type B, n (%) |
42 (23.46) |
|
Type C, n (%) |
117 (65.36) |
|
Lesion Location |
|
|
LAD, n (%) |
95 (53.07) |
|
RCA, n (%) |
54 (30.17) |
|
LCX, n (%) |
28 (15.64) |
|
LM, n (%) |
2 (1.11) |
|
Balloon Length (mm), Mean ± SD |
14.47 ± 2.53 |
|
Balloon Diameter (mm), Mean ± SD |
2.19 ± 0.39 |
|
Balloon Pressure for Pre-dilatation (atm), Mean ± SD |
14.27 ± 0.36 |
|
Inflation Cycle (number), Mean ± SD |
3.13 ± 2.85 |
|
Procedural Success, n (%) |
179 (97.81) |
|
Device Success, n (%) |
179 (97.81) |
|
Stent Placement Post Pre-Dilatation, n (%) |
179 (100) |
|
ACC/AHA: American College of Cardiology/American Heart Association; LAD: Left Anterior Descending; LCX: Left Circumflex Artery; LM: Left Main Artery; RCA: Right Coronary Artery |
|
Device performances and procedural outcomes
The Optima SC semi-compliant PTCA balloon catheter was used for vessel pre-dilatation in all 183 cases. Devices and procedural success, defined as successful lesion crossing, balloon inflation and deflation, and final achievement of TIMI III flow without major complications, were achieved in 97.8% of cases (n=179). In four patients, the catheter could not be successfully delivered- in one case due to extensive intraluminal thrombus, in two due to severe lesion calcification, and in one case due to marked vessel tortuosity, necessitating procedural modification or deferral. Performance ratings of the device were consistently high. Investigator-assessed balloon attributes such as pushability, trackability, deflation efficiency, and marker visibility were rated above satisfactory in >94% of cases. Other technical parameters, including balloon crossability, and compliance, were also rated above satisfactory in more than 90% of procedures. No procedural difficulties were encountered with balloon inflation, deflation, or withdrawal.
Importantly, no intraprocedural complications such as perforation, dissection, or spasm were reported. During the mean follow-up of 2.6 ± 0.4 months, one patient had suffered angina, and one patient required repeat vascularization because of target lesion failure. Post-procedural and discharge outcomes are summarized in Table 3. The Investigator-rated performance metrics of the Optima SC Balloon Catheter has been shown in Figure 1.
|
Post-Procedural Characteristics |
n=179 |
|
TIMI Flow III Post-Procedure, n (%) |
179 (100) |
|
Post-Procedure % Stenosis, Mean ± SD |
3.97 ± 1.57 |
|
Procedure Related Complication, n (%) |
0 (0) |
|
Device Related Complication, n (%) |
0 (0) |
|
Any In-Hospital Clinical Event/MACE, n (%) |
0 (0) |
|
Any Clinical Event/MACE till FU*, n (%) |
2 (1.1) |
|
TIMI: Thrombolysis in Myocardial Infarction; MACE: Major Adverse Cardiac Event; FU*: Follow-up till 3 months |
|
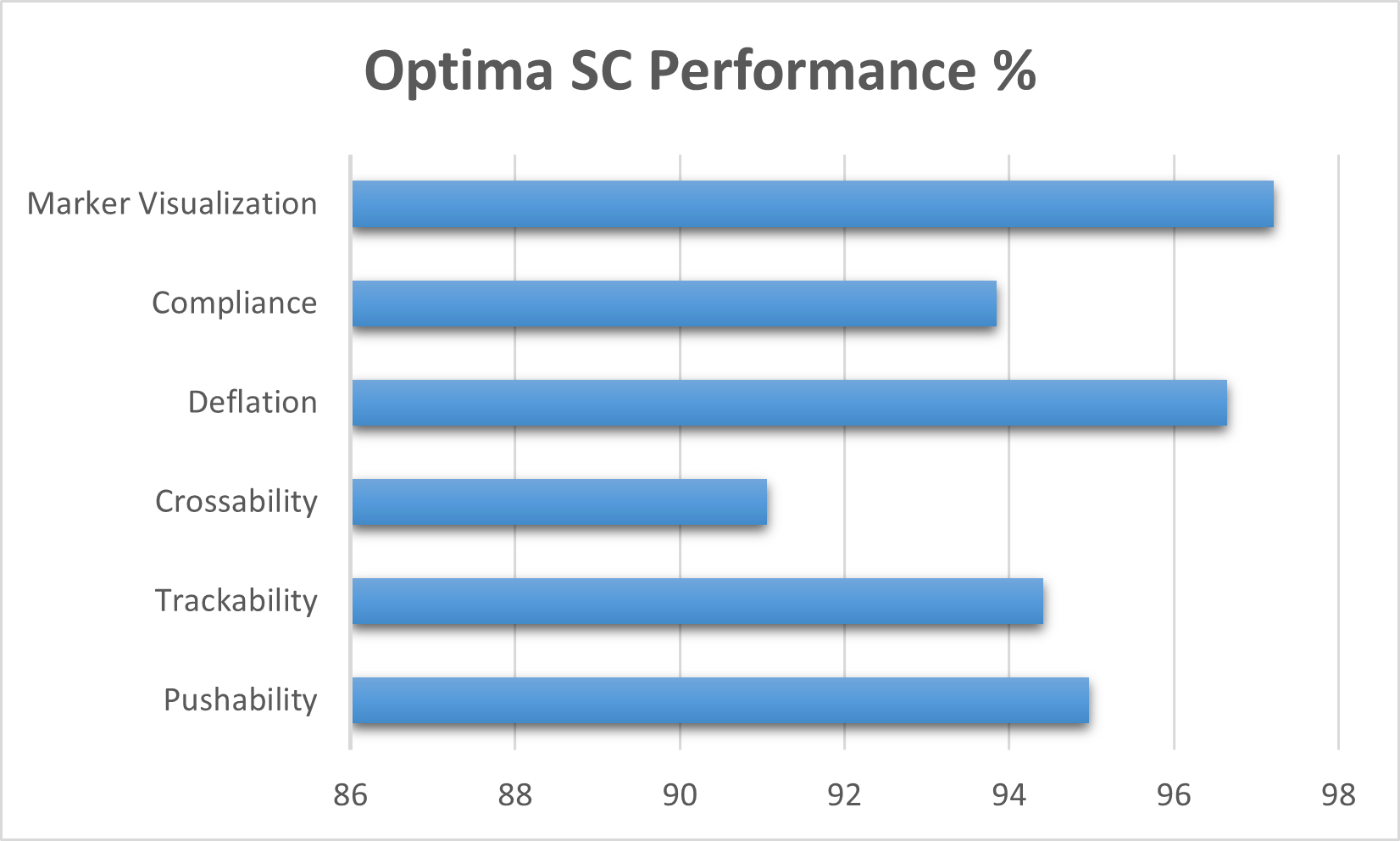
Figure 1. Operator assessment of technical performance parameters for optima SC semi-compliant PTCA balloon (n=179).
Discission
This post-marketing, multicenter, retrospective study evaluated the real-world safety and performance of the Optima SC semi-compliant PTCA balloon catheter in 183 consecutive PCI procedures. Device- and procedure-related success was achieved in 97.8% of the cases. In the four unsuccessful procedures, the catheter advancement was impeded by extensive intraluminal thrombus, heavy concentric calcification, and marked vessel tortuosity. Importantly, none of these failures led to acute vessel injury or clinically significant ischemia, and all were managed safely through procedural deferral or alternative plaque-modifying strategies.
Among the 179 successfully treated patients, no in-hospital MACE, SAE, or device-related complications were recorded. During the 3-month follow-up, one patient experienced recurrent angina, and one patient had target lesion failure, supporting a favorable short-term clinical safety profile.
The procedural success rate observed with Optima SC is consistent with post-marketing evaluations of contemporary semi-compliant balloons, which report success rates between 95 and 100% in broadly inclusive, real-world PCI cohorts [7,8]. While these studies reflect routine clinical practice, differences in balloon material properties, compliance, and lesion complexity preclude direct device-level equivalence. Accordingly, the observed alignment in success rates should be interpreted as contextual benchmarking within the same functional device class rather than evidence of identical mechanical performance.
Consistent short-term clinical safety has also been reported in prior MACE-based evaluations of semi-compliant balloon use. Vorucchu et al. documented 0% MACE at discharge and a 3.2% MACE rate at 12 months in an unselected cohort of 125 patients undergoing PCI with semi-compliant balloons [9], providing appropriate contextual support for the absence of early MACE observed in the present study.
Insights from the OCT-guided randomized study by Cuculi et al. further inform the role of semi-compliant balloons in lesion preparation. In that study, pre-dilatation with conventional semi-compliant balloons resulted in a stent expansion index comparable to non-compliant balloons immediately after stent implantation, suggesting non-inferior preparatory efficacy in simple coronary lesions. However, superior final stent expansion following high-pressure non-compliant balloon post-dilatation highlighted the influence of balloon compliance and inflation characteristics on optimal stent deployment [10]. At 1-year follow-up, clinical event rates were low and comparable between treatment groups, although the study was not powered for clinical endpoints and should therefore be interpreted as procedural and safety reassurance rather than definitive comparative evidence.
Supporting these observations, studies by Bach et al. and Goel et al. have shown that adverse outcomes following PTCA are not intrinsically related to balloon compliance when appropriate technique and lesion selection are applied [11,12]. Collectively, these data support the clinical safety of semi-compliant balloons within their intended use.
Semi-compliant balloons remain the work-horse device for coronary lesion preparation, offering a balance between deliverability with controlled radial force. The present findings support the use of Optima SC as a reliable pre-dilatation tool across a broad spectrum of real-world clinical scenarios, including acute coronary syndromes where rapid and atraumatic vessel expansion is essential. In appropriately selected lesions SC balloons may reduce the need for aggressive non-compliant balloon post-dilatation, potentially minimizing the risk of vessel injury.
There are certain limitations to this study. The retrospective, single-arm design, modest sample size, and short follow-up duration limit conclusions regarding long-term outcomes. Routine intravascular imaging was not mandated, precluding quantitative assessment of stent expansion. Finally, the absence of a comparator arm limits direct performance comparisons. Prospective randomized studies with longer follow-up and systematic imaging are needed to further validate these findings.
Conclusion
In this multicenter real-world series, the Optima SC Semi-compliant PTCA balloon catheter demonstrated high procedural success and an excellent short-term safety profile up to three months. Delivery failures were observed only in select complex cases where additional intervention may be necessary. These results confirm Optima SC as a dependable pre-dilatation balloon for contemporary PCI and provide a rationale for further prospective evaluation in complex coronary subsets.
Conflict of Interest
The authors report no conflicts of interest regarding the content herein.
Funding
The study was funded by Translumina Therapeutics Private Limited.
Acknowledgements
The authors acknowledge Dr. Nancy Chugh and Dr. Brijesh Mishra from Translumina Therapeutics Private Limited for their critical feedback on the Manuscript.
References
2. Engelen SE, Robinson AJB, Zurke YX, Monaco C. Therapeutic strategies targeting inflammation and immunity in atherosclerosis: how to proceed? Nat Rev Cardiol. 2022 Aug;19(8):522–42.
3. Faroux L, Campelo-Parada F, Munoz-Garcia E, Nombela-Franco L, Fischer Q, Donaint P, et al. Procedural Characteristics and Late Outcomes of Percutaneous Coronary Intervention in the Workup Pre-TAVR. JACC Cardiovasc Interv. 2020 Nov 23;13(22):2601–13.
4. Doll JA, Hira RS, Kearney KE, Kandzari DE, Riley RF, Marso SP, et al. Management of Percutaneous Coronary Intervention Complications: Algorithms From the 2018 and 2019 Seattle Percutaneous Coronary Intervention Complications Conference. Circ Cardiovasc Interv. 2020 Jun;13(6):e008962.
5. Pradhan A, Vishwakarma P, Bhandari M, Sethi R, Chandra S, Chaudhary G, et al. Clinical Outcomes of a Non-Compliant Balloon Dilatation Catheter: MOZEC™ NC Study. Int J Environ Res Public Health. 2022 Dec 4;19(23):16231.
6. Dini CS, Tomberli B, Mattesini A, Ristalli F, Valente S, Stolcova M, et al. Intravascular lithotripsy for calcific coronary and peripheral artery stenoses. EuroIntervention. 2019 Oct 20;15(8):714–21.
7. Özel E, Taştan A, Öztürk A, Özcan EE, Uyar S, Şenarslan Ö. What is better for predilatation in bioresorbable vascular scaffold implantation: a non-compliant or a compliant balloon? Anatol J Cardiol. 2016 Apr;16(4):244–9.
8. Vani P, Patel A, Abhishek K, Goyal K. Clinical performance and safety of the Vector® percutaneous transluminal coronary angioplasty balloon catheter: A single-arm, multicenter, retrospective post-marketing clinical study. ARYA Atheroscler. 2024;20(6):17–26.
9. Kitani S, Igarashi Y, Tsuchikane E, Nakamura S, Seino Y, Habara M, et al. Efficacy of drug-coated balloon angioplasty after directional coronary atherectomy for coronary bifurcation lesions (DCA/DCB registry). Catheter Cardiovasc Interv. 2021 Apr 1;97(5):E614–23.
10. Cuculi F, Bossard M, Zasada W, Moccetti F, Voskuil M, Wolfrum M, et al. Performing percutaneous coronary interventions with predilatation using non-compliant balloons at high-pressure versus conventional semi-compliant balloons: insights from two randomised studies using optical coherence tomography. Open Heart. 2020 Jan 23;7(1):e001204.
11. Bach RG, Kern MJ, Aguirre FV, Donohue TJ, Bell C, Penick D. Effects of percutaneous transluminal coronary angioplasty balloon compliance on angiographic and clinical outcomes. Am J Cardiol. 1993 Oct 15;72(12):904–7.
12. Goel P, Agarwal R, Kaul U, Wasir HS. Effects of balloon compliance on angiographic and clinical outcomes after PTCA. Int J Cardiol. 1995 Aug;51(1):1–4.
