Abstract
The global incidence of metabolic disorders is on the rise, posing a significant challenge to public health. With remarkable advancements in diagnostic tools and clinical procedures, our understanding of the etiology and underlying pathophysiology of these disorders has expanded considerably. Furthermore, the utilization of in vitro and in vivo experimental models, preceding clinical investigations, has catalyzed numerous breakthroughs in biomedicine, particularly in the identification and development of potential drug candidates for the management of metabolic disorders. Natural compounds isolated from various sources have garnered extensive attention as prospective drug candidates for the treatment of conditions such as diabetes, obesity, heart-related diseases, and cancer. This interest is partly attributed to their inherent antioxidant and anti-inflammatory properties. Concurrently, intensive research efforts have been directed towards enhancing the bioactivity and bioavailability of these compounds through selected drug delivery strategies. In this article, we provide valuable insights into recent advancements that shed light on the role of inflammatory-mediated responses in the initiation of metabolic disorders, with a specific focus on conditions like diabetes mellitus, obesity, heart-related diseases, cancer, and related drug delivery systems. Additionally, we explore the promising potential of natural products in managing these metabolic disorders. Furthermore, we present lists of potential biological targets suitable for high throughput screening in the drug discovery and development process. Finally, we delve into the findings gleaned from preclinical and clinical studies, setting the stage for the identification of suitable approaches in the realm of phytochemical drug delivery systems that hold promise for the treatment of metabolic disorders.
Keywords
Natural products, Inflammation, Metabolic disorders, Drug delivery, Diabetes
Introduction
Metabolism, a highly orchestrated process encompassing the breakdown of ingested foods into simpler elements, is essential for all life forms, including humans [1]. In eukaryotes, this intricate process serves as the primary source of energy required for growth and survival. Any disruptions in this process can have adverse consequences on the species' viability [2]. Regrettably, recent years have witnessed a concerning surge in metabolism-related issues, collectively referred to as metabolic disorders. Individuals afflicted by metabolic disorders often exhibit distinct characteristics compared to those with normal metabolism, often stemming from various pathological conditions giving rise to unique phenotypes [3]. Presently, the most prevalent metabolic disorders encompass diabetes mellitus, obesity, heart-related diseases, and cancer [4]. Although substantial knowledge has been amassed regarding the etiology and pharmacological management of these disorders, their mechanistic underpinnings remain complex and partially understood. Nonetheless, chronic inflammation has emerged as a pivotal contributor to the initiation, progression, and transition of these metabolic disorders [5,6]. Notably, the stimulation of pro-inflammatory cytokines in response to the release of endogenous danger-associated ligands has been consistently observed in a wide spectrum of metabolic disorder-related conditions [7].
A growing body of evidence suggests that natural products and their bioactive compounds, particularly phytochemicals, offer a myriad of health benefits. Among the most extensively researched areas in natural products is their potential application in the treatment of diabetes, obesity, cardiovascular issues, and various cancers [8]. These benefits are often attributed to their capacity to target oxidative stress-related pathways and the intricate regulatory network governing the inflammatory processes [9]. In this comprehensive review, we delve into the current understanding of the pathophysiology underlying diabetes, obesity, heart-related diseases, and cancer in connection with inflammation-mediated induction of metabolic disorders. Furthermore, we offer a concise discussion on the promising role of natural products in managing these conditions, including a list of potential biological targets for phytochemicals. We also examine findings from both preclinical and clinical studies, followed by an exploration of contemporary approaches to phytochemical drug delivery systems employed in the treatment of metabolic disorders.
Diabetes and Its Pathophysiology
The pathophysiology of diabetes mellitus is intricately linked to two fundamental factors: insulin levels and the body's capacity to effectively utilize this hormone. Insulin plays a pivotal role in facilitating the entry of blood glucose into cells, where it undergoes metabolism to generate energy. Consequently, any conditions that disrupt the normal physiological functions of insulin can lead to disturbances in glucose levels. Diabetes mellitus encompasses several types, with type 1 (T1) and type 2 (T2) diabetes mellitus being the most widely recognized forms. While these two types exhibit distinct pathogenic mechanisms, a common pathophysiological hallmark shared by both type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) is the impaired ability of insulin to facilitate the entry of glucose into cells.
T1DM is characterized by the dysfunction of pancreatic beta cells, leading to their incapacity to produce insulin. This disruption is often linked to an autoimmune disease [10]. In this autoimmune condition, the immune system, instead of safeguarding the body against external threats, targets various internal systems, tissues, or cells, including the insulin-producing pancreatic beta cells. This relentless assault compromises their ability to generate adequate insulin levels, resulting in a substantial increase in blood glucose levels and the manifestation of hyperglycemia. While significant progress has been made in unraveling the pathophysiological aspects of T1DM in recent years, the autoimmune nature of this diabetes type remains incompletely understood. Nevertheless, certain insights into the involvement of the immune system in the onset of T1DM have emerged. Notably, more than three decades ago, it was observed that diabetic patients exhibited elevated expression of a molecule known as human leukocyte antigen (HLA) [11]. As this molecule plays a crucial role in regulating immune responses by encoding various related proteins involved in antigen presentation, any alterations in its expression or function may disrupt self-tolerance mechanisms.
In contrast, severe hyperglycemia in patients with T2DM is primarily attributed to insulin resistance, rather than the destruction of beta cells. This condition arises from peripheral tissues and cells failing to respond effectively to insulin, impairing their ability to uptake blood glucose. Consequently, hyperglycemia ensues, even when the circulating insulin levels are within the physiological range. This situation sets off a cycle where beta cells continue producing insulin in response to the persistently elevated glucose levels, ultimately leading to their failure to secrete insulin when T2DM is diagnosed [12].
Several factors have been proposed to influence beta cell function and insulin production, including the relatively recent concept of gut-related hormones, also known as incretins. Two gut hormones, glucagon-like peptide-1 (GLP-1) and gastric inhibitory polypeptide (GIP), act as messengers to stimulate insulin secretion following glucose ingestion. However, this mechanism is not fully activated when glucose is administered intravenously. In individuals with T2DM, the secretion of incretins, particularly GLP-1, is significantly reduced, hampering the induction of insulin production after meals. This imbalance also results in increased glucagon secretion and the conversion of glycogen to glucose, contributing to elevated blood glucose levels [13].
The precise mechanism underlying insulin resistance remains somewhat elusive. Nevertheless, a clear association exists between insulin resistance and fat accumulation, particularly in cases of obesity. Both the liver and muscles are key players in the development of insulin resistance, potentially due to their capacity to store excessive fat [14]. Excess fat accumulation, often associated with obesity resulting from excessive calorie intake without adequate physical activity, significantly contributes to this condition. Specifically, fat buildup in beta cells can impair their function, hindering their ability to produce insulin at physiological levels and ultimately leading to the dysregulation of blood glucose levels [14].
Genetics also exert an influence on the pathophysiology of T2DM, despite the condition not displaying a strong inheritance pattern. Nonetheless, family history and genetic predisposition are recognized as risk factors for T2DM and warrant careful consideration [15].
Obesity
The pathogenesis of obesity is intricate, with a multitude of contributing factors spanning environmental, social, behavioral, physiological, medical, and genetic domains [16]. Environmental shifts resulting from advancements in technology and healthcare have led to significant lifestyle modifications. Infectious diseases, which were once the primary cause of death, are now better controlled, leading to changes in behavior and physical activity. Factors such as increased access to various modes of transportation and the widespread availability of electronic devices have reduced physical activity levels. Furthermore, easy access to high-calorie foods exacerbates the issue.
Genetic factors also play a pivotal role in the pathogenesis of obesity. Remarkably, the heritability of body mass index (BMI) falls within the range of 40% to 70% [17]. Researchers have identified several monogenic mutations or alterations associated with obesity pathogenesis, with particular interest in deficiencies of leptin and melanocortin-4 receptors. These receptors are instrumental in regulating human energy homeostasis [18]. Studies have consistently revealed that obesity often involves a deficiency of these proteins. Additionally, the vagal nerve is linked to obesity pathogenesis, serving as the key connection between the brain and the gut for satiety regulation [19]. The vagal nerve receives information from the gastrointestinal tract after the ingestion process through various mechanisms, including mechanical stimulation, release of gut hormones, activation of chemoreceptors, and direct actions of certain nutritive compounds [20]. Gastric distension following feeding stimulates the first mechanism, while the second mechanism is mediated by various gut hormones. A multitude of gut hormones, including cholecystokinin, peptide YY (PYY), pancreatic polypeptide (PP), glucagon-like peptide-1 (GLP-1), ghrelin, insulin, and leptin, have been identified [21]. Despite their differing functions, these hormones collectively aim to regulate food intake and gastric emptying.
Upon receiving information from peripheral receptors, the vagal nerve transmits this data to the area postrema and nucleus of the solitary tract complex in the brainstem, where it undergoes processing before being relayed to the dorsal motor nucleus [22]. Modulation of this pathway can lead to various events related to the control of gastric emptying, absorption rates, and alterations in the secretion of gut hormones [23]. Given the pivotal role of the vagal nerve, any conditions that disrupt its ability to receive information from the gut may result in dysregulation of energy balance.
The onset and progression of cancer are intimately tied to DNA damage. Given that our cells are continually exposed to various stressors that can harm DNA, our body has developed several mechanisms to address this issue, including cell-cycle arrest, DNA repair, cellular senescence, and apoptosis [24]. These mechanisms are rigorously controlled by the p53 family, often referred to as the guardian of the genome [25]. Any factors that disrupt the functionality of the p53 family can potentially pave the way for the development of cancer.
A significant proportion of human cancers, more than half, can be traced back to missense mutations in the transcription factor p53 family [26]. Among the various mutation sites, the DNA-binding domain (DBD) appears to be the most susceptible site for mutation within the p53 family [27]. Under normal conditions, the expression of p53 is maintained at a very low level. However, when a specific stressor impacts a cell, p53 promptly increases the expression of murine/human double minute 2 (MDM2). Intriguingly, MDM2 also possesses the ability to implement negative feedback regulation on p53, preventing its expression from rising further [28].
The p53 family's role in mitigating DNA damage encompasses apoptosis, cell-cycle arrest, and senescence mechanisms. In apoptosis, p53 can initiate this process through either extrinsic or intrinsic pathways. The former pathway is mediated by the action of death receptors, while the latter involves the release of cytochrome c into the cytosolic region of mitochondria [29]. Caspase-3 appears to play a pivotal role in executing the final stages of both pathways. To facilitate and activate the cell cycle, the cyclin-dependent kinases (CDKs) family is of paramount importance, and inhibiting this protein family is essential to halt cellular replication, a beneficial effect in preventing the division of cancer cells. The p53 family regulates CDKs by inducing the activation of the p21 protein, which is responsible for CDK inhibition [30]. Lastly, p53 can activate cellular senescence through its influence on certain genes, including p21, p16-Rb, and B-cell Translocation Gene 2 BTG2 [31]. This mechanism is particularly critical in curbing the progression and dissemination of cancer cells. Several other mechanisms are also associated with p53's role in safeguarding cells against cancerous threats. Some of these mechanisms involve its ability to prevent events such as cancer cell migration to other tissues, angiogenesis, oxidative stress, and drug resistance. Additionally, p53 can induce autophagy and promote genome stabilization.
Natural Products to Treat Diabetes
A deficiency of functional β-cells within the Langerhans islets leads to insulin resistance, resulting in persistently elevated blood sugar levels and eventually culminating in diabetes mellitus [11]. The term "Diabetes Mellitus" originates from the Greek language, with "Diabetes" meaning "a passer through" and "Mellitus" meaning "sweet" [32]. When the body ceases to produce or efficiently utilize insulin, it poses significant health risks to the cardiovascular system, blood vessels, eyes, and kidneys. The global prevalence of diabetes is on a rapid rise, making it a prominent global health concern. In 2019, the International Diabetes Federation (IDF) estimated that 463 million adults were affected by diabetes, a number projected to increase to 578 million by 2030 and a staggering 700 million by 2045 [33]. Additionally, around 374 million individuals worldwide were living with undiagnosed diabetes in 2017 [34]. Diabetes mellitus is categorized into two distinct types: type 1 and type 2. In the case of type T1DM, the immune system erroneously targets and attacks β-cells triggered by environmental factors such as chemicals and viruses [35]. As a result, exogenous insulin becomes imperative for managing type 1 diabetes [36]. Approximately 10% of all diabetic patients contend with this condition, which predominantly affects young individuals [37].
In contrast, type 2 diabetes mellitus (T2DM), also known as "non-insulin-dependent diabetes," typically develops in adulthood and is characterized by the body's inefficient use of insulin, a condition medically termed "peripheral tissue resistance" [38]. Factors such as childhood sunlight exposure and maintaining a healthy body weight have been found to reduce the risk of developing T2DM [39]. While medications like metformin, sulfonylurea, and insulin are currently available and scientifically proven for treating T2DM, newer medications like α-glucosidase inhibitors, thiazolidinediones, glucagon-like peptide-1 receptor agonists, pramlintide, and dipeptidyl peptidase-4 inhibitors have emerged with limited supporting evidence. Therefore, the quest for effective drugs with minimal side effects, including severe hypoglycemia, and safety concerns in some cases, underscores the importance of exploring alternative therapies for diabetes management. In most instances, the advantages of availability, affordability, and safety associated with alternative treatments would outweigh their disadvantages.
Persistent efforts are being made to explore diabetes and uncover novel therapeutic avenues, including the identification of natural products with anti-diabetic properties. Individuals with diabetes and cancer have turned to a wide array of alternative treatments to manage their condition [40]. Both pre-clinical and clinical trials have been conducted on various natural products for diabetes and cancer treatment [41].
Numerous naturally occurring substances exhibit potential in regulating blood sugar levels in diabetic patients. Mechanisms underlying their anti-diabetic effects include the inhibition of digestive enzymes like α-glucosidase and α-amylase [42], modulation of glucose uptake and the expression of glucose transporters, enhancement of insulin secretion and pancreatic β-cell proliferation, alleviation of insulin resistance, and regulation of oxidative stress. The extensive array of molecules derived from natural products that have undergone clinical trials highlights the ongoing promise of natural products as a source for the development of innovative therapeutics.
Animal models in predicting idiosyncratic drug-induced liver injury (DILI)
Considering the unpredictability of idiosyncratic Drug-Induced Liver Injury (DILI), it remains challenging to prospectively study the patients susceptible to its effects. While animal models are pivotal in biomedical research, they face limitations in replicating idiosyncratic DILI, exhibiting idiosyncrasies distinct from those in humans. Existing animal models often involve high drug doses or other manipulations causing direct and immediate liver injury, thereby presenting histological disparities from human idiosyncratic DILI.
These dissimilarities between animal models and human idiosyncratic DILI, such as distinct HLA genotypes associated with increased risk in humans, pose difficulties in developing animal models representative of this condition. Drugs requiring specific HLA genotypes further complicate animal model development. However, certain idiosyncratic DILI instances are not associated with specific HLA genotypes, potentially due to drug reactions with various endogenous proteins that subsequently generate peptides binding to various HLA molecules expressed in humans or animals.
Recent advances in cancer treatment, focusing on immune checkpoint inhibitors targeting PD-1 and CTLA-4, have unveiled liver-related adverse effects including DILI. Immune checkpoint inhibitors not only induce DILI but also elevate the risk of idiosyncratic DILI when combined with other drugs. Studies using PD-1 knockout mice treated with anti-CTLA-4 and amodiaquine, a drug linked to idiosyncratic DILI, exhibit liver injury resembling human idiosyncratic DILI in histology. However, this injury, mediated by CD8 T cells, doesn't result in overt liver failure.
Despite this, drugs associated with severe liver injury often manifest as mild liver injury in most individuals, indicating a potential innate immune response to these drugs without the specific HLA and T cell receptors required for an adaptive immune response leading to injury. Studying the innate immune response to drugs might offer insights into early immune events, aiding in screening drug candidates for potential idiosyncratic DILI risks [43].
Drug Delivery Approaches for Bioactive Compounds in the Treatment of Diabetes
The grapevine (Vitis vinifera) is renowned for its abundance of phenolic compounds, well-documented for their potent antioxidant properties. Teschke and colleagues made clinical investigation analysis and concluded that most of the mechanistic steps leading to idiosyncratic DILI remain unclear [43]. Liposomes, recognized for their capacity to enhance drug efficacy in various medical contexts [44-46], were employed to encapsulate these compounds using an extrusion technique, yielding impressive entrapment efficiencies exceeding 80% for both cyanidin and delphinidin. In vitro investigations revealed that the free forms of these compounds could reduce albumin glycation by 30.5% for delphinidin and 46% for cyanidin. Remarkably, when formulated into liposomal delivery systems, glycation of albumin decreased substantially to 8.5% for delphinidin and 14.6% for cyanidin. Furthermore, in vivo experiments demonstrated that liposomal delivery significantly outperformed the free compounds in reducing albumin and HbA1c glycation rates in diabetic mice. These results underscore the potential of natural compound formulations in advancing diabetic therapy (Figure 1).
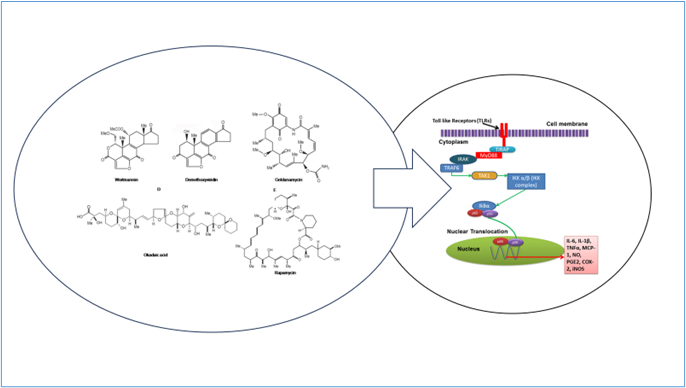
Figure 1. Exploration of bioactive compounds in diabetes treatment.
In a parallel study, Yücel and colleagues harnessed a similar liposomal delivery system to encapsulate resveratrol, a natural compound, in two distinct forms: PEGylated and non-PEGylated liposomes [47]. They employed a combination of dipalmitoylphosphatidylcholine (DPPC) and cholesterol to craft multi-bilayered nanoparticles with a size of 215 nm. When they treated diabetic (streptozotocin-induced) pancreatic cell lines with both resveratrol solution and resveratrol-loaded liposomes for 24 hours, insulin concentrations exhibited a notable increase, with liposomal formulations showing a more substantial effect, while glucose concentrations decreased. In vitro assessments highlighted the significant antioxidant activity of liposomal formulations within pancreatic cells compared to their free counterparts. This suggests a promising avenue for diabetes mellitus therapy, with a particular focus on alleviating oxidative stress.
Mao and collaborators delved into the potential of Echinacea purpurea as an antidiabetic agent. E. purpurea extract is known to contain an array of phenolic compounds and isobutylamides, showcasing antidiabetic activity. To enhance its effectiveness, the extract was incorporated into chitosan/silica nanoparticles, resulting in particles with a size of 218 nm, an entrapment efficacy of 66.9%, and a drug loading of 39.9%. Notably, this formulation exhibited the capacity to reduce oxidative stress in LC-540 cells, manifesting robust antioxidant activity. Significantly, in an in vivo study employing streptozotocin (STZ)-induced diabetes rat models, these nanoparticles managed to normalize blood glucose levels, enhance insulin resistance, and elevate plasma fibroblast growth factor 21 (FGF21) resistance compared to the free extract. This highlights the potential of these nanoparticles in improving diabetic therapy. Gold nanoparticles, an alternative type of nanoparticles, were also explored for their antidiabetic potential. They synthesized gold nanoparticles using an aqueous extract from Cassia fistula stem bark. Gold nanoparticles have garnered attention for their green synthesis using natural compounds [48,49]. This study encompassed various characterizations, including ultraviolet–visible spectroscopy, Fourier transform infrared spectroscopy, and scanning electron microscopy, to assess absorbance patterns, functional groups, and nanoparticle size, respectively. The outcomes demonstrated that gold nanoparticles prepared from C. fistula stem bark aqueous extract exhibited promising hypoglycemic activity compared to the aqueous extract. Evaluation criteria included serum glucose levels, body weight, kidney function, liver function, and lipid profile. The administration of gold nanoparticles was associated with a reduction in serum biochemistry parameters in rats with streptozotocin-induced diabetes, affirming the potential of C. fistula-derived gold nanoparticles to enhance diabetic therapy.
Drug Delivery Approaches for Bioactive Compounds in the Treatment of Obesity
The utilization of drug delivery systems incorporating natural compounds has also found application in the realm of obesity treatment. Salacia chinensis, a prominent Ayurvedic medicine, has garnered attention for its potential pharmacological effects in this context. Researchers have embarked on a study involving gold nanoparticles loaded with S. chinensis to investigate its anti-obesity properties [50]. The investigation was conducted on obese rats subjected to a high-fat diet (HFD). Initially, the nanoparticles were meticulously characterized for their physicochemical attributes, revealing a spherical shape with crystalline properties. In the subsequent in vivo study, these nanoparticles exhibited the capacity to mitigate several obesity-related parameters in HFD-fed rats. These included changes in body weight, resistin levels, adipose index, inflammatory markers, BMI, leptin, CRI, adiponectin, AI, liver marker enzymes, lipid profiles, and AMPK signaling proteins. Additionally, the histopathological assessment of the liver showcased promising outcomes, with a reduction in hepatocyte degradation following the administration of Salacia chinensis-loaded nanoparticles. In a similar vein, Ansari et al. employed a comparable approach by developing gold nanoparticles synthesized using Smilax glabra rhizome [21]. These nanoparticles measured 21 nm in size and exhibited exceptional cellular uptake properties. The administration of these nanoparticles to HFD-fed rats demonstrated superior anti-obesity activity across various parameters. These encompassed lipid profiles, liver markers, hormone levels such as leptin, adiponectin, and resistin, as well as histopathological evaluations.
To address the issues of bioavailability and solubility associated with resveratrol as an anti-obesity agent, Wan and colleagues formulated PLGA nanoparticles encapsulating resveratrol [51,52]. These nanoparticles were crafted using an oil-in-water emulsion method, yielding particles sized at 176.1 nm with a zeta potential of -22.6 mV. Moreover, they exhibited remarkable entrapment efficiency (97%) and drug loading (14.9%), coupled with sustained release characteristics within the gastrointestinal tract and excellent physical stability profiles. Importantly, when compared to free resveratrol, the administration of PLGA nanoparticles demonstrated enhanced anti-obesity activity by influencing lipogenesis, promoting lipolysis, and reducing hepatocellular proliferation. Furthermore, other researchers explored the isolation of cellulose nanocrystals from grapes and assessed their anti-obesity potential [53-63]. These nanocrystals were isolated using sodium hydroxide and subjected to bleaching with sulfuric acid. In a rat obesity model, the nanocellulose displayed anti-obesity activity based on observations of body weight, lipid profiles, liver function, and kidney function when compared to a positive control group given grape seed powder. Taking a different administration route, scientists devised nanofiber patches for the transdermal delivery of curcumin. The transdermal route has gained recognition as an alternative to conventional oral delivery for various drugs, with numerous studies indicating improved bioavailability via this route [64-73]. These nanofibers were created using polyvinyl alcohol and gelation, resulting in a formulation characterized by fiber diameters of 200–250 nm and high reproducibility [74-78]. The effectiveness of transdermal delivery was evaluated by monitoring parameters such as body weight, blood parameter levels, and MRI imaging. The results indicated a reduction in leptin levels following transdermal curcumin delivery, and MRI imaging showed a decrease in adipose tissue by approximately 4–7%. Consequently, this highlights the potential of transdermal delivery as an alternative route for administering natural compounds in the context of obesity therapy [78-87].
RUCAM in Drug and Herb Induced Liver Injury
The Roussel Uclaf Causality Assessment Method (RUCAM), formerly known as CIOMS (Council for International Organizations of Medical Sciences), is a widely adopted and well-established tool for quantitatively assessing causality in suspected cases of Drug-Induced Liver Injury (DILI) and Herbal and Dietary Supplement-Induced Liver Injury (HILI). The historical background and original research validate the preference for using RUCAM as the sole term for future cases, thereby discontinuing the use of CIOMS to enhance simplicity and clarity. RUCAM offers a structured, standardized, and hepatotoxicity-specific diagnostic approach that assigns scores to specific key criteria. This method enables the assignment of quantitative causality grades for each suspect drug or herb in a case report. A consensus among experts from Europe and the United States, established in collaborative meetings, laid the foundation for the initial criteria of RUCAM, catering to the needs of clinicians and practitioners caring for patients with suspected DILI and HILI. Additional criteria were subsequently integrated into RUCAM, resulting in its validation and contributing to the timely and precise diagnosis of liver injuries. For over two decades, RUCAM has been successfully applied by physicians, regulatory agencies, authors of case reports, and pharmaceutical companies in numerous countries. Their hands-on experience, coupled with emerging data on DILI and HILI characteristics, as well as some ambiguities related to factors such as alcohol use and exclusions of non-drug causes, prompted the revision of RUCAM. The primary goal was to reduce variability in causality assessments among different observers and within the same observer, create clearly defined and objective core components, and streamline the application of its criteria. In this updated version of RUCAM, the authors present the revised scale, building upon the well-accepted original version. Authors recommend its use for clinical, regulatory, publication, and expert purposes to facilitate the valid establishment of causality in suspected cases of DILI and HILI. This updated version not only simplifies the handling of the criteria but also promotes an internationally harmonized approach to causality assessment, serving as a fundamental tool for practitioners in the field [88-97].
Limitations and Future Prospects
Muscle-associated metabolic disorders, particularly Type 2 Diabetes (T2D), have been a subject of intense research and therapeutic development. Various strategies have been employed to address these conditions, focusing on specific druggable proteins, including DPP4, PTB1B, SGLT, PPARγ, and GLP-1R. These efforts have yielded promising results, showcasing the potential for novel treatment options. To effectively combat metabolic diseases such as insulin resistance (IR) and T2DM, it is crucial to delve deeper into the molecular mechanisms of these target proteins and their roles in skeletal muscle (SM) metabolism [54,98-106]. A comprehensive understanding of these processes is fundamental for the development of more efficient therapeutic approaches. Among the crucial regulatory factors associated with myogenesis and SM metabolism, MSTN stands out as a key player in the regulation of insulin resistance and muscle growth. Investigating these aspects further is imperative to refine our therapeutic strategies for managing metabolic conditions linked to these intricate processes. Throughout history, natural compounds derived from herbal plants have played a significant role in the treatment of SM-associated metabolic disorders, including diabetes. The extensive pharmacological exploration of natural inhibitors or modulators that target proteins such as DPP4, PTB1B, SGLT, GLP-1R, and PPARγ promises to expand our knowledge and provide updated information about their therapeutic applications [79]. This review aims to contribute to the identification of safer natural inhibitors or agonists that target these specific proteins. By doing so, it aspires to enhance the management of diabetes and related metabolic disorders [106-117]. The pursuit of natural compounds as potential therapeutics offers a promising avenue for improving the treatment and care of individuals affected by these conditions.
Also, recently Teschke et.al, published a review on the exploration of natural products to uncover novel active ingredients has sparked significant interest in both clinical and scientific circles. These compounds hold potential for developing new nature-based drugs and augmenting existing conventional and herbal medications used in treating human ailments. Past and present nature-derived drugs have primarily originated from plants, fungi, bacteria, and occasionally from animals or maritime sources like algae. Future endeavors are likely to emphasize phytochemicals within plants, particularly those recognized for their therapeutic potential in traditional herbal medicines like Traditional Chinese Medicine (TCM). The authors emphasis to gain acceptance, new herbal drugs must demonstrate efficacy against well-defined diseases via randomized clinical trials (RCTs) while showcasing a favorable risk-benefit profile by avoiding severe adverse effects. Tropical plants, abundant in diverse species within tropical forests, warrant further clinical investigation. Despite concerns about potential reductions in plant numbers and diversity due to mismanagement, attention has gravitated toward Sustainable Development Goal 3 (SDG3), which reinforces global health and well-being within the UN's 2030 Agenda. Overall, the review signifies that the development of new drugs from natural products could significantly contribute to improving health conditions and preserving well-being in alignment with these goals [118-120].
Conclusion
Metabolic disorders are complex conditions that often necessitate the use of multiple medications to achieve an effective pharmacological response. In this context, the utilization of pharmaceutical preparations derived from natural products holds promise as a safer option. Natural products are widely recognized for their lower risk of triggering harmful adverse effects, making them a viable choice for addressing metabolic disorders. However, it is essential to acknowledge that the slow progress of animal and clinical studies aimed at demonstrating the efficacy and safety of natural products has posed a significant challenge in advancing biomedical and pharmaceutical research in this field. Therefore, it is imperative to address this issue promptly and decisively to narrow the gap in providing scientific evidence supporting the benefits of phytochemicals derived from plants in the management of metabolic disorders. Such efforts are crucial for providing substantial support in the ongoing fight against the rising incidence of diseases associated with metabolic disorders.
List of Abbreviations
BMI: Body Mass Index; PYY: Peptide YY; PP: Pancreatic Polypeptide; GLP-1: Glucagon-Like Peptide-1; DBD: DNA-Binding Domain; H-i: Herb- induced; HILI: Herbal and Dietary Supplement-Induced Liver Injury (HILI); Human Leukocyte Antigen (HLA); MDM2: Murine/human Double Minute 2; CDKS: Cyclin-Dependent Kinases (CDKs); IDF: International Diabetes Federation; DPPC: Dipalmitoylphosphatidylcholine; FGF21: Fibroblast Growth Factor 21; HFD: High-Fat Diet; RUCAM: Roussel Uclaf Causality Assessment Method; DILI: Drug-Induced Liver Injury; T1DM: Type 1 Diabetes Mellitus; T2DM: Type 2 Diabetes Mellitus; GLP-1: Glucagon-Like Peptide-1; GIP: Gastric Inhibitory Polypeptide
References
2. Agrawal AK, Aqil F, Jeyabalan J, Spencer WA, Beck J, Gachuki BW, et al. Milk-derived exosomes for oral delivery of paclitaxel. Nanomedicine: Nanotechnology, Biology and Medicine. 2017 Jul 1;13(5):1627-36.
3. Kim KL, Sung G, Sim J, Murray J, Li M, Lee A, et al. Supramolecular latching system based on ultrastable synthetic binding pairs as versatile tools for protein imaging. Nature Communications. 2018 Apr 27;9(1):1712.
4. Ahn K, Ji H, Kim HE, Cho H, Sun Q, Shi S, et al. Raphanus sativus L. seed extracts induce apoptosis and reduce migration of oral squamous cell carcinoma KB and KBCD133+ cells by downregulation of β-catenin. Nutrition and Cancer. 2020 Nov 16;72(8):1378-89.
5. Li M, Lee A, Kim KL, Murray J, Shrinidhi A, Sung G, et al. Autophagy caught in the act: a supramolecular FRET pair based on an ultrastable synthetic host–guest complex visualizes autophagosome–lysosome fusion. Angewandte Chemie. 2018 Feb 19;130(8):2142-7.
6. Alatrash G, Jakher H, Stafford PD, Mittendorf EA. Cancer immunotherapies, their safety and toxicity. Expert Opinion on Drug Safety. 2013 Sep 1;12(5):631-45.
7. Alberti KG, Zimmet P, Shaw J. International Diabetes Federation: a consensus on Type 2 diabetes prevention. Diabetic Medicine. 2007 May;24(5):451-63.
8. Murray J, Sim J, Oh K, Sung G, Lee A, Shrinidhi A, et al. Enrichment of specifically labeled proteins by an immobilized host molecule. Angewandte Chemie International Edition. 2017 Feb 20;56(9):2395-8.
9. Ali AN, Saeed NA, Omear HA. The anticancer properties of Artemisia aucheri boiss extract on HT29 colon cancer cells. Journal of Gastrointestinal Cancer. 2021 Mar;52:113-9.
10. Ali MY, Jannat S, Jung HA, Choi JS. Insulin–mimetic dihydroxanthyletin-type coumarins from Angelica decursiva with protein tyrosine phosphatase 1B and α-glucosidase inhibitory activities and docking studies of their molecular mechanisms. Antioxidants. 2021 Feb 15;10(2):292.
11. Hewitt CS, Abutaleb NS, Elhassanny AE, Nocentini A, Cao X, Amos DP, et al. Structure–activity relationship studies of acetazolamide-based carbonic anhydrase inhibitors with activity against Neisseria gonorrhoeae. ACS Infectious Diseases. 2021 Mar 25;7(7):1969-84.
12. Alshehri MA. Cardioprotective properties of Artemisia herba alba nanoparticles against heart attack in rats: A study of the antioxidant and hypolipidemic activities. Saudi Journal of Biological Sciences. 2022 Apr 1;29(4):2336-47.
13. Park KM, Baek K, Ko YH, Shrinidhi A, Murray J, Jang WH, et al. Mono‐allyloxylated cucurbit [7] uril acts as an unconventional amphiphile to form light‐responsive vesicles. Angewandte Chemie. 2018 Mar 12;130(12):3186-90.
14. Alzahrani AJ. Potent antioxidant and anticancer activities of the methanolic extract of Calligonum comosum (L'Her) fruit hairs against human hepatocarcinoma cells. Saudi Journal of Biological Sciences. 2021 Sep 1;28(9):5283-9.
15. Kishor C, Arya T, Reddi R, Chen X, Saddanapu V, Marapaka AK, et al. Identification, biochemical and structural evaluation of species-specific inhibitors against type I methionine aminopeptidases. Journal of Medicinal Chemistry. 2013 Jul 11;56(13):5295-305.
16. Anand A, Komati A, Katragunta K, Shaik H, Nagendla NK, Kuncha M, et al. Phytometabolomic analysis of boiled rhizome of Nymphaea nouchali (Burm. f.) using UPLC-Q-TOF-MSE, LC-QqQ-MS & GC–MS and evaluation of antihyperglycemic and antioxidant activities. Food Chemistry. 2021 Apr 16;342:128313.
17. Ambrose JA, Singh M. Pathophysiology of coronary artery disease leading to acute coronary syndromes. F1000prime Reports. 2015;7.
18. Abdelbaky MS, Ibrahim HS, Hassan ML, Sayed ZE. Nanoparticles effects of red grape (Vitis vinifera) seeds and grape seeds powder on obese hyperlipidemic rats. ARC Journal of Nutrition and Growth. 2016;2:1-5.
19. Chitturi KL, Garimella S, Marapaka AK, Kudle KR, Merugu R. Single pot green synthesis, characterization, antitumor antibacterial, antioxidant activity of bimetallic silver and copper nanoparticles using fruit pulp of palmyra fruit. Journal of Bionanoscience. 2018 Apr 1;12(2):284-9.
20. Park KM, Kim J, Ko YH, Ahn Y, Murray J, Li M, et al. Dye-cucurbit [n] uril complexes as sensor elements for reliable pattern recognition of biogenic polyamines. Bulletin of the Chemical Society of Japan. 2018 Jan 15;91(1):95-9.
21. Ansari S, Bari A, Ullah R, Mathanmohun M, Veeraraghavan VP, et al. Gold nanoparticles synthesized with Smilax glabra rhizome modulates the anti-obesity parameters in high-fat diet and streptozotocin induced obese diabetes rat model. Journal of Photochemistry and Photobiology B: Biology. 2019 Dec 1;201:111643.
22. Aeluri R, Ganji RJ, Marapaka AK, Pillalamarri V, Alla M, Addlagatta A. Highly functionalized tetrahydropyridines are cytotoxic and selective inhibitors of human puromycin sensitive aminopeptidase. European Journal of Medicinal Chemistry. 2015 Dec 1;106:26-33.
23. Hariprasad KS, Anand A, Rathod BB, Zehra A, Tiwari AK, Prakasham RS, et al. Neoteric Synthesis and Biological Activities of Chromenopyrazolones, Tosylchromenopyrazolones, Benzoylcoumarins. ChemistrySelect. 2017 Nov 13;2(32):10628-34.
24. An J, Kim S, Shrinidhi A, Kim J, Banna H, Sung G, et al. Purification of protein therapeutics via high-affinity supramolecular host–guest interactions. Nature Biomedical Engineering. 2020 Nov;4(11):1044-52.
25. reza Ariamoghaddam A, Ebrahimi-Hosseinzadeh B, Hatamian-Zarmi A, Sahraeian R. In vivo anti-obesity efficacy of curcumin loaded nanofibers transdermal patches in high-fat diet induced obese rats. Materials Science and Engineering: C. 2018 Nov 1;92:161-71.
26. Ganji RJ, Reddi R, Gumpena R, Marapaka AK, Arya T, Sankoju P, et al. Structural basis for the inhibition of M 1 family aminopeptidases by the natural product actinonin: Crystal structure in complex with E. coli aminopeptidase N. Protein Science. 2015 May;24(5):823-31.
27. Arulselvan P, Fard MT, Tan WS, Gothai S, Fakurazi S, Norhaizan ME, et al. Role of antioxidants and natural products in inflammation. Oxidative Medicine and Cellular Longevity. 2016 Oct 10;2016.
28. Astrup A. Healthy lifestyles in Europe: prevention of obesity and type II diabetes by diet and physical activity. Public Health Nutrition. 2001 Apr;4(2b):499-515.
29. Li M, Kim S, Lee A, Shrinidhi A, Ko YH, Lim HG, Kim HH, Bae KB, Park KM, Kim K. Bio-orthogonal supramolecular latching inside live animals and its application for in vivo cancer imaging. ACS Applied Materials & Interfaces. 2019 Nov 5;11(47):43920-7.
30. Atanasov AG, Zotchev SB, Dirsch VM, Supuran CT. Natural products in drug discovery: Advances and opportunities. Nature Reviews Drug Discovery. 2021 Mar;20(3):200-16.
31. Bakhaidar RB, Naveen NR, Basim P, Murshid SS, Kurakula M, Alamoudi AJ, et al. Response Surface Methodology (RSM) Powered Formulation Development, Optimization and Evaluation of Thiolated Based Mucoadhesive Nanocrystals for Local Delivery of Simvastatin. Polymers. 2022 Nov 28;14(23):5184.
32. Baek SJ, Hammock BD, Hwang IK, Li QX, Moustaid-Moussa N, Park Y, et al. Natural products in the prevention of metabolic diseases: lessons learned from the 20th KAST frontier scientists workshop. Nutrients. 2021 May 31;13(6):1881.
33. Li M, Lee A, Kim S, Shrinidhi A, Park KM, Kim K. Cucurbit [7] uril-conjugated dyes as live cell imaging probes: investigation on their cellular uptake and excretion pathways. Organic & Biomolecular Chemistry. 2019;17(25):6215-20.
34. Balakumar P, Maung-U K, Jagadeesh G. Prevalence and prevention of cardiovascular disease and diabetes mellitus. Pharmacological Research. 2016 Nov 1;113:600-9.
35. Komati A, Anand A, Shaik H, Mudiam MK, Babu KS, Tiwari AK. Bombax ceiba (Linn.) calyxes ameliorate methylglyoxal-induced oxidative stress via modulation of RAGE expression: identification of active phytometabolites by GC-MS analysis. Food & function. 2020;11(6):5486-97.
36. Salomon E, Schmitt M, Marapaka AK, Stamogiannos A, Revelant G, Schmitt C, et al. Aminobenzosuberone scaffold as a modular chemical tool for the inhibition of therapeutically relevant M1 aminopeptidases. Molecules. 2018 Oct 11;23(10):2607.
37. Egharevba GO, Dosumu OO, Oguntoye SO, Njinga NS, Dahunsi SO, Hamid AA, et al. Corrigendum to “Antidiabetic, antioxidant and antimicrobial activities of extracts of Tephrosia bracteolata leaves”[Heliyon 5 (8)(August 2019) e02275]. Heliyon. 2019 Sep 1;5(9).
38. Chandrasekhar S, Shrinidhi A. Selective aldehyde reduction in ketoaldehydes with NaBH4-Na2CO3-H2O at room temperatures. Synthetic Communications. 2014 Jul 18;44(14):2051-6.
39. Berthoud HR. Vagal and hormonal gut–brain communication: from satiation to satisfaction. Neurogastroenterology & Motility. 2008 May;20:64-72.
40. Ahmad R, AlLehaibi LH, AlSuwaidan HN, Alghiryafi AF, Almubarak LS, AlKhalifah KN, et al. Evaluation of clinical trials for natural products used in diabetes: An evidence-based systemic literature review. Medicine. 2021 Apr 4;100(16):e25641.
41. Ahmad R, AlLehaibi LH, Alshammari AK, Alkhaldi SM. Quality evaluation of the clinical trials for natural products used in cancer: An evidence-based literature review. Applied Sciences. 2020 Nov 10;10(22):7961.
42. Yin D, Hao J, Jin R, Yi Y, Bodduluri SR, Hua Y, et al. Epidermal Fatty Acid‒Binding Protein Mediates Depilatory-Induced Acute Skin Inflammation. Journal of Investigative Dermatology. 2022 Jul 1;142(7):1824-34.
43. Teschke R, Uetrecht J. Mechanism of idiosyncratic drug induced liver injury (DILI): Unresolved basic issues. Annals of Translational Medicine. 2021 Apr;9(8):730.
44. Chandrasekhar S, Shrinidhi A. Useful extensions of the Henry reaction: expeditious routes to nitroalkanes and nitroalkenes in aqueous media. Synthetic Communications. 2014 Oct 18;44(20):3008-18.
45. Kurakula M, Basim P. Biopolymer-Lipid Hybrid Composites and their Advances in Bio-imaging and Drug Delivery. Journal of Radiology and Medical Imaging. 2021;4(1):1041.
46. Pillalamarri V, Arya T, Haque N, Bala SC, Marapaka AK, Addlagatta A. Discovery of natural product ovalicin sensitive type 1 methionine aminopeptidases: molecular and structural basis. Biochemical Journal. 2019 Mar 29;476(6):991-1003.
47. Yücel Ç, Karatoprak GŞ, Aktaş Y. Nanoliposomal resveratrol as a novel approach to treatment of diabetes mellitus. Journal of Nanoscience and Nanotechnology. 2018 Jun 1;18(6):3856-64.
48. Mao CF, Sudirman S, Lee CC, Tsou D, Kong ZL. Echinacea purpurea ethanol extract improves male reproductive dysfunction with streptozotocin–nicotinamide-induced diabetic rats. Frontiers in Veterinary Science. 2021 Apr 28;8:651286.
49. Boltjes A, Shrinidhi A, van de Kolk K, Herdtweck E, Dömling A. Gd‐TEMDO: Design, Synthesis, and MRI Application. Chemistry–A European Journal. 2016 May 23;22(22):7352-6.
50. Bom MJ, Van Der Heijden DJ, Kedhi E, Van Der Heyden J, Meuwissen M, Knaapen P, et al. Early detection and treatment of the vulnerable coronary plaque: can we prevent acute coronary syndromes?. Circulation: Cardiovascular Imaging. 2017 May;10(5):e005973.
51. Wan S, Zhang L, Quan Y, Wei K. Resveratrol-loaded PLGA nanoparticles: enhanced stability, solubility and bioactivity of resveratrol for non-alcoholic fatty liver disease therapy. Royal Society Open Science. 2018 Nov 14;5(11):181457.
52. Bala SC, Haque N, Pillalamarri V, Reddi R, Kashyap R, Marapaka AK, et al. Discovery of a new class of type 1 methionine aminopeptidases that have relaxed substrate specificity. International Journal of Biological Macromolecules. 2019 May 15;129:523-9.
53. Perrin CL, Shrinidhi A, Burke KD. Isotopic-perturbation NMR study of hydrogen-bond symmetry in solution: temperature dependence and comparison of OHO and ODO hydrogen bonds. Journal of the American Chemical Society. 2019 Oct 8;141(43):17278-86.
54. Bottazzo G, Florin-Christensen A, Doniach D. Islet-cell antibodies in diabetes mellitus with autoimmune polyendocrine deficiencies. The Lancet. 1974 Nov 30;304(7892):1279-83.
55. Srujana MS, Thota RS, Anand A, Anusha K, Zehra A, Prasad K, et al. Raphanus sativus (Linn.) fresh juice priming moderates sucrose-induced postprandial glycemia as well as postprandial glycemic excursion in rats. Indian Journal of Traditional Knowledge. 2019 Apr;18(2):339-45.
56. Shrinidhi A. Diels‐Alder Reaction with Hydrophilic Dienes and Dienophiles. ChemistrySelect. 2016 Aug 1;1(12):3016-21.
57. Pavan Kumar P, Siva B, Anand A, Tiwari AK, Vekata Rao C, Boustie J, et al. Isolation, semi-synthesis, free-radicals scavenging, and advanced glycation end products formation inhibitory constituents from Parmotrema tinctorum. Journal of Asian Natural Products Research. 2020 Oct 2;22(10):976-88.
58. Bray MS, Loos RJ, McCaffery JM, Ling C, Franks PW, Weinstock GM, et al. NIH working group report—using genomic information to guide weight management: From universal to precision treatment. Obesity. 2016 Jan;24(1):14-22.
59. Chandrasekhar S, Shrinidhi A. Chloral hydrate as a water carrier for the efficient deprotection of acetals, dithioacetals, and tetrahydropyranyl ethers in organic solvents. Synthetic Communications. 2014 Jul 3;44(13):1904-13.
60. Cai W, Liu J, Zheng L, Xu Z, Chen J, Zhong J, et al. Study on the anti-infection ability of vancomycin cationic liposome combined with polylactide fracture internal fixator. International Journal of Biological Macromolecules. 2021 Jan 15;167:834-44.
61. Marapaka AK, Nocentini A, Youse MS, An W, Holly KJ, Das C, et al. Structural characterization of thiadiazolesulfonamide inhibitors bound to neisseria gonorrhoeae α-carbonic anhydrase. ACS Medicinal Chemistry Letters. 2022 Dec 6;14(1):103-9.
62. Hwang BW, Kim YE, Kim M, Han S, Bok S, Park KM, et al. Supramolecular hydrogels encapsulating bioengineered mesenchymal stem cells for ischemic therapy. RSC Advances. 2018;8(34):18771-5.
63. Capasso R, Izzo AA. Gastrointestinal regulation of food intake: general aspects and focus on anandamide and oleoylethanolamide. Journal of Neuroendocrinology. 2008 May;20:39-46.
64. Cazarolli LH, Pereira DF, Kappel VD, Folador P, Figueiredo MD, Pizzolatti MG, et al. Insulin signaling: a potential signaling pathway for the stimulatory effect of kaempferitrin on glucose uptake in skeletal muscle. European Journal of Pharmacology. 2013 Jul 15;712(1-3):1-7.
65. Perrin CL, Shrinidhi A. Enthalpic and entropic contributions to the basicity of cycloalkylamines. Chemical Science. 2020;11(32):8489-94.
66. Basim P, Gorityala S, Kurakula M. Advances in functionalized hybrid biopolymer augmented lipid-based systems: A spotlight on their role in design of gastro retentive delivery systems. Archives of Gastroenterology Research. 2021 Mar 2;2(1):35-47.
67. Shrinidhi A. ‘Reverse Micellar Aggregates’ in Phase Transfer Catalyzed Nucleophilic Displacement Reactions. ChemistrySelect. 2016 Jul 1;1(10):2479-84.
68. Chang W, Li K, Guan F, Yao F, Yu Y, Zhang M, et al. Berberine pretreatment confers cardioprotection against ischemia–reperfusion injury in a rat model of type 2 diabetes. Journal of Cardiovascular Pharmacology and Therapeutics. 2016 Sep;21(5):486-94.
69. Marapaka AK, Sankoju P, Zhang G, Ding Y, Ma C, Pillalamarri V, et al. Development of peptidomimetic hydroxamates as PfA-M1 and PfA-M17 dual inhibitors: Biological evaluation and structural characterization by cocrystallization. Chinese Chemical Letters. 2022 May 1;33(5):2550-4.
70. Chang W, Zhang M, Li J, Meng Z, Xiao D, Wei S, et al. Berberine attenuates ischemia-reperfusion injury via regulation of adenosine-5'-monophosphate kinase activity in both non-ischemic and ischemic areas of the rat heart. Cardiovascular Drugs and Therapy. 2012 Dec;26:467-78.
71. Shrinidhi A. Microwave-assisted chemoselective reduction of conjugated nitroalkenes to nitroalkanes with aqueous tri-n-butyltin hydride. Cogent Chemistry. 2015 Dec 31;1(1):1061412.
72. Chao IC, Chen Y, Gao MH, Lin LG, Zhang XQ, Ye WC, et al. Simultaneous determination of α-glucosidase inhibitory triterpenoids in Psidium guajava using HPLC–DAD–ELSD and pressurized liquid extraction. Molecules. 2020 Mar 11;25(6):1278.
73. Chen RC, Wang J, Yu YL, Sun GB, Sun XB. Protective effect of total saponins of Aralia elata (Miq) Seem on lipopolysaccharide-induced cardiac dysfunction via down-regulation of inflammatory signaling in mice. RSC Advances. 2015;5(29):22560-9.
74. Zhong X, Song R, Shan D, Ren X, Zheng Y, Lv F, et al. Discovery of hepatoprotective activity components from Thymus quinquecostatus celak. by molecular networking, biological evaluation and molecular dynamics studies. Bioorganic Chemistry. 2023 Nov 1;140:106790.
75. Marapaka AK, Pillalamarri V, Gumpena R, Haque N, Bala SC, Jangam A, et al. Discovery, Structural and Biochemical Studies of a rare Glu/Asp Specific M1 Class Aminopeptidase from Legionella pneumophila. International Journal of Biological Macromolecules. 2018 Dec 1;120:1111-8.
76. Khaltaev N, Axelrod S. Countrywide cardiovascular disease prevention and control in 49 countries with different socio-economic status. Chronic Diseases and Translational Medicine. 2022 Dec 25;8(04):296-304.
77. Shrinidhi A, Perrin CL. Cyclohexeno [3, 4] cyclodec-1, 5-diyne-3-ene: A Convenient Enediyne. Organic Letters. 2021 Aug 24;23(17):6911-5.
78. Anand A, Priyanka U, Nayak VL, Zehra A, Babu KS, Tiwari AK. Nutritional composition and antioxidative stress properties in boiled tuberous rhizome of Neel Kamal (Nymphaea nouchali Burm. f.). Indian Journal of Natural Products and Resources (IJNPR)[Formerly Natural Product Radiance (NPR)]. 2019 Jun 9;10(1):59-67.
79. Kieffer TJ. Gastro-intestinal hormones GIP and GLP-1. Annales d'endocrinologie. 2004 Feb 1;65(1):13-21.
80. Kurakula M, Patel DB, Patel B, Gorityala S, Basim P. Functionalized Nanocarriers for Drug delivery: Amalgam of Biopolymers and Lipids. Journal of Nanomedicine. 2021;4(1):1037.
81. Kim SH, Hur HJ, Yang HJ, Kim HJ, Kim MJ, Park JH, et al. Citrus junos tanaka peel extract exerts antidiabetic effects via AMPK and PPAR-both in vitro and in vivo in mice fed a high-fat diet. Evidence-based Complementary and Alternative Medicine. 2013 Jan 1;2013.
82. Reddi R, Ganji RJ, Marapaka AK, Bala SC, Yerra NV, Haque N, et al. Puromycin, a selective inhibitor of PSA acts as a substrate for other M1 family aminopeptidases: Biochemical and structural basis. International Journal of Biological Macromolecules. 2020 Dec 15;165:1373-81.
83. Park KM, Shrinidhi A, Murray J, Kim K. Guest‐responsive, non‐proteolytic harvest of a cell‐sheet using controllable host‐guest chemistry. Israel Journal of Chemistry. 2018 Apr;58(3-4):461-5.
84. Kurakula M, Gorityala S, Patel DB, Basim P, Patel B, Kumar Jha S. Trends of chitosan based delivery systems in neuroregeneration and functional recovery in spinal cord injuries. Polysaccharides. 2021 Jun 15;2(2):519-37.
85. Ma J, Liu Y, Valladolid-Acebes I, Recio-López P, Peng G, Li J, et al. ATF5 is a regulator of ER stress and β-cell apoptosis in different mouse models of genetic-and diet-induced obesity and diabetes mellitus. Cellular Signalling. 2023 Feb 1;102:110535.
86. Addlagatta A, Marapaka AK. Development of selective inhibitors against malarial M1 family aminopeptidase. Acta Crystallographica Section A Foundation and Advances. 2017 Jan 1; 73(a2):C237.
87. Abutaleb NS, Shrinidhi A, Bandara AB, Seleem MN, Flaherty DP. Evaluation of 1, 3, 4-Thiadiazole Carbonic Anhydrase Inhibitors for Gut Decolonization of Vancomycin-Resistant Enterococci. ACS Medicinal Chemistry Letters. 2023 Mar 21;14(4):487-92.
88. Anusha K, Anand A, Babu KS, Tiwari AK. Bombax ceiba calyces regulate carbohydrate and lipid digesting enzyme's actions, display insulin sensitizing and antioxidant activities in vitro: A nutritional and phytochemicals examination. Indian Journal of Traditional Knowledge. 2022 Apr;21(2):323-31.
89. Danan G, Teschke R. RUCAM in drug and herb induced liver injury: the update. International Journal of Molecular Sciences. 2015 Dec 24;17(1):14.
90. Addlagatta A, Gumpena R, Marapaka A, Allanki AD, Jun JH, Sijwali PS, et al. Study of quinoxaline-based new compounds against aminopeptidase N, and Plasmodium falciparum malarial parasite. Abstracts of Papers of the American Chemical Society. 2014 Mar 16;247:1155.
91. Sailakshmi AS, Anand A, Madhusudana K, Nayak VL, Zehra A, Babu KS, et al. Diospyros melanoxylon (Roxb.): A tribal fruit that maintains euglycemic state after consumption and cools oxidative stress. Indian Journal of Natural Products and Resources (IJNPR)[Formerly Natural Product Radiance (NPR)]. 2018 Dec 28;9(3):194-203.
92. Chow HC, So TH, Choi HC, Lam KO. Literature Review of Traditional Chinese Medicine Herbs–Induced Liver Injury From an Oncological Perspective With RUCAM. Integrative Cancer Therapies. 2019 Aug;18:1534735419869479.
93. Shrinidhi A, Perrin CL. Nucleophilic Addition of Enolates to 1, 4-Dehydrobenzene Diradicals Derived from Enediynes: Synthesis of Functionalized Aromatics. ACS omega. 2022 Jun 23;7(26):22930-7.
94. Teschke R, Eickhoff A, Schulze J, Danan G. Herb-induced liver injury (HILI) with 12,068 worldwide cases published with causality assessments by Roussel Uclaf Causality Assessment Method (RUCAM): an overview. Translational Gastroenterology and Hepatology. 2021;6:51.
95. Teschke R, Zhu Y, Jing J. Herb-induced liver injury in Asia and current role of RUCAM for causality assessment in 11,160 published cases. Journal of Clinical and Translational Hepatology. 2020 Jun 6;8(2):200.
96. Butera R, Shrinidhi A, Kurpiewska K, Kalinowska-Tłuścik J, Dömling A. Fourfold symmetric MCR's via the tetraisocyanide 1, 3-diisocyano-2, 2-bis (isocyanomethyl) propane. Chemical Communications. 2020;56(73):10662-5.
97. Priyanka U, Anand A, Bhargavi K, Zehra A, Tiwari AK. Presence of postprandial antidysmetabolic and antioxidative stress properties in aqueous methanol extract of seeds and tuber of aquatic food plant Nymphaea nouchali (Burm. f.). Cogent Food & Agriculture. 2016 Dec 31;2(1):1249172.
98. Teschke R, Danan G. Idiosyncratic drug-induced liver injury (DILI) and herb-induced liver injury (HILI): Diagnostic algorithm based on the quantitative Roussel Uclaf Causality Assessment Method (RUCAM). Diagnostics. 2021 Mar 6;11(3):458.
99. Shrinidhi A. Reaction of Henry adducts with aqueous sodium borohydride. Cogent Chemistry. 2015 Dec 31;1(1):1080210.
100. Kurakula M, Gorityala S, Basim P, Sun M, Mannem C. Rationalizing Nanodeliverables for Effective L-Asparaginase Delivery in Chemotherapy: Update 2020. Cancer Science & Research. 2021;4:1-7.
101. Lee RT, Libby P. The unstable atheroma. Arteriosclerosis, Thrombosis, and Vascular Biology. 1997 Oct;17(10):1859-67.
102. Lee YS, Olefsky J. Chronic tissue inflammation and metabolic disease. Genes & Development. 2021 Mar 1;35(5-6):307-28.
103. Lehnert T, Sonntag D, Konnopka A, Riedel-Heller S, König HH. Economic costs of overweight and obesity. Best practice & research Clinical Endocrinology & Metabolism. 2013 Apr 1;27(2):105-15.
104. Li D, Yao S, Zhou Z, Shi J, Huang Z, Wu Z. Hyaluronan decoration of milk exosomes directs tumor-specific delivery of doxorubicin. Carbohydrate Research. 2020 Jul 1;493:108032.
105. Komati A, Anand A, Nagendla NK, Madhusudana K, Mudiam MK, Babu KS, et al. Bombax ceiba calyx displays antihyperglycemic activity via improving insulin secretion and sensitivity: Identification of bioactive phytometabolomes by UPLC‐QTof‐MS/MS. Journal of Food Science. 2022 Apr;87(4):1865-81.
106. Li XL, Zhou J, Chen ZR, Chng WJ. P53 mutations in colorectal cancer-molecular pathogenesis and pharmacological reactivation. World Journal of Gastroenterology: WJG. 2015 Jan 1;21(1):84.
107. Lim HK, Bae W, Lee HS, Jung J. Anticancer activity of marine sponge Hyrtios sp. extract in human colorectal carcinoma RKO cells with different p53 status. BioMed Research International. 2014 Jan 1;2014.
108. Abenavoli L, Scarlata GG, Scarpellini E, Boccuto L, Spagnuolo R, Tilocca B, et al. Metabolic-Dysfunction-Associated Fatty Liver Disease and Gut Microbiota: From Fatty Liver to Dysmetabolic Syndrome. Medicina. 2023 Mar 17;59(3):594.
109. Shah HG, Rathod V, Basim P, Gajera B, Dave RH. Understanding the impact of multi-factorial composition on efficient loading of the stable ketoprofen nanoparticles on orodispersible films using Box-Behnken Design. Journal of Pharmaceutical Sciences. 2022 May 1;111(5):1451-62.
110. Liu N, Yang HL, Wang P, Lu YC, Yang YJ, Wang L, et al. Functional proteomic analysis revels that the ethanol extract of Annona muricata L. induces liver cancer cell apoptosis through endoplasmic reticulum stress pathway. Journal of Ethnopharmacology. 2016 Aug 2;189:210-7.
111. Deepthi S, Anusha K, Anand A, Manasa A, Babu KS, Mudiam MK, et al. Micronutrients and phytochemicals content in various rice (Oryza sativa Linn.) samples control carbohydrate digestion variedly and present differential antioxidant activities: an in vitro appraisal. Indian Journal of Traditional Knowledge. 2020 Oct;19(4):821-31.
112. Bastos RG, Rodrigues SD, Marques LA, de Oliveira CM, Salles BC, Zanatta AC, et al. Eugenia sonderiana O. Berg leaves: Phytochemical characterization, evaluation of in vitro and in vivo antidiabetic effects, and structure-activity correlation. Biomedicine & Pharmacotherapy. 2023 Sep 1;165:115126.
113. Basim P, Ajjarapu S, Kurakula M. Conductive polymers for drug and bioactives delivery. In: Ali N, Bilal M, Khan A, Nguyen TA, Gupta RK, (Eds). Smart Polymer Nanocomposites: Design, Synthesis, Functionalization, Properties, and Applications. Elsevier. 2023 Jan 1; pp. 263-78.
114. Lumeng CN, Saltiel AR. Inflammatory links between obesity and metabolic disease. The Journal of Clinical Investigation. 2011 Jun 1;121(6):2111-7.
115. Bhatt J, Shah H, Basim P, Morris K, Haware R. In Structure–Mechanics Study of Cocrystals to Optimize Tablet Size. InNIPTE Conference 2018.
116. Anand A, Priyanka U, Madhusudana K, Nayak VL, Zehra A, Babu KS, et al. Administration of roasted barley and roasted horse gram powders pacified chronic sucrose-induced dysglycemia and dyslipidemia in rats and exerted In Vitro potent antioxidative stress effect. Pharmacognosy Magazine. 2018;14(59s):s578-s590.
117. Ejeh S, Uzairu A, Shallangwa GA, Abechi SE, Ibrahim MT, Ramu R. Cheminformatics study of some indole compounds through QSAR modeling, ADME prediction, molecular docking, and molecular dynamic simulation to identify novel inhibitors of HCV NS5B protease. Journal of the Indian Chemical Society. 2023 Mar 1;100(3):100955.
118. Kumar TK, Siva B, Anand A, Anusha K, Mohabe S, Reddy AM, et al. Comprehensive Lichenometabolomic Exploration of Ramalina conduplicans Vain Using UPLC-Q-ToF-MS/MS: An Identification of Free Radical Scavenging and Anti-Hyperglycemic Constituents. Molecules. 2022 Oct 9;27(19):6720.
119. Teschke R, Xuan TD. Active nature based ingredients for drug discovery with pivotal role of clinical efficacy: Review and prospective. Journal of Modern Medicinal Chemistry. 2020 Apr 24;8(1):4-18.
120. Ahmad K, Shaikh S, Lim JH, Ahmad SS, Chun HJ, Lee EJ, et al. Therapeutic application of natural compounds for skeletal muscle-associated metabolic disorders: A review on diabetes perspective. Biomedicine & Pharmacotherapy. 2023 Dec 1;168:115642.
