Introduction
In 2020 we published a series of 18 patients who underwent neoadjuvant chemotherapy (NACT) and vaginal radical trachelectomy (VRT) as a fertility sparing alternative in stage 1B2 cervical cancer [1]. We concluded that this could be a safe fertility-sparing option in a selected group of women with stage 1B2 cervical cancer. We found a recurrence rate of 21% (three patients). All these patients had adenocarcinoma, lymphovascular space invasion (LVSI) present and a partial response to NACT. In our cohort in 78% of the women who were eligible fertility preservation was achieved. In this overview we provide an update of the data including an additional number of patients who have received this treatment in the meantime. Finally, we give advice about options for fertility sparing surgery.
Update of Data
Between our last published data and moment of writing, four more patients were eligible for NACT followed by VRT, and follow up for the entire group was extended with another 18 months. Between September 2009 and February 2021, 22 patients diagnosed with stage 1B2 cervical cancer (FIGO 2018) were scheduled to receive fertility-preserving treatment by neoadjuvant chemotherapy and VRT with pelvic lymph node dissection (intention to treat). One patient was excluded from all analyses as described in our study [1]. Median time of follow-up was 53.9 months (range 2.0-122.3). Patient-, tumor-, and treatment characteristics of all patients are provided in Table 1.
| Variable | Total (n=22) |
| Age, median (range) BMI, median (range) |
30 (25-36) 23.1 (20.0-30.8) |
| Histology Squamous cell carcinoma Adenocarcinoma |
16 (72.3) 6 (27.3%) |
| Grade 1 2 3 |
2 (9.1%) 12(54.5%) 8 (36.4% |
| LVSI Yes No |
13 (59.0%) 9 (41.0%) |
| Tumor size horizontal spread, median (mm, range) depth of invasion, median (mm, range) |
9 (20-38) 7 (5-16) |
| Number of lymph nodes, median (mm, range) | 21 (12-43) |
| Lymph node metastasisyes no |
1 (4.5%) 21 (95.5% |
| Follow up, median (months, range) | 53.9 (2.0-122.3) |
| Cycles of NACT (median, range) | 6 (3-6) |
| Recurrence Yes No |
4 (18.1%) 18 (81.8%) |
| Status Dead Alive |
1 (4.5%) 21 (95.5%) |
| NACT: Neoadjuvant Chemotherapy; BMI: Body Mass Index; LVSI: Lymph Vascular Space Invasion | |
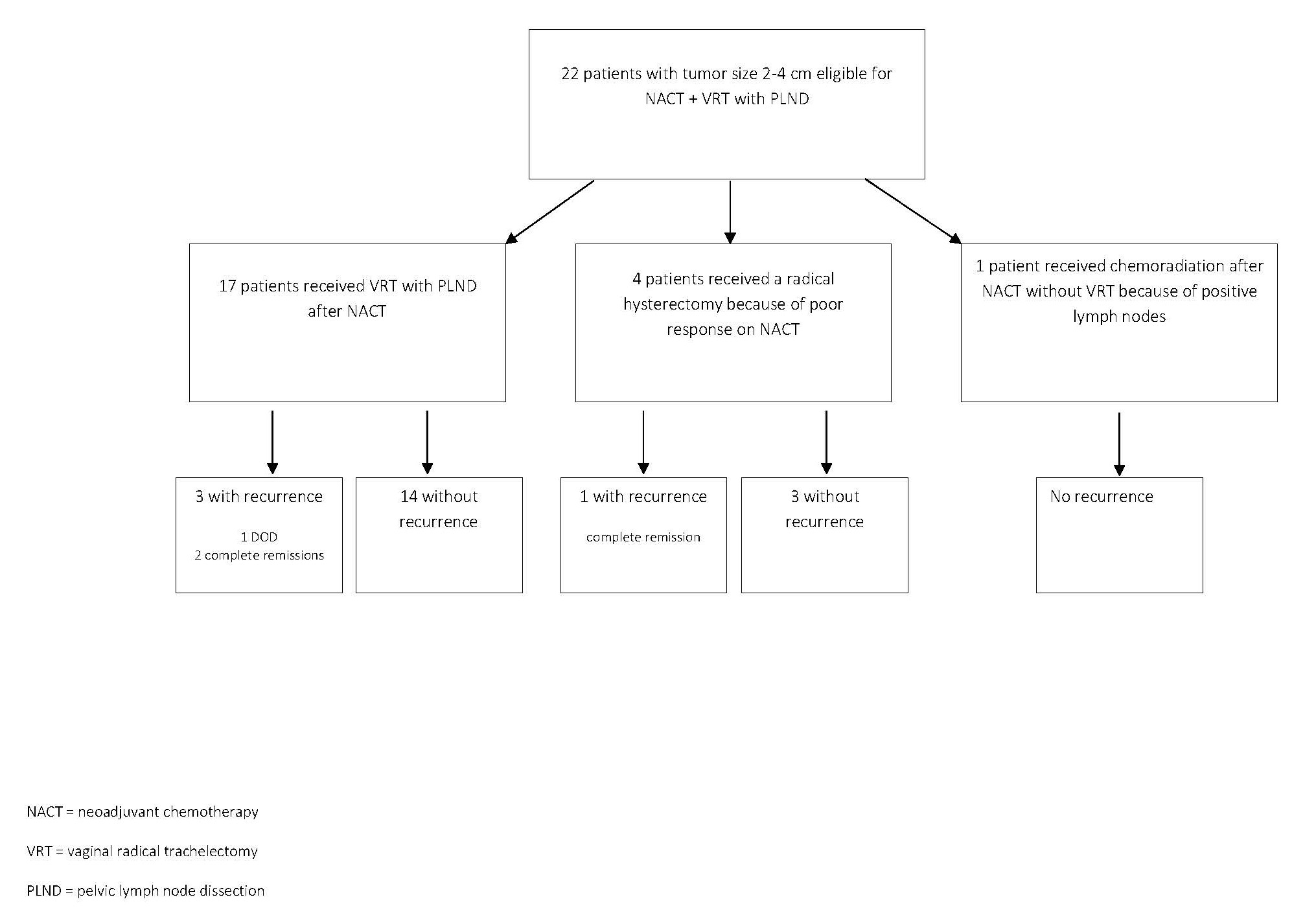
17 patients of the total underwent a VRT after chemotherapy while five patients did not get VRT after chemotherapy. One patient had a suspicious lymph node during surgery. Frozen section analysis showed metastatic disease. The VRT was cancelled, and chemoradiation was given instead. Four patients had a poor response on chemotherapy with a remaining tumor size larger than 2 cm after chemotherapy. They underwent a radical hysterectomy with pelvic lymph node dissection (PLND) (Figure 1). In one of them the final histology report showed a tumor size less than 2 cm, whereas the other three indeed had a tumor size over 2 cm in final histology.
Figure 1.
Oncological outcome
Four patients developed a recurrence: three after VRT and one in a poor-responder on NACT after radical hysterectomy. Recurrences were localized in the pelvis in three women, in one woman the recurrence had spread to higher lymph nodes outside the pelvis. Median time to recurrence was 23.6 months (range 18.1–105.7). One recurrence occurred in a patient with squamous cell carcinoma (LVSI positive), three recurrences occurred in patients with adenocarcinoma, in which also LVSI was present. All patients with recurrent disease had residual disease after NACT. Three of the four women who developed recurrent disease are still alive and had a complete remission after chemoradiation (n=1) or surgery followed by chemoradiation (n=2). Unfortunately, one young woman deceased after treatment with radical hysterectomy for a recurrence and additionally chemotherapy and a human papillomavirus 16 cancer vaccine (ISA101/ISA101b).
Fertility and obstetrical outcomes
Table 2 describes fertility and obstetrical outcomes. Sixteen out of 22 patients (72.7 %) had fertility preserved. Six patients lost their fertility: three due to radical hysterectomy after poor response on chemotherapy, one received adjuvant radiotherapy because of a positive lymph node, and two developed premature ovarian insufficiency (POI), most likely as a consequence of the chemotherapy.
| n(%) | |
| VRT Preserved fertility Active desire to conceive Referred to fertility treatment Pregnancies |
16 out of 22 (72.7%) 8 out of 16 (50.0%) 5 out of 8 (62.5%) 6 |
| Conception Spontaneous After IUI After IVF/ IVF-ICSI |
2 2 2 |
| Outcome pregnancies (n=4) Abortion first trimester 32-36 WOG >37 WOG |
2 1 3 |
| VRT: Vaginal Radical Trachelectomy; IUI: Intra-Uterine Insemination; IVF: In vitro Fertilisation; ICSI: Intracytoplasmatic Sperm Injection; WOG: Weeks of Gestation | |
Data on fertility rates are missing for 2 women as they were lost to follow-up since our last published study.
Five patients underwent in vitro fertilization (IVF) treatment just before NACT was started. Two of them succeeded in obtaining oocytes for vitrification. In two patients IVF was unsuccessful because the response to ovarian hyperstimulation was very poor.
Of the 16 women with preserved fertility, to date at least eight expressed the desire to conceive (in two patients unknown). Five of them were referred to a fertility clinic for intra-uterine insemination or IVF.
Currently, four women had six pregnancies, including four live births born after 32 weeks of pregnancy and two first trimester miscarriages. One of the patients who underwent a radical hysterectomy had frozen embryos that were used for surrogacy. The surrogate mother is now pregnant.
Discussion
Comparison to new literature
Since our study, Kasius et al. published a robust literature overview of neoadjuvant chemotherapy in fertility sparing cervical cancer treatment [2]. They concluded a recurrence rate of 13% after NACT and trachelectomy (either abdominal or vaginal) and a live birth rate of 24%. However, this is based on heterogenic patient populations with sometimes unfavorable factors (such as tumor size, non-squamous pathology or LVSI present). Also, the treatment protocol itself varied among studies: different chemotherapy schedules were used, and surgical procedures varied in radicality.
Literature remains heterogenic and consists currently of small cohort studies only. After publication of our paper, one other publication appeared from another Dutch group [3]. This study assessed the feasibility, safety, oncological and obstetric outcomes in patients with cervical tumors 1B2 of 1B3 (FIGO 2018) with neoadjuvant chemotherapy in preparation for abdominal radical trachelectomy. Their cohort consisted of 19 patients. In line with our paper, they concluded that it may be a feasible and safe option in a selection of patients.
There are also some differences. They included patients with larger tumors. The mean clinical tumor size was 4.4 cm, ranging from 3.5 to 6 cm. In our study, tumor size was deliberately 4 cm at the most. The neoadjuvant chemotherapy regime was similar (i.e. cisplatin and paclitaxel, 6 cycles), but they performed abdominal radical trachelectomy (ART) after NACT instead of VRT.
ART is believed to have less results with regards to fertility preservation and obstetrical outcomes [4]. In that respect, reduction of the tumor with neo-adjuvant chemotherapy to perform less radical surgery and improve fertility outcomes seems more logical, i.e., simple or radical vaginal trachelectomy. In addition, for women with stage 1B2 (FIGO 2018) cervical cancer who are undergoing ART, it would be preferable to perform ART without NACT. Chemotherapy carries a risk of premature ovarian insufficiency as we observed in our series of patients. A systematic review including ten studies [4] showed more pregnant women in the NACT followed by VRT group than in the ART group (21% versus 7%). More women attempted to conceive and succeeded after NACT followed by VRT than after ART (70% versus 21%). Finally, they showed a higher chance on a life birth in women treated with NACT followed by VRT (63% versus 43% in ART). Nevertheless, although numbers do not correspond to other studies, Tesfai et al. found similar pregnancy rates after NACT followed by ART in their patients with 1B2 or 1B3 cervical cancer compared to NACT and VRT [3].
Aside from a vaginal or open abdominal approach, the trachelectomy could also be performed as a minimally invasive procedure. The minimally invasive approach might have some important peri-operative advantages, but since the publication of the LACC trial concerns exist about the safety of this approach in women with cervical cancer [5]. Literature is very scarce when it comes to laparoscopic or robot-assisted trachelectomy. Studies have a retrospective nature and numbers are small. A multi-institutional international registry study (International Radical Trachelectomy Assessment – IRTA – study) recently completed the assessment of open versus minimally invasive radical trachelectomy [6]. Preliminary results have shown similar progression-free and overall survival for patients with tumors up to 2 cm undergoing open abdominal radical trachelectomy versus minimally invasive radical trachelectomy. It is not yet known how this approach impacts radicality of the procedure or fertility numbers.
The CONTESSA/NEOCON-F study is a multicenter, prospective phase 2 study in which the safety of neoadjuvant chemotherapy and fertility-sparing surgery in patients with cervical cancer FIGO stage (2018) IB2 (2–4 cm) and a wish to preserve fertility is evaluated [7]. The advantage of this trial is that certain aspects are standardized, such as timing and mode of evaluation of response to NACT, but which chemotherapy used is up to the participating center. This large prospective trial is expected to provide more uniform evidence on the oncological safety and fertility and obstetrical outcome after fertility-sparing treatment in this group of patients.
Only patients with a complete or partial response (residual tumor <2 cm) will receive fertility-sparing treatment that consists of a simple trachelectomy or conization to enlarge their pregnancy rate. ART and VRT – which are with respect to the parametrium more radical procedures - will not be one of the surgical options. Several retrospective studies have indeed shown very low rates of parametrial involvement in patients with stage IA2 and IB1 (FIGO 2018) cervical cancer with favorable pathologic characteristics who have undergone radical hysterectomy [8]. This raises the question whether these patients might be eligible to less radical surgery, such as simple hysterectomy or conization combined with bilateral PLND. Data on this subject are expected from 3 studies: ConCerv, SHAPE, and GOG 278. The SHAPE trial is a randomized trial, comparing simple hysterectomy and radical hysterectomy in this group of cervical cancer patients. The objective is to determine if overall survival is significantly different between the two procedures and whether simple hysterectomy is associated with less morbidity. Importantly, all tumor grades were eligible as well as the presence of LVSI [9]. As Kasius and colleagues stated, it remains the question whether the results of these trials can be translated to patients with cervical tumors <2 cm after receiving NACT [2].
In our presented series of patients who were intended to undergo NACT and VRT with PLND, recurrences occurred in women with unfavorable pathological characteristics, such as non-squamous histology and LVSI present. This is in line with findings in the study of Tesfai and colleagues [3] in which ART, a more radical procedure than VRT, is performed after NACT. It is noteworthy that these recurrences are often distant metastasis and not local. It is the question if outcomes for these women would have been the same if they would have undergone a radical hysterectomy instead. However, unfavorable prognostic factors defined as non-responsiveness and non-squamous pathology and LVSI present can help in patient selection for fertility-sparing surgery.
Conclusion
Neoadjuvant chemotherapy followed by some form of fertility sparing surgery seems feasible in women with stage 1B2 (FIGO 2018) cervical cancer. While awaiting the results of the SHAPE trial and NEOCON study we recommend performing a vaginal radical trachelectomy as the middle ground between abdominal radical trachelectomy and simple trachelectomy or conization on the other end of the spectrum.
The most important issue with regards to oncological safety of the procedure is the assessment of those patients who would be eligible. Prognosis seems worse in patients with unfavorable characteristics, such as non-squamous histology, presence of LVSI, and high differentiation grade.
Taking all these considerations into account, careful counseling of women with a wish for fertility preservation, using shared decision-making principles, is very important.
References
2. Kasius JC, van der Velden J, Denswil N, Tromp J, Mom C. Neo-adjuvant chemotherapy in fertility-sparing cervical cancer treatment. Best Practice & Research Clinical Obstetrics & Gynaecology 2021.
3. Tesfai F, Kroep J, Gaarenstroom K, de Kroon C, van Loenhout R, Smit V, et al. Fertility-sparing surgery of cervical cancer >2 cm (International Federation of Gynecology and Obstetrics 2009 stage IB1–IIA) after neoadjuvant chemotherapy. Int J Gynecol Cancer. 2020 Jan;30(1):115-121.
4. Van Kol N, Vergeldt TFM, Bekkers RLM. Abdominal radical trachelectomy versus chemotherapy followed by vaginal radical trachelectomy in stage 1B2 (FIGO 2018) cervical cancer. A systematic review on fertility and recurrence rates. Gynecol Oncol. 2019 Dec;155(3):515-521.
5. Ramirez P, Frumovitz M, Pareja R, Lopez A, Vieira M, Ribeiro R, et al. Minimally Invasive versus Abdominal Radical Hysterectomy for Cervical Cancer. N Engl J Med. 2018;379:1895-1904.
6. Salvo G, Pareja R, Ramirez PT. Minimally invasive radical trachelectomy: Considerations on surgical approach. Best Pract Res Clin Obstet Gynaecol. 2021 Feb 20:S1521-6934(21)00018-3.
7. Plante M, van Trommel N, Lheureux S, Oza AM, Wang L, Sikorska K, et al. FIGO 2018 stage IB2 (2-4 cm) cervical cancer treated with neo-adjuvant chemotherapy followed by fertility sparing surgery (CONTESSA); neo-adjuvant chemotherapy and conservative surgery in cervical cancer to preserve fertility (NEOCON-F). A pmhc, dgog, GCIG/CCRN and multicenter study. Int J Gynecol Canc. 2019;29(5):969-975.
8. Ramirez PT, Pareja R, Rend GJ. Management of lowrisk early-stage cervical cancer: Should conization, simple trachelectomy, or simple hysterectomy replace radical surgery as the new standard of care? Gynecol Oncol. 2014 Jan;132(1):254-259.
9. Plante M. The SHAPE trial. http://www.gcig.igcs.org/ Spring2012/2012_june_shape_trial.pdf