Abstract
Background: Metabolic syndrome (MS) is an important clinical and public health problem, especially in Western society, and is affecting more and more people worldwide. Although most cases of MS are considered obese and insulin-resistant, they are characterized by inflammatory cytokine activity and chronic low-grade inflammation. Also, assumptions about the role of MS and organ interaction during target organ damage become increasingly important.
Method: For this study, online research and screening were carried out in electronic databases such as PubMed, Web of Science, and Google Scholar; using the terms “metabolic syndrome” and “organ crosstalk” as keywords.
Results: Due to the unpredictable clinical course and treatment uncertainty in this multi-factor chronic disease, increased organ interaction and complications lead to an increase of biomarkers in blood circulation, each of which has diagnostic potential. Therefore, a better understanding of inflammatory triggers and organ interferences will also help determine therapeutic targets and prevent organ damage associated with metabolic syndrome.
Conclusion: This mini review presents an overview of the relationship between MS and organ crosstalk. More research is needed to fully elucidate the relationship between MS and organ crosstalk, the pathological conditions it causes, and biomarkers related to MS.
Keywords
Metabolic syndrome, Organ crosstalk, Inflammation, COVID-19, Biomarker
Introduction
Today, metabolic syndrome (MS) has been regarded as a very important disease due to its complex multifactorial etiology and damage to different organs. In general, obesity, dyslipidemia, hypertension, hyperglycemia, and insulin resistance are the main metabolic abnormalities of the MS. Compared with the others; in particular, insulin resistance and central obesity are considered the main causes in the pathogenesis [1,2]. The interactions between organs in the human body are called ‘organ crosstalk’. This can be part of physiological reflexes that protect the organs during various stimuli or can be part of pathological conditions where an injury to an organ releases cytokines that causes several effects on other organs [3]. In response to various metabolic needs, the human body has developed an interorgan communication system where individual tissues can affect the metabolic pathways of distant tissues. In the MS, dysregulation of this communication system plays an important role in diseases including diabetes, obesity, atherosclerosis, kidney and liver diseases [4,5]. It is known that organ crosstalk is altered in pathological conditions such as obesity and type 2 diabetes, and MS could be considered as a systemic disease since its pathology involves the interplay of multiple organs [5]. Also, adipose tissue is recognized to be an active metabolic and endocrine organ which directly and indirectly contributes to the metabolic organ interactions and related diseases (Figure 1) [6,7]. Furthermore, adipokines and myokines play a crucial role in the interactions among adipose tissue, skeletal muscle and other organs [8]. While the studies in human and animal models confirm the relationship between gut microbiota and MS, it is claimed that changes in host-microbiota interactions may be responsible for the current MS prevalence [9]. Notably, many circulating mediators and biomarkers have been reported in recent publications, which are evidence of MS-related organ interactions (Table 1) [5,8].
| Related organ or tissue | Related or associated mediator or markers* | Target or affected organs |
|---|---|---|
| Liver-derived and associated biomarkers | Adropin, Hepassocin, Selenoprotein P, Fetuin A, FGF21, RBP4, etc. |
Adipose tissue, muscle, gut and brain |
| Adipose tissue-derived mediator/markers | Leptin, Adiponectin, FABP4, Lipocalin-2, etc. |
Liver, muscle, and brain |
| Vasculature-associated biomarkers |
GLP-1, Ghrelin, Adiponectin, L5, ANG1 and 2, Irisin, FABP4, ANGPTL3,4,8, etc. | The vasculature is affected by many mediators secreted by adipose tissue, liver, muscle, and gut |
| Muscle-derived mediator/ markers | IL-6, Myostatin, Irisin, IL-15, MOTS-c, etc. | Adipose tissue and liver |
| Gut-derived and endocrine mediator/markers | Insulin, Ghrelin, Glucagon, Sero- tonin, GIP, GLP-1 | Adipose tissue, liver, muscle, and muscle |
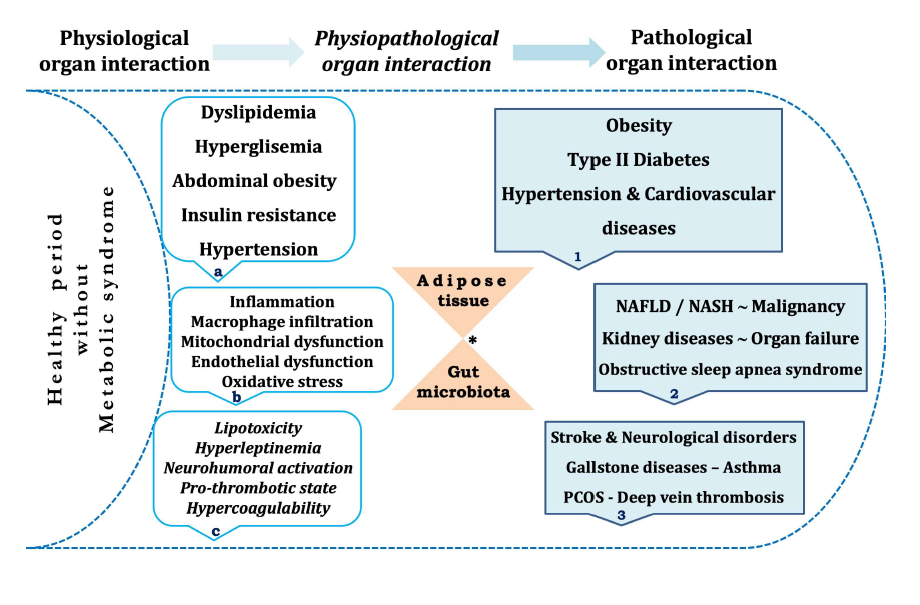
* While interaction of inflammatory markers and adipocytokines contribute to MS development, ongoing disease and its complications also contribute many organ crosstalk and related diseases or organ failure over the years
Table 1: The biomarkers that play a role in the pathogenesis and progression of metabolic syndrome as well as inter- organ interaction can be used for diagnostic purposes.
Aim and Methods
Since MS and obesity both damaged several tissues and organs, it was aimed to draw attention to the relationship between MS and organ crosstalk. Also, analyzing the original aspects of MS and organ interaction, this mini review will help promote healthy living and develop new diagnostic and treatment strategies for metabolic diseases. For this purpose, it was performed by using electronic database search which included PubMed, Web of Science, and Google Scholar. The terms ‘metabolic syndrome’ and ‘organ crosstalk’ or ‘organ interaction’ have been used as keywords for online research and screening.
Discussion
Physiological and pathological organ crosstalk
Crosstalk between organs is part of the normal physiology of human body providing an important physiological function responsible for maintenance of whole metabolic homeostasis. Any organ injuries may be potentiated through crosstalk, resulting in a far greater burden of illness when compared to those situations in which dysfunctions of organs are created individually. Also, there may be pathophysiological pathways of organ crosstalk that could be targets for specific intervention. Coordinated responses to inflammation across many organ systems are part of the physiological responses to the organ injury and metabolic changes [10]. For example, cytokine driven epithelial and endothelial dysfunction is a crucial causative factor in the pathological organ interactions. Also, mitochondrial and immune cell dysfunctions are the other causes. Usually, increased circulating molecules such as damaged tissue-induced DAMP and PAMP caused by low-grade inflammation and gut microbiota are the other contributing factors to this process [3,7].
Relationship between metabolic syndrome and organ interaction
In the PubMed and the other indexes, the data show that there has been a notable increase in publications about the organ crosstalk over the last decade [3,5]. Previously, Lusis et al. [4] proposed a system-based approach to the complex pathogenesis of MS, unlike the traditional genetic and biochemical approach. According to them, there were many interactions between the components of MS traits associated with diabetes and cardiovascular diseases. As we all know, good blood sugar control can bring better clinical results in diabetic patients, thereby delaying cardiovascular complications and organ damage [1]. It has been reported by some other authors that adipokine, cytokine and inflammatory mediators produced in adipose tissue and liver are effective in inter-organ interaction and contribute to insulin resistance [11,12]. Metabolic syndrome, which is also closely related to obesity with multiorgan involvement, is characterized by endothelial dysfunction, atherogenic dyslipidemia, visceral adiposity, and insulin resistance. These disorders are interrelated and share common pathophysiological mechanisms, pathways, and mediators [6]. Insulin resistance and hyperglycemia, which become chronic over time, not only affect the cardiovascular system, kidneys, and liver but also may play a role in long-term damage and failure of these organ systems. In fact, it can be said that MS is an important cornerstone in terms of the interaction between organs, and therefore, it contributes significantly to the pathogenesis and course of organ crosstalk. For instance, MS stimulates the development of the premature atherosclerotic disease, and it is closely related to the occurrence of other cardiovascular complications such as heart attack and cardiac failure [13]. Similarly, insulin resistance and adipokine-mediated metabolic dysregulation have been associated with NAFLD, and hyperinsulinemia may induce inflammatory processes by leading to liver damage [12,14]. Indeed, the development of NAFLD in obese and MS patients is a typical example of organ interaction. Also, musculoskeletal pathologies have emerged as another area associated with MS-related conditions [8]. Yeo et al. [15] reported that gut-skeletal muscle or gut-liver interactions play a crucial role in the development of the insulin resistance of skeletal muscle and NAFLD.
In the MS, inflammation (metaflammation) induced by excessive nutrient intake, recurrent oxidative stress, and mitochondrial dysfunction changes lead to progressive tissue damage in the heart, liver, and other organs over time [3,15]. Adipose tissue, skeletal muscle, pancreas and liver are themselves sites of inflammation in the MS and obesity. Low-grade inflammation of adipose tissue is an important step contributing to pathological organ abnormalities and symbolizes the relationship with MS as in the examples of diabetes, liver, kidney, and cardiovascular diseases [16]. Moreover, patients with MS have an increased frequency of renal vascular sclerosis, tubulointerstitial fibrosis, and tubular atrophy [17]. Although obesity, insulin resistance, and the MS are associated with changes to the gut microbiota, the mechanism of adipose tissue-gut microbiota interaction in the patients with MS is unknown fully. In patients with MS, both gut microbiota and visceral adipose tissue are thought to be responsible for inflammatory changes and damage to target organs over time [9,18]. In particular, NAFLD, which is frequently seen in MS and obese patients, is described as an epiphenomenon of the organ crosstalk and interaction [14].
Biomarkers and mediators associated with organ interaction in metabolic syndrome
Circulating biomarkers diversity is a fledgling but rapidly growing issue in the diagnosis and treatment of MS. Besides genetic biomarkers, MS-associated markers include dyslipidemia, oxidative stress, inflammatory, and cardio-metabolic biomarkers [19]. A number of circulating biomarkers related to obesity and metabolic dysfunction could serve as the risk of MS, the indicator of metabolic organ interactions, and early markers of elevated another disease risk such as type 2 diabetes and NAFLD [3]. The interaction of inflammatory markers, cytokines, and adipokines contributes to the interaction between MS and related organs. While chronic inflammation leads to abnormal cytokine production in obesity and MS, RNA expressions of proinflammatory cytokines, especially TNF-α, IL-1, and IL-6, increase in patients with obesity [20]. Additionally, as miRNAs are released into the bloodstream, they serve as key messengers between cells and tissues, participating in the metabolic organ crosstalk and facilitating interaction [21]. Besides, the increase in circulating modified LDL rates leads to several metabolic disorders associated with high cardiovascular risk and systemic inflammation [22]. At the systemic level, changes in circulating mediator levels such as TNF-α and adiponectin lead to decreased liver and muscle insulin sensitivity through inflammation, toxicity, and increased accumulation of ectopic lipids [8,23]. Srikanthan et al. [24] reported a diagnostic marker panel consisting of the levels of leptin, adiponectin, paraoxonase, Ox-LDL, IL-6, IL-10, TNF-α, uric acid, PAI-1, and ghrelin in the circulation of patients with MS.
Metabolic syndrome is a progressive disease involving various diseases with specific abnormalities at different time intervals. These abnormalities can be diagnosed and monitored by serum biomarkers [24]. Therefore, creating a biomarker panel with known and predictable links to MS can be a tool to identify people at risk and intervene if necessary. In a recent study of Priest and Toronto [5], attention is drawn into inter-organ crosstalk in MS, and many mediators and biomarkers are reported to be secreted from the liver, adipose tissue, skeletal muscle, vascular system, intestine, and endocrine system (Table 1). Because MS affects multiple interacting organ systems (for being a systemic disease), system-level analysis of disease etiopathogenesis is essential for both complete elucidations of its pathophysiology and new treatment methods [25]. For instance, the administration of recombinant GDF15 improves insulin resistance and glucose tolerance by decreasing food intake [26]. Therefore, for circulating biomarkers, improvements in risk estimates have the potential to positively contribute to MS diagnosis and treatment processes. Also, MS is a risk factor that influences COVID-19 progression and prognosis. Cytokine release syndrome -an acute systemic inflammatory immune response- is the major hallmark of the COVID-19, which is characterized by fever and multiple organ dysfunction [27]. Therefore, the rapid course of COVID-19 cases with obese, diabetic, hypertensive, or liver damage indicates the importance of this risk group for monitoring and treatment. As in the control of other epidemics, education of the population about the health hazard of the MS would be very important. Specifically, to prevent MS before it happens, targeting the young population may be a better plan than trying to improve it once it happens.
Conclusion
Despite the increasing number of publications on organ interaction, what is known about the mechanism of crosstalk and the detailed role of interactions is not yet sufficient. However, hypotheses about the role of organ interaction in the course of MS and target organ damage are becoming increasingly important. Due to its complex multifactorial etiology, MS causes damage to the many organs. Specifically, insulin resistance plays a central role in MS pathogenesis and promotes the majority of metabolic impairments, including the organ diseases that originate from MS. For example, the change in white fat tissue and adipocytokine release due to obesity characterizes lowgrade inflammation involved in the pathogenesis of liver and heart disorders. As individuals with MS have signs of organ damage such as inflammation and metabolic dysregulation, this and similar changes should be handled holistically for diagnostic purposes, and also treatment management should be directed accordingly. Hopefully, ongoing studies in this area may open new avenues for the diagnosis and management of MS.
Conflicts of Interest
The authors declare no conflict of interest.
Author Contribution
Review concept and design: FA; drafting of the manuscript: FA and SA; critical revision of the manuscript: All authors.
List of Abbreviations
ANG1: Angiopoietin 1; ANGPTL3: Angiopoietin-like; CRP: C-reactive Protein; DAMPs: Damage-associated Molecular Patterns; FABP4: Fatty Acid-binding Protein 4; FGF21: Fibroblast Growth Factor; GDF15: Growth/ differentiation Factor 15; GIP: Glucose-dependent Insulinotropic polypeptide; GLP-1: Glucagon-like Peptide-1; IL-6: Interleukin-6; L5: Electronegative low-density lipoprotein; LPS: Lipopolysaccharide; MCP- 1: Monocyte Chemoattractant Protein-1; MOTS-c: Mitochondrial-encoded peptide; NAFLD: Nonalcoholic Fatty Liver Disease; PAI-1: Plasminogen Activator Inhibitor-1; PAMPs: Pathogen-associated Molecular Patterns; PON-1: Paraoxonase 1; Ox-LDL: Oxidized Lowdensity Lipoprotein; RBP4: Retinol-Binding Protein; TNF-α: Tumor Necrosis Factor Alpha.
References
2. Mendrick DL, Diehl AM, Topor LS, Dietert RR, Will Y, La Merrill MA, et al. Metabolic syndrome and associated diseases: from the bench to the clinic. Toxicological Sciences. 2018 Mar 1;162(1):36-42.
3. Armutcu F. Organ crosstalk: the potent roles of inflammation and fibrotic changes in the course of organ interactions. Inflammation Research. 2019 Jul 20;68:825- 39.
4. Lusis AJ, Attie AD, Reue K. Metabolic syndrome: from epidemiology to systems biology. Nature Reviews Genetics. 2008 Nov;9(11):819-30.
5. Priest C, Tontonoz P. Inter-organ cross-talk in metabolic syndrome. Nature Metabolism. 2019 Dec 9:1-2.
6. Romacho T, Elsen M, Röhrborn D, Eckel J. Adipose tissue and its role in organ crosstalk. Acta physiologica. 2014 Apr;210(4):733-53.
7. Chung HS, Choi KM. Adipokines and myokines: a pivotal role in metabolic and cardiovascular disorders. Current Medicinal Chemistry. 2018 Jun 1;25(20):2401-15.
8. Collins KH, Herzog W, MacDonald GZ, Reimer RA, Rios JL, Smith IC, Zernicke RF, Hart DA. Obesity, metabolic syndrome, and musculoskeletal disease: common inflammatory pathways suggest a central role for loss of muscle integrity. Frontiers in Physiology. 2018 Feb 23;9:112.
9. Festi D, Schiumerini R, Eusebi LH, Marasco G, Taddia M, Colecchia A. Gut microbiota and metabolic syndrome. World Journal of Gastroenterology: WJG. 2014 Nov 21;20(43):16079.
10. Prowle JR. Organ cross-talk in shock and critical illness. ICU Management & Practice 2018;18(3).
11. Gancheva S, Jelenik T, Alvarez-Hernandez E, Roden M. Interorgan metabolic crosstalk in human insulin resistance. Physiological Reviews. 2018 Jul 1;98(3):1371- 415.
12. Alam S, Mustafa G, Alam M, Ahmad N. Insulin resistance in development and progression of nonalcoholic fatty liver disease. World Journal of Gastrointestinal Pathophysiology. 2016 May 15;7(2):211.
13. Esser N, Paquot N, Scheen AJ. Inflammatory markers and cardiometabolic diseases. Acta Clinica Belgica. 2015 Jun 1;70(3):193-9.
14. Zhang X, Ji X, Wang Q, Li JZ. New insight into interorgan crosstalk contributing to the pathogenesis of nonalcoholic fatty liver disease (NAFLD). Protein & cell. 2018 Feb 1;9(2):164-77.
15. Yeo YH, Lai YC. Redox regulation of metabolic syndrome: recent developments in skeletal muscle insulin resistance and non-alcoholic fatty liver disease (NAFLD). Current Opinion in Physiology. 2019 Jun 1;9:79-86.
16. Esser N, Legrand-Poels S, Piette J, Scheen AJ, Paquot N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Research and Clinical Practice. 2014 Aug 1;105(2):141-50.
17. Singh AK, Kari JA. Metabolic syndrome and chronic kidney disease. Current opinion in nephrology and hypertension. 2013 Mar 1;22(2):198-203.
18. Kuvat N, Tanriverdi H, Armutcu F. The relationship between obstructive sleep apnea syndrome and obesity: A new perspective on the pathogenesis in terms of organ crosstalk. The Clinical Respiratory Journal. 2020 Feb 29;14(7):595-604.
19. Robberecht H, Hermans N. Biomarkers of Metabolic Syndrome: Biochemical Background and Clinical Significance. Metabolic Syndrome and Related Disorders. 2016 Mar 1;14(2):47-93.
20. Ji C, Guo X. The clinical potential of circulating microRNAs in obesity. Nature Reviews Endocrinology. 2019 Oct 14:1-3.
21. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006 Dec;444(7121):860-7.
22. Akyol S, Lu J, Akyol O, Akcay F, Armutcu F, Ke LY, Chen CH. The role of electronegative low-density lipoprotein in cardiovascular diseases and its therapeutic implications. Trends in Cardiovascular Medicine. 2017 May 1;27(4):239-46.
23. Cao H. Adipocytokines in obesity and metabolic disease. Journal of Endocrinology. 2014 Feb 1;220(2):T47- 59.
24. Srikanthan K, Feyh A, Visweshwar H, Shapiro JI, Sodhi K. Systematic review of metabolic syndrome biomarkers: a panel for early detection, management, and risk stratification in the West Virginian population. International Journal of Medical Sciences. 2016;13(1):25.
25. Saklayen MG. The Global Epidemic of the Metabolic Syndrome. Current hypertension reports. 2018 Feb 1;20(2):12.
26. Emmerson PJ, Wang F, Du Y, Liu Q, Pickard RT, Gonciarz MD, et al. The metabolic effects of GDF15 are mediated by the orphan receptor GFRAL. Nature medicine. 2017 Oct;23(10):1215-1219.
27. Costa FF, Rosário WR, Farias AC, de Souza RG, Gondim RS, Barroso WA. Metabolic syndrome and COVID-19: An update on the associated comorbidities and proposed therapies. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2020 Jun 11;14(5):809-814.
