Short Communication
Lower extremity fractures in individuals with a spinal cord injury (SCI) cause significant morbidity [1] and contribute to excess mortality [2]. Early identification of persons at highest risk for fracture is possible using bone mineral density (BMD) testing by dual-energy X-ray absorptiometry (DXA) imaging [3]. Recent guidelines by the International Society for Clinical Densitometry (ISCD) recommend that all adults with a SCI resulting in permanent motor or sensory dysfunction should have a DXA scan of the total hip, proximal tibia, and distal femur as soon as they are medically stable [4], in order to assess bone health and fracture risk. A number of demographic and SCI-related factors, comorbidities and use of certain medications can also be used to determine those at highest risk for fracture [5]. A recent study reported that providers who care for patients with a SCI do utilize these BMD-related and clinical risk factors to identify subgroups of patients at highest risk for these events and to target pharmacological therapies for fracture prevention accordingly [6].
There are substantial differences in the pathophysiology of SCI-related bone loss compared with senile or postmenopausal osteoporosis [7-9]. It is not clear whether pharmacological therapies that are efficacious to treat senile osteoporosis and prevent fractures in the able-bodied population [10-12] are also effective in sublesional osteoporosis. In a cohort of Veterans with a chronic SCI, we recently reported that bisphosphonates, which may reduce fracture risk by upwards of 40% in the elderly able-bodied population [13] were not significantly associated with decreased lower extremity fracture risk [14]. However, in that report [14], we did not examine the relationship of long-term use of bisphosphonates with incident fractures. Sufficient duration of bisphosphonate use may be an important consideration for fracture risk reduction. In fact, among able-bodied postmenopausal women, transiliac bone biopsy data suggest that reductions in cortical porosity, a major determinant of bone strength [15,16], occur only after using alendronate for two years or more [17]. Moreover, studies that examine the efficacy of medications to prevent fracture are generally designed to include at least 36 months of therapy [11,12]. The goal of the current analysis was to examine the association of duration of use of oral bisphosphonates with risk of lower extremity (LE) fractures in male Veterans with a SCI.
Methods
Men with a traumatic SCI were identified from the 2016 Veterans Health Administration (VHA) Allocation Resource Center (ARC) list using ICD-9 codes for SCI and treatment in a VHA SCI bed section or outpatient clinic [18]. ARC is a cumulative list of Veterans who have ever received healthcare from a VHA facility. Creation of a cohort of Veterans with traumatic SCI was described in an earlier publication [14]. VHA pharmacy data files were used to identify filled prescriptions for a Food and Drug Administration (FDA) approved oral bisphosphonates for osteoporosis (alendronate, ibandronate, or risedronate).
A matched nested case control study was performed. Cases were defined as men who were adherent to oral bisphosphonate therapy (defined as at least 80% of prescriptions refilled, calculated as medication possession ratio (MPR)) over a period of two years or more [19]. The date of a case’s first prescription served as his index date. Controls were defined as men who did not receive any FDA approved drug therapy for osteoporosis during the study period. The date of the control’s closest outpatient encounter to the case’s index date (within 365 days), served as his index date.
Covariates
A number of important risk factors including demographics (age at index date, race [white, black, other, or missing]), medication use (prescriptions within 90 days of the index date for anticonvulsants, opioids or corticosteroids), prevalent LE fractures (FY2005- FY2010) [21] and SCI-related characteristics: duration of injury (acute [≤ 2 years], chronic [>2 years] or missing) level of injury (paraplegia, tetraplegia or missing), and completeness of injury (complete, incomplete or missing) [5] and having had a prior DXA [20], were considered in the analyses.
Statistical Analyses
Cases and controls were censored at last date of followup within the study period, or fracture (first incident LE fracture (ICD-9 codes 808, 820-829) or pathological LE fracture (ICD-9 codes 733.10, 733.14-733.16, 733.19), or death. To reduce the effect of medical coding variability, we considered LE fracture ICD-9 codes with the same first three digits occurring within 120 days of each other to be the same fracture, consistent with previous research [21]. Cases were also censored if they were switched to another FDA approved osteoporosis medication. Within each nested cohort, each case was matched up to four controls using incidence density sampling. This allowed for cases and controls to be matched only on person time at risk of fracturing to minimize bias, contrary to our previous study where we matched on both person time at risk and propensity quintiles because we had a larger sample [14].
Conditional logistic regressions were used to examine relevant outcomes, which permitted unbiased estimates and 95% confidence intervals of odds ratios of incident LE fracture. Results were also confirmed via stratified Cox regression analyses and we report Hazard Ratios and 95% confidence intervals of the Hazard Ratios. Regressions were adjusted for covariates demonstrated to be statistically significant different at baseline (see Table 1). These included age at index date, corticosteroid use within 90 days of index date, opioid use within 90 days of index date, extent of SCI, prior fracture, and whether there was a prior DXA done. All statistical analyses were performed using SAS 9.2 and STATA version 14 statistical packages. A p-value of <0.05 was considered statistically significant.
There were some missing data with respect to level (6.98%), extent (13.06%) and duration of SCI (5.97%) in men. To account for this, without losing information, we included another category labeled “missing.”
This study was reviewed and approved by the Institutional Review Boards at the two VA facilities where this research was conducted. A HIPAA waiver and a waiver of informed consent were obtained.
Results
We identified 7,989 male Veterans with a traumatic SCI who had utilized VHA health care between fiscal years (FY) 2010-2015 after applying the exclusions noted above. Of these, 267 had at least one filled prescription for an oral bisphosphonate. Among these, only 157 (58.80%) were adherent with this therapy for at least one year. Among these adherent cases, 65 were adherent with oral bisphosphonate therapy for at least two years (24 months), 42 were adherent for 24-36 months and 28 were adherent for 36 to 48 months. Baseline characteristics of the study population (at two years [24 months]) are shown in Table 1. There was no significant association of long-term use of oral bisphosphonates (defined as use of up to 24 months) and incident LE fracture (HR 0.97 [95% CI 0.25 to 3.75]) (Figure 1). Similarly, there was no significant association of oral bisphosphonate use with incident LE fractures among the 42 individuals who were adherent for 3 years (HR 1.17 [95% CI 0.26 to 5.35]) or among the 28 who were adherent for four or more years (HR 1.02 [95% CI 0.13 to 7.89]).
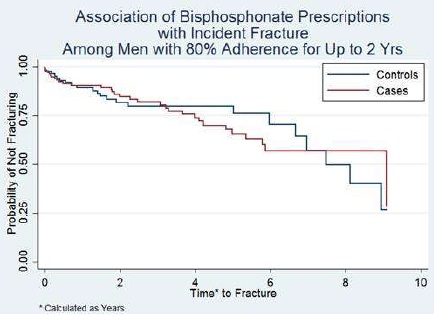
Figure 1: Life-table analysis to incident fracture in cases with oral bisphosphonate prescriptions who were at least 80% adherent to medication up to 2 years vs. controls.
| DEMOGRAPHIC AND CLINICAL CHARACTERISTICS | CASES Oral Bisphosphonate Script+ (n=65) |
CONTROLS No Osteoporosis Scripts+ + (n=7,722) |
P-Value |
| *Age (mean, sd) < 50 > 50 |
57.09 ± 13.79 12 (18.46) 53 (81.54) |
55.14 ± 14.06 2,528 (32.74%) 5,194 (67.26) |
0.02 |
| Race (%) White Black Other | 45 (69.23%) 10 (15.38%) 10 (15.38%) |
5,124 (66.36%) 1,618 (20.95%) 980 (12.69%) |
0.50 |
| Anticonvulsant use (%) | 3 (4.62%) | 257 (3.33%) | 0.48 |
| Corticosteroid use (%) | 4 (6.15%) | 94 (1.22%) | 0.009 |
| Opioid use (%) | 7 (10.77%) | 300 (3.89%) | 0.01 |
| SCI CHARACTERISTICS | |||
| Level of injury (%) Paraplegia Tetraplegia Missing |
29 (44.62%) 31 (47.69%) 5 (7.69%) |
3,434 (44.47%) 3,749 (48.55%) 539 (6.98%) |
0.97 |
| Extent of injury (%) Complete Incomplete Missing |
34 (52.31%) 21 (32.31%) 10 (15.38%) |
2,965 (38.40%) 3,750 (48.56%) 1,007 (13.04%) |
0.03 |
| Prevalent Lower Extremity Fractures (%) | 11 (16.92%) | 4 (0.05) | <0.0001 |
| Duration of SCI Related Injury (%) Acute Chronic Missing |
12 (18.46 %) 49 (75.38%) 4 (6.15%) |
1,375 (17.81 %) 5,886 (76.22%) 461 (5.97%) |
0.99 |
| DXA prior to Index Date (%) | 45 (69.23%) | 875 (11.33%) | <0.0001 |
| + Patient with prescription for alendronate, ibandronate, or risedronate *Age at time of prescription or index date P-Value from Chi-Square or from Fisher’s Exact Test |
|||
Discussion
We did not find a significant association between oral bisphosphonate therapy with incident LE fractures among men with a traumatic SCI, despite adherence with this therapy for at least two years, and in some cases, up to four years. However, few (n=267) in this cohort who had at least one filled prescription for an oral bisphosphonate who were adherent at one year (n=157) continued to be adherent at two years (n=65, 41.4%) and beyond 2 years (n=42, 26.8% at three, and n= 28, 17.8% at four years).
Prevention of the first sentinel fracture event is important in persons with a SCI, not only because of its potential complications [1,2], but also because these fractures are associated with upwards of a fourfold increased risk of additional fractures over time [5]. Reports of the relationship of oral bisphosphonate use with positive changes in BMD, a central predictor of fractures in persons with a SCI [3], are inconsistent [22]. This report extends our prior work, which revealed no overall association of filled prescriptions for bisphosphonates with incident fractures [14], to suggest that, even with long-term adherence to oral bisphosphonate therapies, use of these medications is not associated with risk of LE fractures in men with a traumatic SCI.
We found that there was poor adherence over time with oral bisphosphonate therapy in our cohort of male Veterans with a traumatic SCI. This finding is aligned with prior reports of low adherence to oral bisphosphonate therapies for fracture prevention in the able-bodied population [23]. We have previously reported in a case series, that side effects, predominantly gastrointestinal, were the main reason for discontinuation of oral bisphosphonates in Veterans with a SCI [24].
There are a number of limitations to this work. First, the number of individuals in our cohort who were adherent with oral bisphosphonate use over time was limited. Additionally, administrative databases do not contain information on whether the medications were actually taken and if taken, were used correctly, which is an important consideration, as oral bisphosphonates are very poorly absorbed if taken with food or beverages other than water [25].
In conclusion, in men with a traumatic SCI, long-term adherence with bisphosphonate prescription refills is not associated with reductions in lower extremity fractures. New therapies to prevent fractures in individuals with a SCI are needed.
Acknowledgements
The views expressed in this article are those of the authors and do not necessarily represent the views of the Departments of Defense or Veterans Affairs or the US government. This work was supported by the Department of Defense CMDRP program award number for grant is W81XWH-16-1-0413 and log number is SC150092.
References
2. Carbone LD, Chin AS, Burns SP, Svircev JN, Hoenig H, Heggeness M, et al. Mortality after lower extremity fractures in men with spinal cord injury. Journal of Bone and Mineral Research. 2014 Feb;29(2):432-9.
3. Abderhalden L, Weaver FM, Bethel M, Demirtas H, Burns S, Svircev J, et al. Dual-energy X-ray absorptiometry and fracture prediction in patients with spinal cord injuries and disorders. Osteoporosis international. 2017 Mar 1;28(3):925-34.
4. Morse LR, Biering-Soerensen F, Carbone LD, Cervinka T, Cirnigliaro CM, Johnston TE, et al. Bone Mineral Density Testing in Spinal Cord Injury: 2019 ISCD Official Position. Journal of Clinical Densitometry. 2019 Oct 1;22(4):554-66.
5. Bethel M, Weaver FM, Bailey L, Miskevics S, Svircev JN, Burns SP, et al. Risk factors for osteoporotic fractures in persons with spinal cord injuries and disorders. Osteoporosis International. 2016 Oct 1;27(10):3011-21.
6. Weaver FM, Etingen B, Guihan M, Ray C, Priebe M, Burns S, et al. Spinal cord injury providers’ perspectives on managing sublesional osteoporosis. The Journal of Spinal Cord Medicine. 2019 Dec 24:1-7.
7. Battaglino RA, Lazzari AA, Garshick E, Morse LR. Spinal cord injury-induced osteoporosis: pathogenesis and emerging therapies. Current Osteoporosis Reports. 2012 Dec 1;10(4):278-85.
8. Goltzman D. The Aging Skeleton. Advances in Experimental Medicine and Biology. 2019;1164:153-60.
9. Warriner AH, Patkar NM, Curtis JR, Delzell E, Gary L, Kilgore M, et al. Which fractures are most attributable to osteoporosis? Journal of Clinical Epidemiology. 2011 Jan 1;64(1):46-53.
10. Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, et al. Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA. 1999 Oct 13;282(14):1344-52.
11. Black DM, Cummings SR, Karpf DB, Cauley JA, Thompson DE, Nevitt MC, et al. Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet (London, England). 1996 Dec 7;348(9041):1535-41.
12. Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. The New England Journal of Medicine. 2007 May 3;356(18):1809-22.
13. Chua WM, Nandi N, Masud T. Pharmacological treatments for osteoporosis in very elderly people. Therapeutic Advances in Chronic Disease. 2011 Jul;2(4):279-86.
14. Carbone LD, Gonzalez B, Miskevics S, Ray C, Etingen B, Guihan M, et al. Association of Bisphosphonate Therapy With Incident of Lower Extremity Fractures in Persons With Spinal Cord Injuries or Disorders. Archives of Physical Medicine and Rehabilitation. 2020;101(4):633- 41.
15. McCalden RW, McGeough JA, Barker MB, Court- Brown CM. Age-related changes in the tensile properties of cortical bone. The relative importance of changes in porosity, mineralization, and microstructure. The Journal of Bone and Joint Surgery. 1993 Aug 1;75(8):1193-205.
16. Martin RB. Porosity and specific surface of bone. Critical Reviews in Biomedical Engineering. 1984;10(3):179-222.
17. Roschger P, Rinnerthaler S, Yates J, Rodan GA, Fratzl P, Klaushofer K. Alendronate increases degree and uniformity of mineralization in cancellous bone and decreases the porosity in cortical bone of osteoporotic women. Bone. 2001 Aug 1;29(2):185-91.
18. Smith BM, Evans CT, Ullrich P, Burns S, Guihan M, Miskevics S, et al. Using VA data for research in persons with spinal cord injuries and disorders: lessons from SCI QUERI. Journal of Rehabilitation Research & Development. 2010 Dec 30;47(8):679-88.
19. Curtis JR, Cai Q, Wade SW, Stolshek BS, Adams JL, Balasubramanian A, et al. Osteoporosis medication adherence: physician perceptions vs. patients’ utilization. Bone. 2013 Jul 1;55(1):1-6.
20. Lala D, Craven BC, Thabane L, Papaioannou A, Adachi JD, Popovic MR, et al. Exploring the determinants of fracture risk among individuals with spinal cord injury. Osteoporosis International. 2014 Jan 1;25(1):177-85.
21. Logan WC, Jr., Sloane R, Lyles KW, Goldstein B, Hoenig HM. Incidence of fractures in a cohort of veterans with chronic multiple sclerosis or traumatic spinal cord injury. Archives of Physical Medicine and Rehabilitation. 2008 Feb 1;89(2):237-43.
22. Soleyman-Jahi S, Yousefian A, Maheronnaghsh R, Shokraneh F, Zadegan SA, Soltani A, et al. Evidencebased prevention and treatment of osteoporosis after spinal cord injury: a systematic review. European Spine Journal. 2018 Aug 1;27(8):1798-814.
23. Fatoye F, Smith P, Gebrye T, Yeowell G. Real-world persistence and adherence with oral bisphosphonates for osteoporosis: a systematic review. BMJ open. 2019 Apr 1;9(4):e027049.
24. Le B, Ray C, Gonzalez B, Miskevics S, Weaver FM, Priebe M, et al. Reasons for Initiation and Discontinuation of Pharmacological Therapies of Osteoporosis in Veterans With Spinal Cord Injury and Disorder. Journal of Clinical Densitometry. 2019 Jun 15.
25. Pazianas M, Abrahamsen B, Ferrari S, Russell RG. Eliminating the need for fasting with oral administration of bisphosphonates. Therapeutics and clinical risk management. 2013;9:395-402.
