Abstract
Background: At present, progesterone administration is widely used. There is no agreement on whether estradiol (E2) addition should be supplement to progesterone (P) as luteal phase support (LPS). The present meta-analysis was conducted to clarify whether E2 supplementation as LPS has beneficial effect on the clinical outcome after in vitro fertilization (IVF)/intracytoplasmic sperm injection (ICSI).
Methods: MEDLINE, Google Scholar and the Cochrane Library were searched from the incipiency until May 2019. The primary outcome was clinical pregnancy rate (CPR), ongoing pregnancy rate (ongoing PR), embryo implantation rate (IR), spontaneous abortion rate (AR). Thirteen, seven, four, and ten studies were included while evaluating the effect of E2 supplementation on CPR, ongoing PR, IR, and AR, respectively.
Results: The results showed that E2+P group had significant higher CPR (OR=1.39, P=0.0001) and ongoing PR (OR=1.36, P=0.01) than that of P-only group. No significant differences were found between IR (P=0.47) and AR (P=0.43) between E2+P and P- only groups. Subgroup analysis of the GnRH-a/ant showed that GnRH-a protocol group had a significant higher CPR (OR=1.37, P=0.02) and ongoing PR (OR=1.76, P=0.002) with E2+P, whereas GnRH-ant protocol group had no difference in CPR (P=0.6), ongoing PR (P=0.44), IR (P=0.18), and AR (P=0.87) among E2+P and P- only groups.
Conclusions: The results suggest that E2 supplementation as LPS has beneficial effect on the clinical outcomes only during cycles with GnRH-a protocol.
Keywords
Luteal phase support; Estradiol; IVF; ICSI; Ovarian stimulation
Abbreviations
ART: Assisted Reproductive Technology; IVF-ET: In vitro Fertilization - Embryo Transfer; ICSI: Intracytoplasmic Sperm Injection; LPS: Luteal Phase Support; GnRH-ant: Gonadotrophin-Releasing Hormone antagonist; GnRH-a: Gonadotrophin-Releasing Hormone agonist; E2: Estrogen; P: Progesterone.
Summary
This study aimed to clarify whether estradiol (E2) supplementation as luteal phase support (LPS) has benefit effect on the clinical outcomes after IVF/ICSI. A meta-analysis was conducted. E2+P group had significant higher CPR and ongoing PR than that of P-only group. GnRH-a protocol group had a significant higher CPR and ongoing PR with E2+P, whereas GnRH-ant protocol group had no difference in CPR, ongoing PR, IR, and AR among E2+P and P- only groups. So, E2 supplementation as LPS has beneficial effect on the clinical outcomes only during cycles with GnRH-a protocol.
Background
Controlled ovarian hyper-stimulation during in vitro fertilization/ intracytoplasmic sperm injection (IVF/ ICSI) always results in a defective luteal phase because of the pituitary down-regulation with Gonadotropinreleasing hormone agonist (GnRH-a) and the loss of granular cell after ovarian retrieval [1]. So, it is necessary to provide luteal phase support (LPS) in IVF/ICSI cycles, especially in cycles with long GnRH-a protocol.
In a physiological condition, the corpus luteum secretes not only progesterone (P) but also estradiol (E2). At present, P administration is widely used. There is no agreement on whether E2 addition should be supplement to P as LPS. The level of E2 falls sharply after ovulation and slowly rises again during the luteal phase. The role of E2 in the process of embryo implantation is unclear.
Some studies have indicated that the decrease of E2 levels in mid-luteal or late luteal might have negative effect on the clinical pregnancy [2]. The difference of E2 level between pregnant and non-pregnant women starts to appear on the 9th day to 10th day after the luteinizing hormone (LH) peak [3]. Therefore, endometrial maturation with good receptivity also needs adequate concentrations of E2 level during the widow of implantation (WOI) [4]. Additionally, some studies showed that E2 supplementation for LPS would improve the clinical pregnancy outcome [5]. However, some studies did not find beneficial effect of E2 supplementation on reproductive outcomes in women undergoing IVF/ICSI [6].
So far, there were five systematic reviews with different results. Four meta-analyses confirmed that the E2 supplement for LPS in IVF/ICSI cycles has no beneficial effect on pregnancy rate (PR) [8-10,12]. One meta-analysis study [11] addressed this issue, and showed improved clinical pregnancy rate (IR) with E2 supplementation to standard P LPS.
Therefore, it was necessary to evaluate weather supplementation of E2 have beneficial effect on outcome of IVF/ICSI cycles. The present systematic review and meta-analysis aimed to clarify the effect of luteal E2 supplementation.
Methods
Literature retrieval
MEDLINE, Google Scholar and the Cochrane Library were searched from the incipiency until May 2018. The keywords used to search relative studies were as follows: one including terms on luteal phase support/ luteal support, one including terms on estrogen/oestrogen/ oestradiol/estradiol supplementation, one including clinical outcome of IVF/ICSI (clinical pregnancy rate, ongoing pregnancy rate, embryo implantation rate, and abortion rate), and the last one about assisted reproductive technologies (ART, IVF, ICSI, assisted reproduction, in vitro fertilization, intracytoplasmic sperm injection). A number of literatures were generated via combining the subsets with “AND”. Papers published in non-English were excluded. The eligibility of included papers was reviewed by two reviewers independently and the third reviewer was asked to review the paper when the discrepancy was happened.
Study selection and data extraction
Studies which evaluated estrogen supplementation for LPS in women undergoing IVF/ICSI were included and analyzed. The main outcome index was the clinical pregnancy rate (CPR), ongoing pregnancy rate (ongoing PR), embryos implantation rate (IR) and/or abortion rate (AR).
For studies to be eligible, outcome data (with CPR, ongoing PR, IR, and/or AR with/ without estradiol supplementation) were extracted in 2×2 tables. We also recorded the first author, publication year, and study design, sample size, ovarian stimulation protocol, the dose and route of P and E2, and all the reproductive outcomes. Newcastle - Ottawa Quality Assessment Scales was used to evaluate the observational researches’ quality. Two reviewers completed the quality assessment, and any discrepancy was settled by a third reviewer.
Data analysis
The Odds Ratio (OR) with the 2x2 tables was used to conduct a standard meta-analysis. Forest plots were used to evaluate the heterogeneity of the included studies graphically and the l2 value was used to evaluate the heterogeneity quantificationally among studies. An overall OR and its 95% CI were calculated via fixed or random effect model. Because of the low power of X2 test for heterogeneity of a meta-analysis, the statistical significant was considered when the P value <0.1 rather than <0.05, especially in the studies with small sample size. RevMan 5.0 (Cochrane Collaboration, Oxford, UK) was implied to perform the statistical analyses.
Results
Studies selection and characteristics
The search strategy yielded 381 citations. 305 irrelevant studies were excluded after reviewing the title and/ or abstract. Of the 76 remaining publications, 49 were excluded as clinical outcome of IVF/ICSI were not reported or defined. Four studies were system review and meta-analysis. An additional eight papers were excluded because we could not get a 2x2 table from the result (Supplementary Table 1).
The characteristics of included studies are described in Table 1. Fifteen prospective studies were included in our meta-analysis, and implied long protocol with GnRH-a, GnRH-ant, or both undergoing IVF/ICSI. In these studies, there were thirteen studies for clinical pregnancy, seven studies for ongoing pregnancy, four studies for embryo implantation, and ten studies for abortion. A total of 2971 cycles were included in the analyses, comprising 1572 couples received E2 supplementation and 1399 couples received P4 only as LPS in IVF/ICSI cycles. The sample size varied from 44 to 402 cycles. The dose of E2 and P, together with the route of administration and the duration of treatment, is presented. E2 was administered orally in nine studies, transdermally in three studies; vaginally, transdermal/orally, and vaginal/orally in only one study, respectively. P was given vaginally in most studies, while it was administrated by IM injection in four trials, by vaginal/oral, transdermal/oral in only one study, and IM routes in four studies. The duration and doses of E2 and/ or P4 varied between studies.
Meta-analysis
Thirteen, seven, four, and ten studies were obtained in our meta-analysis to assess the effect of E2 supplementation on CPR, ongoing PR, IR, and AR after IVF/ICSI treatment, respectively.
When evaluated the effects of E2 addition on the CPR, thirteen studies were analyzed including 1401 cycles with E2 supplementation and 1228 cycles with P only as LPS. The CPR was significantly increased in women with E2 supplementation as LPS compared with those with P only as LPS. The statistical heterogeneity was moderate with P<0.1 (l2=74%). The random effects model was applied with a combined OR 1.39 (95% CI, 1.18, 1.64; P=0.0001) (Figure 1).
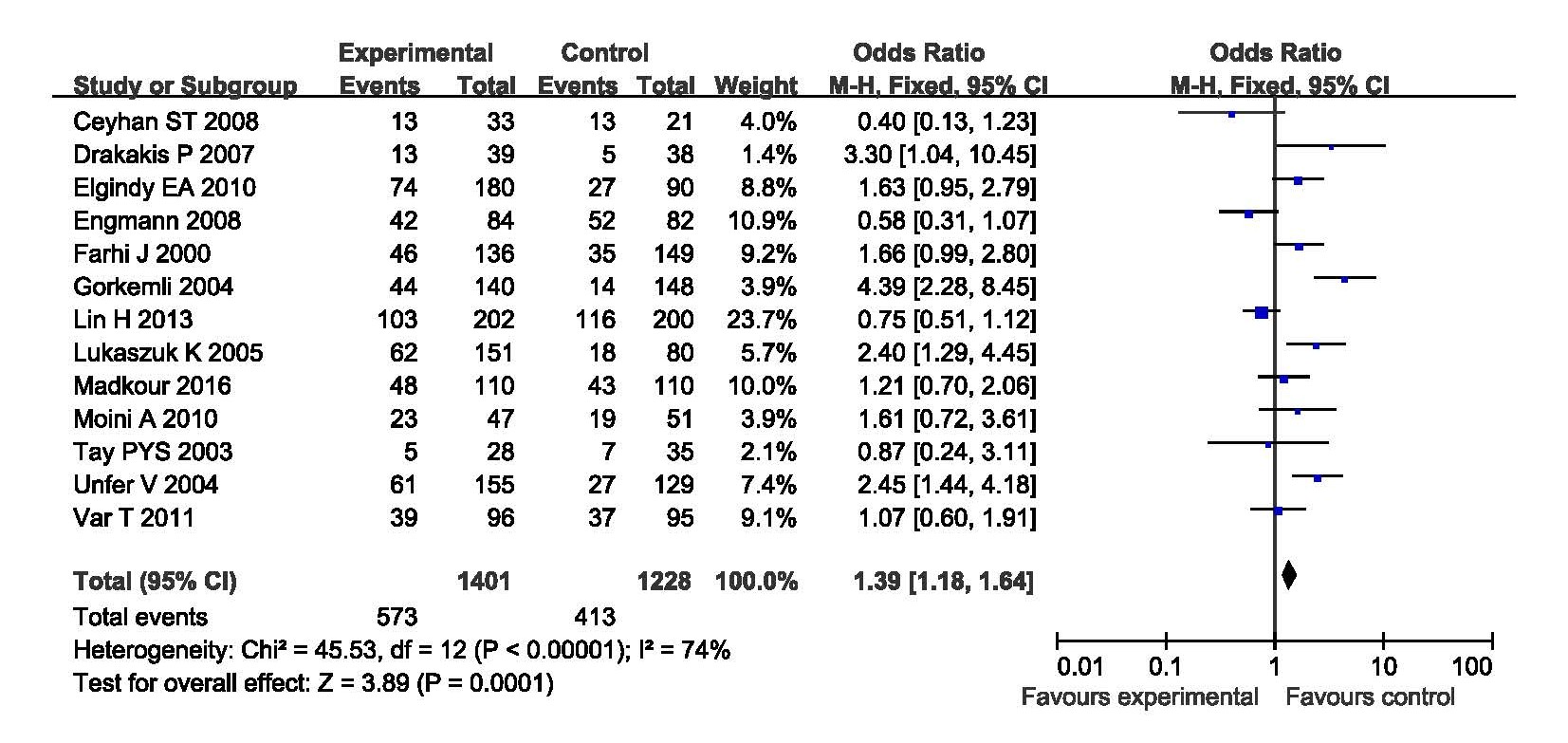
Figure 1. Forest plot showing the results of meta-analysis of studies comparing the effect of E2 + P and P only as LPS on clinical pregnancy after IVF/ICIS.
Seven studies were included to evaluate the effects of E2 addition on the ongoing PR, including 690 cycles with E2 supplementation and 669 cycles with P only as LPS. The PR was significantly increased with E2 supplementation as LPS compared with those with P only as LPS. The statistical heterogeneity was moderate with P<0.1 (l2=58%). The combined OR was 1.36 (95% CI, 1.07, 1.72; P=0.01) (Figure 2).
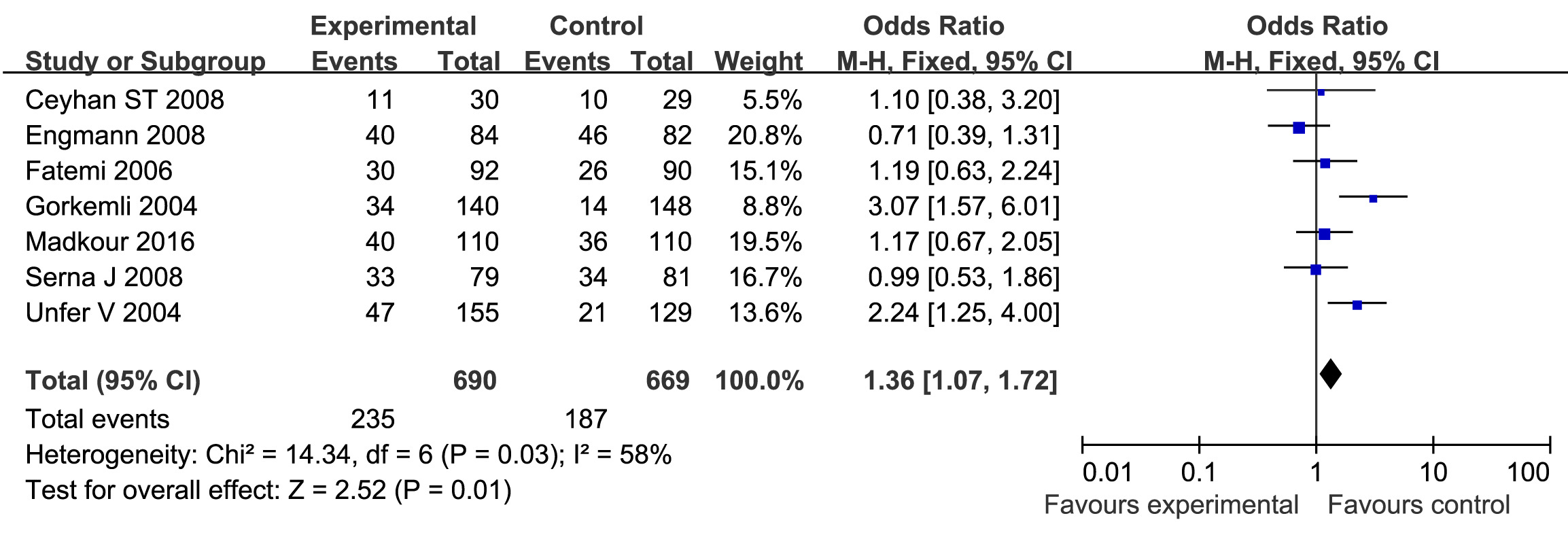
Figure 2. Forest plot showing the results of meta-analysis of studies comparing the effect of E2 + P and P only as LPS on ongoing pregnancy after IVF/ICSI.
Of fifteen studies, ten studies evaluated the effect of E2 supplementation on IR and AR, respectively. These results of the meta-analysis indicated that there was no beneficial effect in patients with E2 supplementation as LPS. The Q statistic P-value for ongoing PR and AR were all below 0.1, indicating heterogeneity of the studies (l2=54%, P=0.09; l2=40%, P=0.09). The random effects model was implied and these combined ORs for IR, AR were 1.08 (95% CI, 0.88, 1.31; P=0.47) and 0.86 (95% CI, 0.58, 1.26; P=0.43), respectively (Supplementary Figures 1 and 2).
We also evaluated the effect of E2 supplementation on the clinical outcome (CPR, ongoing PR, IR, AR) after GnRH-a protocol or GnRH-ant protocol treatment. Interestingly, the results showed that GnRH-a protocol subgroup got an increased CPR (OR 1.37; 95% CI, 1.05, 1.78; P=0.02) and ongoing PR (OR 1.76; 95% CI, 1.22, 2.53; P=0.0002) when E2 supplementation as LPS, whereas GnRH-ant protocol subgroup did not find such effect. Additionally, there were no differences in IR and AR between E2+P and P- only groups when GnRH-a protocol and GnRHant protocol subgroups were analyzed separately (Supplementary Figures 3-6).
These studies included were all have high score via the Newcastle-Ottawa Quality Assessment Scale (not shown). There was no publication bias as a symmetrical shape was seen in the funnel plot in the evaluation the effect of E2 supplementation on CPR, ongoing PR (Supplementary Figures 7 and 8). However, the studies showed modest publication bias when assessing the effect of E2 supplementation on IR and AR (Supplementary Figures 9 and 10).
Discussion
So far, five systematic reviews [8-12] have evaluated whether there was positive effect of E2 supplementation as LPS on the IVF/ICSI cycles and came into different conclusions. As far as we know, this study has the largest sample size of 2971 IVF/ICSI cycles. Besides, subgroup analysis was conducted to assess whether the effect of E2 supplementation between the GnRH-a and GnRH-ant protocol is different.
In the present systematic review, 13, 7, 4, and 10 studies were included assessing the effect of E2 supplementation as LPS on the clinical pregnancy, ongoing pregnancy, embryo implantation and abortion after IVF/ICSI, respectively. Our results indicated that E2 supplementation could improve the clinical pregnancy significantly (combined OR 1.39; 95% CI 1.18, 1.64; P=0.0001) and ongoing pregnancy (combined OR 1.36; 95% CI 1.07, 1.72; P=0.01); however, we did not find significant differences in IR, and AR between E2 + P group and P only group. The conclusion was not completely in agreement with the systematic reviews by Jee et al. [10], Kolibianakis et al. [9] and Gelbaya et al. [8], Huang et al.[12], which suggested that the addition of E2 for LPS have no beneficial effect on outcome of IVF/ICSI. In addition, we found that E2 supplementation for LPS has no beneficial effect on the clinical outcomes in cycles with GnRH-ant protocol.
As we all know, the luteal function is impaired during IVF/ICSI cycles. Additionally, very low level of LH can be seen [13], which is necessary to maintain the function of the corpus luteum in humans [14]. It is well established that corpus luteum secretes both progesterone and E2. Therefore, not only P but also E2 as LPS would be necessary for the development of endometrium before embryo implantation. Whether imply E2 supplementation as LPS during IVF/ICSI cycles is still controversial [15,16].
Stewart et al. [17] identified that there was a significantly different E2 concentration between pregnant and nonpregnant women using donor insemination for the first time. Similarly, there was a rise of luteal E2 level on day 6 in pregnant women compared with non-pregnant women trying to conceive spontaneously. Sharara and McClamrock [18] found that when the ratio of peak E2 to mid-luteal E2 is greater than 5, the embryo implantation rate is reduced. One study found that women received 6 mg E2 have a better clinical pregnancy outcome in comparison to women without E2 supplementation. The positive effect of E2 supplementation for LPS in ICSI cycles with GnRH-a long protocol was confirmed in recent two studies [19]. A researcher reported that E2 addition to progesterone can reduce abortion rates, but this finding was not significant [20]. Anyway, the decline of late luteal E2 level may have negative affect on the development of endometrial before embryo implantation [21]. Increased level of L-selectin ligands-adhesion molecule can be seen in endometrium of the women with the E2 supplementation as LPS during the phase of luteal [22].
However, other studies indicated that there was no association between the decline of mid-luteal E2 and the clinical pregnancy outcome [23]. Muasher and colleagues [24] have also found that E2 levels were similar between pregnant and non-pregnant women up to 10 days after oocyte retrieval. One recent study showed that pregnancy did not associated with the serum level of E2 in mid-luteal phase or with percent E2 decline [25]. The extent of E2 fall during the phase of luteal of ART cycles seem have no negative effect on the clinical pregnancy outcome [26], successful pregnancy in a few cases in which E2 supplementation was unintentionally omitted in donor oocytes cycles raised question on the necessity of E2 addition as LPS [27]. Subsequent studies have shown that luteal E2 is not essential in the endometrial development for embryo imbed [3,16]. In a more recent study, using GnRH antagonist protocol, the endocrine profile (FSH, E2, and P) was similar in group receiving P alone vs. P+ E2 during the luteal phase [17]. During a GnRH-a long protocol, women with 2, 4 or 6 mg E2 supplementation as LPS have similar clinical pregnancy rates, and women with 2 mg E2 have a higher miscarriage rate compared with other groups [28]. These results indicated that E2 supplementation as PLS have no positive effect on the clinical pregnancy outcome, and were in accordance with the findings of previous researches.
In GnRH antagonist cycles, the women were divided into 3 groups according to the LPS protocol: no luteal support, vaginal micronized P, and micronized P+ oral E2, there was no significant differences in estrogen receptor expression on the day of oocyte retrieval, 3, 5, and 10 days after retrieval [29]. In this study, women with P alone has a significantly increased PR expression in the luminal endometrium 3 days after retrieval compared with the other groups. In earlier study, there was no difference between E2 and P receptors as well as endometrial histology in the luteal phase of ovariectomized women with or without E2 [30]. Similarly, in a programmed cycle, biopsies of the endometrium on the 21st day and the 25th day found that there is no adverse effect on endometrial integrity when neglecting luteal E2 supplementation, indicating that a super physiological level of P may make up for the lack of E2 [16,30].
GnRH-a protocol is always associated with impaired luteal function, with decreased production of E2 and P, and shortened duration of luteal phase, impaired secretion of endogenous gonadotropin. So E2 supplementation as LPS would bring beneficial effect on the clinical pregnancy and ongoing pregnancy. Whereas, GnRH-ant cause less pituitary suppression compared with GnRH-a protocol. P4 may be enough for the LPS, and the E2 supplementation seems to have no beneficial effect on the clinical outcomes.
Comparatively speaking, the meta-analysis and systematic review has the merit of the improved accuracy of the OR estimates by comparison to a single study. The present meta-analysis indicated that E2 supplementation to P as luteal phase support has beneficial effect on clinical pregnancy and ongoing pregnancy after IVF/ICSI, especially in cycles with GnRH-a protocol. Evaluating the effect on clinical pregnancy and ongoing pregnancy, there were four and two studies in which the OR was below one, and the combined OR was 1.39 (95% CI 1.18, 1.64) and 1.36 (95% CI 1.07, 1.72). While assessing the impact on implantation and abortion, the ORs of the included studies were greater than unity, although the sample sizes of studies were small.
But then, the main weakness of this study is the higher variable between studies: different dose and duration of E2 administration, different treatment types (IVF/ ICSI) and different ovarian stimulation protocols. The inclusion/exclusion criteria for women and the definition of pregnancy loss are not always clearly stated or varied. Besides, the present meta-analysis only included papers published in English, we would miss some relative papers published in non-English.
Conclusion
Despite the weakness, the present meta-analysis demonstrates that there is an advantage only during cycles with GnRH agonist protocol. However, there was no beneficial effect on IR and AR. The benefits of additional E2 supplementation, the optimal E2 dose, the start and the end times of E2 supplementation should be further clarified.
Competing Interest
The authors declare that they have no conflict of interest.
Funding
This project was supported by the National Natural Science Foundation of China (Grant No. 81401269), and the class General Financial Grant from the China Postdoctoral Science Foundation (Grant No. 2017M620360).
Authors’ Contribution
Jing Zhao and Jie Hao participated in the design of the study and the acquisition of data, performed the statistical analysis, drafted the article and revised it critically. Yanping Li contributed to conception and design, acquisition of data and analysis and interpretation of data. Bin Xu and Yonggang Wang participated in the interpretation of the data and the revision of the article. All authors read and approved the final manuscript.
References
2. Sharara FI, McClamrock HD. Ratio of oestradiol concentration on the day of human chorionic gonadotrophin administration to mid-luteal oestradiol concentration is predictive of in-vitro fertilization outcome. Human Reproduction. 1999 Nov 1;14(11):2777-82.
3. Zegers-Hochschild F, Altieri E. Luteal estrogen is not required for the establishment of pregnancy in the human. Journal of Assisted Reproduction and Genetics. 1995 Mar 1;12(3):224-8.
4. Engmann L, DiLuigi A, Schmidt D, Benadiva C, Maier D, Nulsen J. The effect of luteal phase vaginal estradiol supplementation on the success of in vitro fertilization treatment: a prospective randomized study. Fertility and sterility. 2008 Mar 1;89(3):554-61.
5. Hill MJ, McWilliams GD, Miller KA, Scott Jr RT, Frattarelli JL. A luteal estradiol protocol for anticipated poor-responder patients may improve delivery rates. Fertility and sterility. 2009 Mar 1;91(3):739-43.
6. Karlikaya G, Karagozoglu H, Kumbak B, Lacin S, Guney A, Kahraman S. O-263: Adding estradiol to progesteron for luteal phase support does not improve the clinical outcome in assisted reproductive technologies (ART) cycles. Fertility and Sterility. 2006 Sep 1;86(3):S112.
7. Pritts EA, Atwood AK. Luteal phase support in infertility treatment: a meta-analysis of the randomized trials. Human Reproduction. 2002 Sep 1;17(9):2287-99.
8. Gelbaya TA, Kyrgiou M, Tsoumpou I, Nardo LG. The use of estradiol for luteal phase support in in vitro fertilization/intracytoplasmic sperm injection cycles: a systematic review and meta-analysis. Fertility and Sterility. 2008 Dec 1;90(6):2116-25.
9. Kolibianakis EM, Venetis CA, Papanikolaou EG, Diedrich K, Tarlatzis BC, Griesinger G. Estrogen addition to progesterone for luteal phase support in cycles stimulated with GnRH analogues and gonadotrophins for IVF: a systematic review and meta-analysis. Human Reproduction. 2008 Jun 1;23(6):1346-54.
10. Jee BC, Suh CS, Kim SH, Kim YB, Moon SY. Effects of estradiol supplementation during the luteal phase of in vitro fertilization cycles: a meta-analysis. Fertility and Sterility. 2010 Jan 15;93(2):428-36.
11. Zhang XM, Lv F, Wang P, Huang XM, Liu KF, Pan Y, et al. Estrogen supplementation to progesterone as luteal phase support in patients undergoing in vitro fertilization: systematic review and meta-analysis. Medicine. 2015 Feb;94(8).
12. Huang N, Situ B, Chen X, Liu J, Yan P, Kang X, Kong S, Huang M. Meta-analysis of estradiol for luteal phase support in in vitro fertilization/intracytoplasmic sperm injection. Fertility and Sterility. 2015 Feb 1;103(2):367-73.
13. Tavaniotou A, Devroey P. Effect of human chorionic gonadotropin on luteal luteinizing hormone concentrations in natural cycles. Fertility and Sterility. 2003 Sep 1;80(3):654-5.
14. Casper RF, Yen S. Induction of luteolysis in the human with a long-acting analog of luteinizing hormone-releasing factor. Science. 1979 Jul 27;205(4404):408-10.
15. Ismail Madkour WA, Noah B, Abdel Hamid AM, Zaheer H, Al-Bahr A, Shaeer M, et al. Luteal phase support with estradiol and progesterone versus progesterone alone in GnRH antagonist ICSI cycles: a randomized controlled study. Human Fertility. 2016 Apr 2;19(2):142-9.
16. Younis JS, Ezra Y, Sherman Y, Simon A, Schenker JG, Laufer N. The effect of estradiol depletion during the luteal phase on endometrial development. Fertility and sterility. 1994 Jul 1;62(1):103-7.
17. Stewart DR, Overstreet JW, Nakajima ST, Lasley BL. Enhanced ovarian steroid secretion before implantation in early human pregnancy. The Journal of Clinical Endocrinology & Metabolism. 1993 Jun 1;76(6):1470-6.
18. Sharara FI, McClamrock HD. Ratio of oestradiol concentration on the day of human chorionic gonadotrophin administration to mid-luteal oestradiol concentration is predictive of in-vitro fertilization outcome. Human Reproduction. 1999 Nov 1;14(11):2777-82.
19. Elgindy EA, El-Haieg DO, Mostafa MI, Shafiek M. Does luteal estradiol supplementation have a role in long agonist cycles?. Fertility and Sterility. 2010 May 1;93(7):2182-8.
20. Chen S, Li H, Kong L, Zhu L, Zhang X, Xing F. Estrogen plus progesterone supplementation for luteal phase support in in vitro fertilization and embryo transfer. Fertility and Sterility. 2004 Sep 1;82:S191-2.
21. Smitz J, Devroey P, Camus M, Deschacht J, Khan I, Staessen C, et al. The luteal phase and early pregnancy after combined GnRH-agonist/HMG treatment for superovulation in IVF or GIFT. Human Reproduction. 1988 Jul 1;3(5):585-90.
22. Vlahos NF, Lipari CW, Bankowski B, Lai TH, King JA, Shih IM, Fragakis K, Zhao Y. Effect of luteal-phase support on endometrial L-selectin ligand expression after recombinant follicle-stimulating hormone and ganirelix acetate for in vitro fertilization. The Journal of Clinical Endocrinology & Metabolism. 2006 Oct 1;91(10):4043-9.
23. Narvekar SA, Gupta N, Shetty N, Kottur A, Srinivas MS, Rao KA. The degree of serum estradiol decline in early and midluteal phase had no adverse effect on IVF/ICSI outcome. Journal of human reproductive sciences. 2010 Jan;3(1):25.
24. Muasher S, Acosta AA, Garcia JE, Jones GS, Jones HW. Luteal phase serum estradiol and progesterone in in vitro fertilization. Fertility and Sterility. 1984 Jun 1;41(6):838-43.
25. Friedler S, Zimerman A, Schachter M, Raziel A, Strassburger D, Ron El R. The midluteal decline in serum estradiol levels is drastic but not deleterious for implantation after in vitro fertilization and embryo transfer in patients with normal or high responses. Fertility and Sterility. 2005; 83: 54-60.
26. Nagahara Y, Miyake A, Tasaka K, Kawamura Y, Aono T, Tanizawa O. Possible site of negative and positive feedback action of oestrogen on gonadotrophin secretion in normal women. European Journal of Endocrinology. 1985 Apr 1;108(4):440-4.
27. Stassart JP, Corfman RS, Ball GD. CASE REPORT: Continuation of a donor oocyte pregnancy in a functionally agonadal patient without early oestrogen support. Human Reproduction. 1995 Nov 1;10(11):3061-3.
28. Tonguc E, Var T, Ozyer S, Citil A, Dogan M. Estradiol supplementation during the luteal phase of in vitro fertilization cycles: a prospective randomised study. European Journal of Obstetrics & Gynecology and Reproductive Biology. 2011 Feb 1;154(2):172-6.
29. Hubayter ZR, Vlahos NF, Lai TH, Garcia JA, Zhao Y. The impact of luteal phase support on endometrial estrogen and progesterone receptor expression: a prospective randomized study. Fertility and Sterility. 2008 Sep 1;90:S65.
30. De Ziegler D, Bergeron C, Cornel C, Medalie DA, Massai MR, Milgrom E, et al. Effects of luteal estradiol on the secretory transformation of human endometrium and plasma gonadotropins. The Journal of Clinical Endocrinology & Metabolism. 1992 Feb 1;74(2):322-31.
