Abstract
The nuclear envelope (NE) is the hallmark of eukaryotic cells, serving as a critical physical barrier that compartmentalizes the genome and separates nuclear and cytoplasmic processes to tightly regulate nucleocytoplasmic transport. Biomolecular phase separation, a physicochemical process frequently driven by proteins containing intrinsically disordered regions (IDRs), is a fundamental mechanism governing diverse cellular activities. Yet, its roles at the nuclear envelope remain largely unexplored. In our recent study (Tang et al., Developmental Cell, 2025), we demonstrated how phase separation at the nuclear lamina (NL) orchestrates transcriptional responses to environmental stress in plants. Building on this finding, this commentary synthesizes the emerging insights into the contributions of phase separation across the entire NE, including the NL, nuclear pore complex (NPC) and nuclear membranes, in both plant and animal systems. Collectively, recent advances establish that perinuclear phase separation as a pivotal regulator of genome activity and nucleocytoplasmic transport. This commentary concludes by highlighting the imperative to further investigation into perinuclear phase separation as an essential and dynamic signaling hub that integrates cytoplasmic signals with nuclear responses to various cellular and environmental cues.
Keywords
Nuclear envelope, Nuclear membrane, Nuclear lamina, Nuclear pore complex, Phase separation, IDRs, Condensates
Introduction
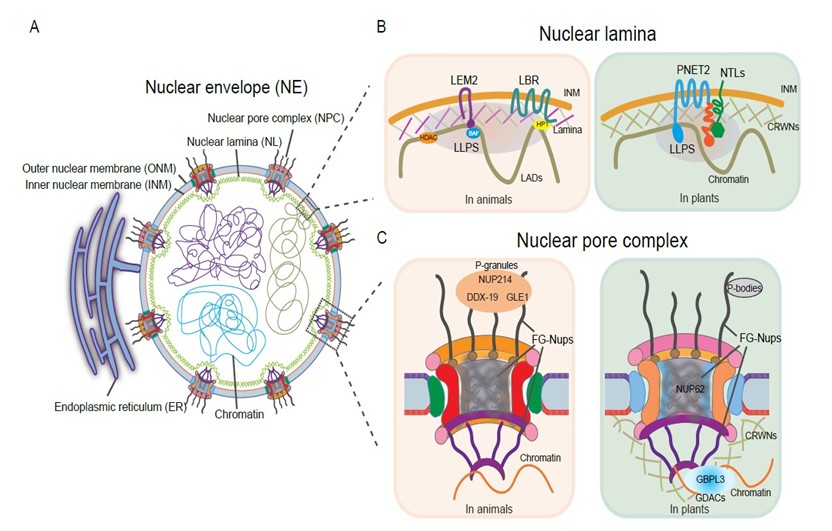
The nuclear envelope is a conserved eukaryotic structure composed of two phospholipid bilayers, the outer and inner nuclear membrane (ONM/INM) (Figure 1A). Underlying the INM, the NL composed of lamins, lamin-like proteins, and their associated INM proteins, forms a peripheral structural meshwork [1,2]. Perforating the NE, the NPCs are structurally partitioned into the cytoplasmic fibrils, the central channel, and the nuclear basket. Nucleoporins (Nups) as the building blocks of the NPC and approximately one third of them contain FG (phenylalanine-glycine)-rich repeats (FG-Nups) that establish the selective permeability barrier for the regulated nucleocytoplasmic transport [3,4]. Although the NE architecture is conserved, its molecular composition diverges among animals, yeast, and plants, suggesting distinct regulatory mechanisms [5]. In humans, NE gene mutations cause a spectrum of severe disorders termed envelopathies [6]. In plant, emerging evidence underscores increasingly critical NE roles [7,8].
Proteins enriched in intrinsically disordered regions (IDRs) exhibit high solvent accessibility and propensity for liquid-liquid phase separation (LLPS) [9]. LLPS forms membrane-less compartments that locally concentrates macromolecules for rapid spatiotemporal organization of biochemical reactions [10]. Notably, membrane associated proteins can also undergo LLPS to participate in membrane remodeling and signaling transduction [11,12]. Consequently, these biomolecular condensates are now widely recognized as key players in various cellular processes. Dysregulated LLPS and condensate dynamics link to a plethora of human diseases, including cancer, neurodegeneration, ageing, and infections. For instance, atypical NE-associated condensates implicate nucleoporin-directed chaperone activities in neurological disorders [13]. Importantly, LLPS also orchestrates plant responses to both developmental cues and environmental stimuli [14,15].
Nuclear Lamina Forms a Distinct Compartment to Regulate Genome Organization and Gene Expression
Nuclear lamina plays a fundamental role in tethering and organizing chromatin, contributing to the characteristic enrichment of silent heterochromatin, also known as lamina-associated domains (LADs) at the nuclear periphery. Advances in high-resolution imaging and mesoscopic modeling reveal that nuclear chromatin itself can organize into active, multiphasic liquid condensates [16,17]. This chromatin-driven microphase separation modulates chromatin dynamics and its interactions with the lamina, which in turn influence nuclear morphology and shape [18]. Additionally, heterochromatin-bound protein 1 alpha (HP1α) is targeted to the nuclear lamina (NL) via the lamin B receptor (LBR), where phosphorylation promotes its assembly into phase-separated droplets [19]. By sequestering compacted chromatin within these HP1α condensates, this phase separation may modulate HP1α's ability to bind H3K9me3-marked chromatin. The NL localizes this phase-separated heterochromatin to the periphery, underscoring that phase separation at the NL can influence global genomic organization. Ultimately, such phase-separation events at the lamina-chromatin interface constitute a critical regulatory layer for gene expression [20], underscoring a dynamic and reciprocal interplay between chromatin organization and nuclear lamina. Of note, the epigenetic landscape of NL is tightly linked to cancer progression. In cancer and aging cells, hypomethylated DNA regions are enriched within LADs, revealing an epigenetic connection between the NL and genetic disorders [21]. Lamin A mutants E161K and K97E, associated with dilated cardiomyopathy, drive aberrant nuclear lamin A aggregation and disrupt the peripheral lamina networks, altering chromatin organization and the epigenetic landscape [22]. Similarly, progerin as the lamin A isoform causing Hutchinson–Gilford progeria syndrome (HGPS), which disrupts heterochromatin–lamina interactions and downregulates H3K9me3 and H3K27me3 [23]. Histone deacetylase HDAC1, which interacts with lamin A/C, is reduced in HGPS fibroblasts, also contributing to transcriptional alterations. A functional link between the NL and DNA methylation may be mediated by LBR interaction with MeCP2, a methyl-CpG binding protein mutated in Rett syndrome [24]. MeCP2 loss induces progeria-like nuclear defects, including reduced lamin and LBR levels and aberrant heterochromatin foci organization. An understanding of phase separation at the NE can provide insights into drug accessibility and target engagement, particularly concerning the efficacy of epigenetic therapies. Given that many NE-associated diseases involve abnormal LLPS, directly targeting these aberrant condensates represents a promising therapeutic strategy [25]. For instance, epigenetic drugs such as the DNA methyltransferase inhibitor decitabine and the histone deacetylase inhibitor vorinostat could potentially be directed to aberrant chromatin condensates.
Plants lack lamin proteins found in animals, instead evolving functional counterparts in plants such as the nucleoskeletal CRWN proteins and the INM protein PNET2 [5,26]. In Arabidopsis, PNET2 maintains proper genome organization and activity. Its loss leads to drastic transcriptome alterations and disrupted global chromatin architecture, restricting growth and activating diverse stress responses [27]. Beyond its middle six transmembrane helices, PNET2 contains a C-terminal IDR that drives LLPS (Figure 1B) [27,28]. PNET2 physically interacts with membrane-tethered NAC transcription factors (NTLs). The IDRs present in both PNET2 and the NTLs promote co-phase separation within the NL, sequestering NTLs to limit their transcriptional activity. Genetically, NTLs function downstream of PNET2 to regulate stress-responsive gene expression, as NTL knockdown in pnet2 mutants largely suppresses the stress-sensitive phenotypes. Environmental stimuli such as heat shock can dissolve the PNET2–NTL condensates, releasing NTLs to activate target genes [28].
Consistent with this model, plant CRWN proteins, which have long coiled-coil structures and IDRs, also undergo LLPS to recruit DNA damage repair regulators, thereby participating in the DNA damage response [29]. Furthermore, the N-terminal domain of PNET2 physically interacts with CRWNs, and their transient co-expression forms nuclear condensates [27], suggesting that LLPS may regulate their functional interplay, a hypothesis requiring further investigation.
The NPC Central Channel is a Highly Dynamic Condensate Structure for Macromolecular Trafficking
The examples above illustrate how LLPS at NL enables dynamic regulation of chromatin organization and gene expression. Meanwhile, the NL and chromatin are also physically and functionally coupled to the NPC. By regulating the activity and localization of key epigenetic modifiers during osteoclast differentiation, NPCs shape the epigenetic landscape and chromatin reorganization, revealing new potential therapeutic targets for bone diseases [30]. The NPC is a highly condensate structure spanning the nuclear membrane. FG-Nups frequently overlap with IDRs, exhibiting the structural characteristics of natively unfolded proteins and forming flexible filaments that line the NPC central channel [31]. The FG repeats drive phase separation into elastic and reversible hydrogels, constituting a selective permeability barrier. This unique biophysical property enables the rapid yet selective nucleocytoplasmic transport [32]. The principles governing FG-NUP phase separation involve multivalent interactions between phenylalanine-glycine motifs, which create a dense, sieve-like meshwork that excludes large cargo while permitting passage of those bound to specific nuclear transport receptors (NTRs) [33]. Unlike cytoplasmic droplets that freely diffuse, NPC condensates are tethered to the nuclear membrane and confined within a cylindrical channel. This spatial constraint of condensed NPC channel imposes distinct biophysical properties: the hydrogel must maintain mechanical stability while allowing rapid sol-gel transitions during transport. As a phase-separated permeability barrier, the NPC governs nuclear drug entry through size, hydrophobicity, and FG-repeat interactions, thereby providing a rational basis for engineering improved nuclear accumulation in future clinical contexts.
Like animal counterparts, plant genomes encode a high proportion of Nups containing IDRs. Beyond transport, the NPC functions as a critical hub for gene regulation. Proximity-labelling proteomics shows plant NPC proximal proteome is enriched in IDR-containing proteins, including chromatin remodelers, transcriptional regulators, and mRNA processing components on the nucleoplasmic side, as well as translation machinery on the cytoplasmic side, indicating an intimate coupling between the NPC and RNA metabolism [34]. Concurrently, LLPS at the NPC also facilitates selective nuclear transport essential for plant defense against pathogens and pests [35,36]. For example, the NUP62 subcomplex undergoes phase separation and forms hydrogels critical for immune regulator transport and disease resistance (Figure 1C) [35]. Conversely, the pathogen effector BcSSP2 disrupts NUP62 LLPS in host cells, thereby suppressing defense and promoting infection [36]. Given component conservation, this physical interaction and the resulting perturbation of NPC LLPS likely represent an evolutionarily conserved strategy by which necrotrophic pathogens to facilitate infection.
Condensate Extensions Surrounding the Nuclear Membrane
The nuclear basket (NB) connects the NL and chromatin at the nuclear periphery [37,38]. In plants, the guanylate-binding protein-like GTPase 3 (GBPL3) is a plant-specific NB component that undergoes LLPS upon immune activation to form nuclear GBPL3 defense-activated condensates (GDACs), where GBPL3 directly binds defense-gene promoters and recruits RNA polymerase II machinery to reprogram host gene expression for disease resistance (Figure 1C) [39]. Functionally, GBPL3 interacts with both the NB components NUP136/NUP82 and the nucleoskeleton CRWN proteins, coordinating gene expression during both plant development and stress responses, and maintains nuclear envelope integrity [37,40]. Adding to this complexity, NUP136/NUP82 shares three conserved motifs with the plant nucleoskeleton KAKU4 and physically interacts with nucleoporin NUP50c to form a conserved NB module [41,42]. Notably, although only NUP136 contains canonical FG repeats, all components including GBPL3 and KAKU4, harbor long IDRs and promote perinuclear condensate formation [37,41,42], indicating that LLPS-dependent regulation. The physical interactions between lamina and Nup proteins and their properties of condensation enable them to form interconnected condensate networks that span the nuclear periphery for diverse molecular and physical functions. While plant systems offer valuable insights, it is important to recognize that many fundamental principles of NPC-associated phase separation have been established in other model organisms. In the C. elegans germline, specialized cytoplasmic condensates known as P granules extend into the NPC cytoplasmic filaments through their FG repeat interactions, driving perinuclear co-coalescence. These condensates at the NPC cytoplasmic face modulate RNA dynamics, including mRNA export [43,44]. More recently, a larger class of germ granules, termed D-compartments, has been identified in the C. elegans germline [45]. The cytoplasmic filament nucleoporins DDX-19, NUP214, and GLE1 are required for the formation of D-compartments themselves and influence the assembly of other surrounding germ granules (Figure 1C). Loss of these nucleoporins disrupts germline immortality and transgenerational epigenetic inheritance [45], identifying D-compartments as a novel class of NPC-associated germ granules essential for RNA surveillance. Similarly, plant NPC cytoplasmic ring is enriched P-body regulators [34]. However, the molecular function and physiological consequences of this association in plants remain unclear.
During mitosis, higher eukaryotic cells undergo nuclear envelope breakdown and reformation. Resealing the nuclear membrane at mitotic exit requires the endosomal sorting complexes required for transport (ESCRT) machinery and its adaptor protein LEM2 (Figure 1B). Notably, LEM2 condenses via LLPS and recruits ESCRT subunits, microtubules, and chromatin, to assemble a macromolecular O-ring seal at the membrane contact site [46]. Concurrently, Lamin B localizes to membrane recycling compartments with biomolecular condensate-like properties, contributing to mitotic partitioning, such as spindle organization and centrosome separation [47]. During meiosis, homolog recognition and pairing are facilitated by the attachment of chromosome pairing centers (PCs) to the nuclear envelope (NE). This attachment creates a phase-separated compartment that links to the chromosome axis through flexible DNA linkages [48]. Thus, phase separation contributes to nuclear envelope activities not only during interphase but also throughout cell division. Furthermore, the core components of the NE, together with its peripheral extensions and associated organelles, to coordinate a diverse array of cellular activities via LLPS.
Conclusions and Future Perspective
The NE is more than a passive physical barrier separating the nucleus and cytoplasm. It functions as a central signaling node orchestrating multifaceted interactions both across the nucleocytoplasmic divide and with proximal organelles. A growing body of research, including studies highlighted in this commentary, highlights phase separation in generating specialized perinuclear condensates (both membrane-less and membrane-bound) key to cellular organization and regulation. Unlike free cytoplasmic droplets, perinuclear condensates are tethered to the nuclear membrane. Moreover, the membrane interface itself may influence condensation through lipid composition and membrane tension, creating a unique microenvironment distinguishing NE-associated LLPS, and enabling directional, organized signal transduction. Recent work, such as that by Tang et al. (2025), advanced our understanding of phase separation at the nuclear lamina in plant stress responses, yet fundamental questions remain. Future research should determine the LLPS prevalence among key NE components and their associated factors, and elucidate how they: (1) sense cellular and environmental signals, (2) coordinate with surrounding macromolecules, and (3) transduce signals via dynamic biomolecular condensates. Addressing these questions in both plant and animal systems is essential for a comprehensive understanding of the NE as a phase-separation-mediated regulatory platform.
Conflict of Interest
The author declared no conflict of interest.
Acknowledgements
This work was supported by National Natural Science Foundation of China (grant number NSFC-2025-32500601). I also apologize to colleagues whose research could not be cited due to space limitations.
References
2. Naetar N, Ferraioli S, Foisner R. Lamins in the nuclear interior - life outside the lamina. J Cell Sci. 2017 Jul 1;130(13):2087–96.
3. Petrovic S, Mobbs GW, Hoelz A. Structure, function and assembly of nuclear pore complexes. Nat Rev Mol Cell Biol. 2026 Jan;27(1):35–54.
4. Makarov AA, Padilla-Mejia NE, Field MC. Evolution and diversification of the nuclear pore complex. Biochem Soc Trans. 2021 Aug 27;49(4):1601–19.
5. Tang Y, Huang A, Gu Y. Global profiling of plant nuclear membrane proteome in Arabidopsis. Nat Plants. 2020 Jul;6(7):838–47.
6. Janin A, Bauer D, Ratti F, Millat G, Méjat A. Nuclear envelopathies: a complex LINC between nuclear envelope and pathology. Orphanet J Rare Dis. 2017 Aug 30;12(1):147.
7. Tang Y. Plant nuclear envelope as a hub connecting genome organization with regulation of gene expression. Nucleus. 2023 Dec;14(1):2178201.
8. Hazelwood OS, Best NB, Ashraf MA. Function of nuclear envelope proteins in plant growth and development. Curr Opin Plant Biol. 2025 Jun;85:102738.
9. Holehouse AS, Kragelund BB. The molecular basis for cellular function of intrinsically disordered protein regions. Nat Rev Mol Cell Biol. 2024 Mar;25(3):187–211.
10. Boeynaems S, Alberti S, Fawzi NL, Mittag T, Polymenidou M, Rousseau F, et al. Protein Phase Separation: A New Phase in Cell Biology. Trends Cell Biol. 2018 Jun;28(6):420–35.
11. MacAinsh M, Muhammedkutty FNK, Prasad R, Zhou HX. Membrane Association of Intrinsically Disordered Proteins. Annu Rev Biophys. 2025 May;54(1):275–302.
12. Zhao YG, Zhang H. Phase Separation in Membrane Biology: The Interplay between Membrane-Bound Organelles and Membraneless Condensates. Dev Cell. 2020 Oct 12;55(1):30–44.
13. Prophet SM, Rampello AJ, Niescier RF, Gentile JE, Mallik S, Koleske AJ, et al. Atypical nuclear envelope condensates linked to neurological disorders reveal nucleoporin-directed chaperone activities. Nat Cell Biol. 2022 Nov;24(11):1630–41.
14. Kim J, Lee H, Lee HG, Seo PJ. Get closer and make hotspots: liquid-liquid phase separation in plants. EMBO Rep. 2021 May 5;22(5):e51656.
15. Emenecker RJ, Holehouse AS, Strader LC. Biological Phase Separation and Biomolecular Condensates in Plants. Annu Rev Plant Biol. 2021 Jun 17;72:17–46.
16. Ashwin SS, Nozaki T, Maeshima K, Sasai M. Organization of fast and slow chromatin revealed by single-nucleosome dynamics. Proc Natl Acad Sci U S A. 2019 Oct 1;116(40):19939–44.
17. Laghmach R, Di Pierro M, Potoyan DA. The interplay of chromatin phase separation and lamina interactions in nuclear organization. Biophys J. 2021 Nov 16;120(22):5005–17.
18. Strom AR, Emelyanov AV, Mir M, Fyodorov DV, Darzacq X, Karpen GH. Phase separation drives heterochromatin domain formation. Nature. 2017 Jul 13;547(7662):241–5.
19. Larson AG, Elnatan D, Keenen MM, Trnka MJ, Johnston JB, Burlingame AL, et al. Liquid droplet formation by HP1α suggests a role for phase separation in heterochromatin. Nature. 2017 Jul 13;547(7662):236–40.
20. Hilbert L, Sato Y, Kuznetsova K, Bianucci T, Kimura H, Jülicher F, et al. Transcription organizes euchromatin via microphase separation. Nat Commun. 2021 Mar 1;12(1):1360.
21. Berman BP, Weisenberger DJ, Aman JF, Hinoue T, Ramjan Z, Liu Y, et al. Regions of focal DNA hypermethylation and long-range hypomethylation in colorectal cancer coincide with nuclear lamina-associated domains. Nat Genet. 2011 Nov 27;44(1):40–6.
22. Nath S, Dutta S, Sarkar SD, Sengupta D, Mitra MK, Sengupta K. Liquid-liquid phase separation of lamin drives altered chromatin organization in cardiomyopathic mutations of lamin A. Nucleic Acids Res. 2025 Jul 19;53(14):gkaf615.
23. Köhler F, Bormann F, Raddatz G, Gutekunst J, Corless S, Musch T, et al. Epigenetic deregulation of lamina-associated domains in Hutchinson-Gilford progeria syndrome. Genome Med. 2020 May 25;12(1):46.
24. Good KV, Vincent JB, Ausió J. MeCP2: The Genetic Driver of Rett Syndrome Epigenetics. Front Genet. 2021 Jan 21;12:620859.
25. Jian Q, Xu Q, Xiang S, Wang R, Wang C, Zhang B, et al. Liquid-liquid phase separation: an emerging perspective on the tumorigenesis, progression, and treatment of tumors. Front Immunol. 2025 Jun 26;16:1604015.
26. Wang H, Dittmer TA, Richards EJ. Arabidopsis CROWDED NUCLEI (CRWN) proteins are required for nuclear size control and heterochromatin organization. BMC Plant Biol. 2013 Dec 5;13:200.
27. Tang Y, Dong Q, Wang T, Gong L, Gu Y. PNET2 is a component of the plant nuclear lamina and is required for proper genome organization and activity. Dev Cell. 2022 Jan 10;57(1):19–31.e6.
28. Tang Y, Liu X, Fang Y, Calvanese E, Hong Y, Gu Y. Nuclear lamina phase separation orchestrates stress-induced transcriptional responses in plants. Dev Cell. 2025 Dec 1;60(23):3156–66.e8.
29. Yin C, Sun A, Guo T, Mao X, Fang Y. Arabidopsis lamin-like proteins CRWN1 and CRWN2 interact with SUPPRESSOR OF NPR1-1 INDUCIBLE 1 and RAD51D to prevent DNA damage. Plant Cell. 2023 Sep 1;35(9):3345–62.
30. Qu Y, Yu Q, Chen Y, Ai H, Wu Y, Ye P, et al. Nuclear pore complex-mediated chromatin reorganization regulates the epigenetic landscape of osteoclasts during differentiation. Genes & Diseases. 2026 Mar 12:102142.
31. Ng SC, Biswas A, Huyton T, Schünemann J, Reber S, Görlich D. Barrier properties of Nup98 FG phases ruled by FG motif identity and inter-FG spacer length. Nat Commun. 2023 Feb 10;14(1):747.
32. Frey S, Rees R, Schünemann J, Ng SC, Fünfgeld K, Huyton T, et al. Surface Properties Determining Passage Rates of Proteins through Nuclear Pores. Cell. 2018 Jun 28;174(1):202–17.e9.
33. Celetti G, Paci G, Caria J, VanDelinder V, Bachand G, Lemke EA. The liquid state of FG-nucleoporins mimics permeability barrier properties of nuclear pore complexes. J Cell Biol. 2020 Jan 6;219(1):e201907157.
34. Tang Y, Yang X, Huang A, Seong K, Ye M, Li M, et al. Proxiome assembly of the plant nuclear pore reveals an essential hub for gene expression regulation. Nat Plants. 2024 Jun;10(6):1005–17.
35. Wang J, Pei G, Wang Y, Wu D, Liu X, Li G, et al. Phase separation of the nuclear pore complex facilitates selective nuclear transport to regulate plant defense against pathogen and pest invasion. Mol Plant. 2023 Jun 5;16(6):1016–30.
36. Wang J, Wu D, Pei G, Wang Y, Liu X, Tian S, et al. The pathogen effector BcSSP2 suppresses the NPC phase separation to facilitate Botrytis cinerea infection. Nat Commun. 2025 Sep 29;16(1):8610.
37. Tang Y, Ho MI, Kang BH, Gu Y. GBPL3 localizes to the nuclear pore complex and functionally connects the nuclear basket with the nucleoskeleton in plants. PLoS Biol. 2022 Oct 21;20(10):e3001831.
38. Guerreiro I, Kind J. Spatial chromatin organization and gene regulation at the nuclear lamina. Curr Opin Genet Dev. 2019 Apr;55:19–25.
39. Huang S, Zhu S, Kumar P, MacMicking JD. A phase-separated nuclear GBPL circuit controls immunity in plants. Nature. 2021 Jun;594(7863):424–9.
40. Reimann TM, Müdsam C, Schachtler C, Ince S, Sticht H, Herrmann C, et al. The large GTPase AtGBPL3 links nuclear envelope formation and morphogenesis to transcriptional repression. Nat Plants. 2023 May;9(5):766–84.
41. Mermet S, Voisin M, Mordier J, Dubos T, Tutois S, Tuffery P, et al. Evolutionarily conserved protein motifs drive interactions between the plant nucleoskeleton and nuclear pores. Plant Cell. 2023 Nov 30;35(12):4284–303.
42. Yang X, Ma Y, Geng B, Liu X, Liu Y, Sun J, et al. NUP50c defines a conserved nuclear basket module with NUP82 and NUP136 to mediate mRNA export and gene regulation in plants. Plant Cell. 2025 Oct 31;37(11):koaf260.
43. Sheth U, Pitt J, Dennis S, Priess JR. Perinuclear P granules are the principal sites of mRNA export in adult C. elegans germ cells. Development. 2010 Apr;137(8):1305–14.
44. Thomas LL, Bodas DM, Seydoux G. FG repeats drive co-clustering of nuclear pores and P granules in the C. elegans germline. Development. 2025 Mar 15;152(6):dev204585.
45. Lu P, Deng B, Li X, Niu X, Qiu Y, Liang Y, et al. A nuclear pore-anchored condensate enables germ granule organization and transgenerational epigenetic inheritance. Nat Struct Mol Biol. 2025 Jul;32(7):1241–54.
46. von Appen A, LaJoie D, Johnson IE, Trnka MJ, Pick SM, Burlingame AL, et al. LEM2 phase separation promotes ESCRT-mediated nuclear envelope reformation. Nature. 2020 Jun;582(7810):115–8.
47. Grindheim AK, Dale H, Novák J, Patil SS, Vedeler A, Saraste J. Annexin A2 and lamin B join membrane recycling compartments for the assembly of biomolecular condensates operating in mitotic partitioning. Front Cell Dev Biol. 2026 Jan 15;13:1744307.
48. Wang R, Tian Y, Liu L, Wang JH, Zhao L, Liu Y, et al. A DNA-containing phase-separated compartment links the nuclear envelope to chromosome axes to promote homolog pairing in C. elegans. Sci Adv. 2025 Oct 10;11(41):eadw5764.