Abstract
Introduction and objective: Iron deficiency is the most common cause of anemia in the United States (US). Several parenteral iron therapies with various dose and administration protocols are available in US. The objective of this analysis was to assess the effectiveness of parental iron products on hemoglobin (Hgb) normalization in real-world clinical practice.
Methods: Data were obtained from the Decision Resources Group (DRG) Real World Evidence US Data Repository. The parenteral iron dose required to correct for iron deficit was calculated using the modified Ganzoni equation.
Results: In total, 2,966 patients were included (68.2% female, 58.4% age ≥ 65 years). After controlling for differences in iron deficit level and other covariates, patients treated with ferric carboxymaltose (FCM) were more likely to have received their full repletion dose within 3 weeks of index date than patients treated with ferumoxytol (FM) (adjusted odds ratio [AOR]=5.10, p<0.001) and other parenteral iron products (OTH [sodium ferric gluconate in sucrose, iron dextran and iron sucrose]) (AOR=17.5, p<0.001). After controlling for differences in baseline Hgb level, receipt of full repletion dose and other covariates, patients treated with FCM were more likely to have normalized serum Hgb within 1 year of index than patients treated with FM (AOR=1.88, p<0.001) or OTH (AOR=1.40, p=0.004). Adjusted mean number of outpatient visits per patient within 1 year after index injection was lower in the FCM group compared with OTH (5.7 vs 11.8, p ≤ 0.001) and FM groups (5.9 vs 9.2, p ≤ 0.001).
Keywords
Iron deficiency anemia; Hemoglobin; Parenteral iron; Real world evidence
Introduction
Iron deficiency anemia (IDA) is caused by an insufficient iron supply for erythropoiesis that leads to a reduction in hemoglobin (Hgb) and circulating red blood cells [1,2]. Iron deficiency is the most common cause of anemia in the United States (US) and occurs most often from blood loss and in patients with chronic diseases and inflammation [3,4]. It was estimated that approximately 10 million people are iron deficient in the US, including 5 million who have IDA [2].
Iron deficiency anemia is associated with burdensome clinical symptoms such as fatigue, dyspnea, headaches, paleness and vertigo [5]. In addition, iron deficiency may reduce an individual’s capacity to exercise [6,7], worsen comorbid diseases and reduce cognitive function [8-10]. An analysis of multiple well-validated patientreported outcome assessments found that people with iron deficiency anemia experience a lower quality of life (QoL) compared with those with normal iron levels [11]. In a cross-sectional observational study, anemia was associated with greater fatigue (p<0.001), lower handgrip strength (p=0.014), an increased number of disabilities (p=0.005) and more depressive symptoms (p=0.002) [12]. Iron deficiency constitutes a significant economic burden to society, increasing health care resource use, reduced work productivity and absenteeism from work [12,13].
The goal of iron replacement therapy is to replenish iron stores and restore Hgb levels [14]. Although oral iron therapy is routinely prescribed for iron deficiency [15], parenteral iron is necessary for patients who cannot tolerate or are non-responsive to oral iron. Parenteral iron is also used in patients with chronic diseases such as inflammatory bowel disease (IBD), cancer, chronic kidney disease (CKD) or when rapid iron replacement is desirable [16]. Several parenteral iron therapies are currently available in the US, including ferric carboxymaltose ([FCM] Injectafer; Luitpold Pharmaceuticals, Shirley, NY, USA), ferumoxytol ([FM] Feraheme; AMAG Pharmaceuticals, Inc., Waltham, MA, USA), iron dextran (INFeD; Allergan, Inc., Ferentino, Italy), sodium ferric gluconate complex in sucrose (Ferrlecit; Sanofi-Aventis, Bridgewater, NJ, USA), and iron sucrose (Venofer; Luitpold Pharmaceuticals, Shirley, NY, USA), which vary in dose and administration protocols [16].
Choice of iron product should depend on individual patient need, tolerability and ease of administration. Iron deficit levels vary between patients; therefore, different doses of iron are required for complete repletion. In addition, patients can achieve iron repletion with less frequent dosing with products that provide more iron per dose. There are limited data on the effectiveness of various parenteral iron products in the real-world setting, and specifically, how choice of therapy affects Hgb normalization. The objective of this analysis was to assess the impact of various parenteral iron products on the likelihood of patients receiving their full expected dose and the likelihood of achieving Hgb normalization in realworld clinical practice.
Methods
Data Source
This observational, retrospective study used real-world data from the Decision Resources Group (DRG) Real World Evidence repository [17], which links medical claims, prescription claims and electronic health records (EHRs) to provide longitudinal patient-level data. The repository covers the majority of the US healthcare system, representing >300 million patients’ medical and pharmacy claims, and electronic health record data. Claims and EHR data are sourced separately and linked together by a Health Insurance Portability and Accountability Act (HIPAA)-compliant encrypted patient key generated by a third party.
Study Design
Patients (aged ≥ 18 years) who were treated with parenteral iron therapy from 3/1/2015−2/28/2017 were included. Patients were required to have their most recent baseline Hgb level recorded <30 days prior to or on the date of their index (first) parenteral iron therapy claim. The most recent baseline Hgb level was required to be below the normal serum Hgb level (<12 g/dL for females and <13.5 g/dL for males). Patients were excluded if they received parenteral iron therapy 6 weeks prior to their index date, had received dialysis, or had insufficient data to identify comorbidities 1 year prior to index date (Figure 1).
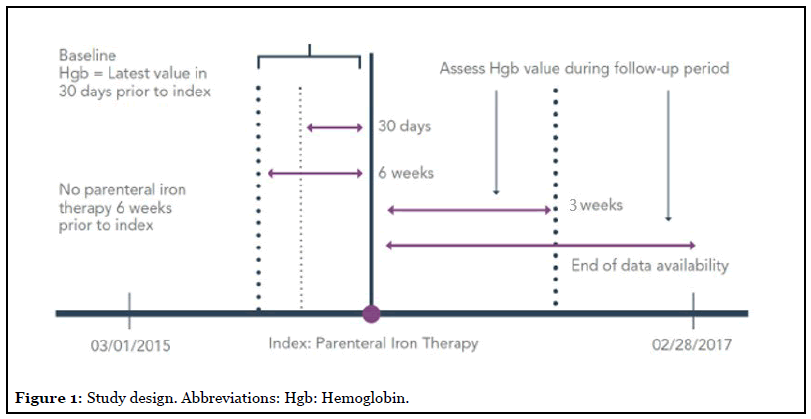
The parenteral iron dose required to correct for iron deficit was calculated using the modified Ganzoni equation [1]. The proportion of patients receiving the full parenteral iron dose required to correct for iron deficit during the 3 weeks after index date, along with the proportion of patients achieving normalized Hgb levels within 1 year after index date were summarized for patients receiving ferric carboxymaltose (FCM), ferumoxytol (FM) and other parenteral iron products (OTH [sodium ferric gluconate in sucrose, iron dextran and iron sucrose]). The categorization of parenteral iron products was based on the amount of iron dose per administration. FCM and FM deliver 750 mg and 510 mg of iron per administration, respectively. Sodium ferric gluconate in sucrose, iron dextran and iron sucrose deliver lower amount of iron (ranging from 50 mg to 200 mg) per administration.
Analysis
The study assessed the clinical effectiveness of different iron products in providing full repletion iron dose and restoring serum Hgb to normal levels in the real-world setting. Multivariable logistic regression models were used to assess how choice of iron product may affect the likelihood of a patient receiving their full repletion dose within 3 weeks of index date, controlling for iron deficit levels, and other compounding factors such as gender, age, comorbid conditions, and use of prescription oral iron therapy. Treatment differences on the likelihood of achieving Hgb normalization within 1 year of start of therapy were evaluated using logistic regressions while controlling for gender, age, comorbidities, prescription oral iron use, baseline Hgb, and if full repletion dose was received within 3 weeks of index date. Differences in healthcare resource utilization in the 12 months after index date between treatment groups were assessed by negative binomial regression, adjusting for Hgb normalization, age, gender, comorbid conditions, and use of prescription oral iron therapy. Pairwise treatment comparisons were made using separate regression models. Data were analyzed using the standard statistical packages of StataCorp 2017. (Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC).
Results
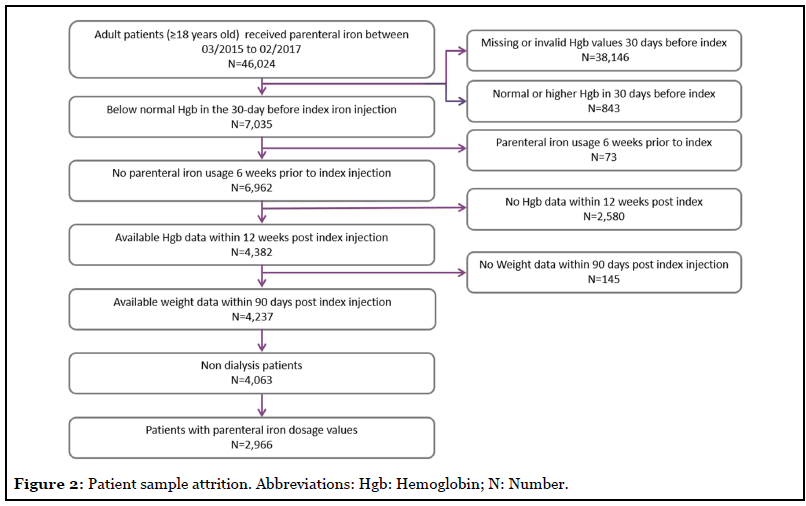
Of the 46,024 patients who received parenteral iron therapy between March 2015 and February 2017, 2,966 patients provided complete claims and EHR data and were included in the analysis (68.2% were female, and 58.4% were age ≥ 65 years). Comorbid conditions, as identified by medical records among the patient cohort, included chronic kidney disease (CKD) (60.1%), congestive heart failure (CHF, 50.1%), inflammatory bowel disease (IBD) (64.4%), and cancer (59.5), (Figure 2 and Table 1). Most patients (95%) did not receive prescription oral iron therapy within 3 months before or after index date.
| All Patients | FCM | FM | Received OTH† | |
|---|---|---|---|---|
| N (%) | N (%) | N (%) | N (%) | |
| Overall Sample | 2,966 (100.0) | 685 (100.0) | 406 (100.0) | 1,875 (100.0) |
| Gender | ||||
| Female | 2,020 (68.1) | 435 (63.5) | 260 (64.0) | 1,325 (70.7) |
| Male | 946 (31.9) | 250 (36.5) | 146 (36.0) | 550 (29.3) |
| Age | ||||
| 18–44 years | 443 (14.9) | 87 (12.7) | 21 (5.2) | 335 (17.9) |
| 45–54 years | 351 (11.8) | 94 (13.7) | 41 (10.1) | 216 (11.5) |
| 55–64 years | 439 (14.8) | 100 (14.6) | 47 (11.6) | 292 (15.6) |
| 65+ years | 1,733 (58.4) | 404 (59.0) | 297 (73.2) | 1,032 (55.0) |
| Comorbidities | ||||
| CKD | 1,783 (60.1) | 309 (45.1) | 324 (79.8) | 1,150 (61.3) |
| CHF | 1,513 (51.0) | 254 (37.1) | 245 (60.3) | 1,014 (54.1) |
| IBD | 1,167 (39.3) | 201 (29.3) | 129 (31.8) | 837 (44.6) |
| Cancer | 1,764 (59.4) | 393 (57.4) | 232 (57.1) | 1,139 (60.8) |
| Prescription oral iron therapy | ||||
| No prescription oral therapywithin 3 months pre- or post- index | 2,817 (95.0) | 661 (96.5) | 384 (94.6) | 1,772 (94.5) |
| 3 months pre-index | 71 (2.4) | 17 (2.5) | 14 (3.5) | 40 (2.1) |
| 3 months post-index | 37 (1.2) | 1 (0.2) | 3 (0.7) | 33 (1.8) |
| 3 months pre- and post-index | 41 (1.4) | 6 (0.9) | 5 (1.2) | 30 (1.6) |
Abbreviations: CKD: Chronic Kidney Disease; CHF: Congestive Heart Failure; FCM: Ferric Carboxymaltose; FM: Ferumoxytol; IBD: Inflammatory Bowel Disease; N: Number; OTH: Other parenteral iron products.
†Sodium ferric gluconate in sucrose, iron dextran and iron sucrose.
Table 1: Cohort demographics.
Most patients were treated with iron sucrose (46.2%, n=1,369), followed by FCM (23.1%, n=685), FM (13.7%, n=406), iron dextran (13.6%, n=404), and sodium ferric gluconate complex in sucrose (3.4%, n=102) (Figure 3).

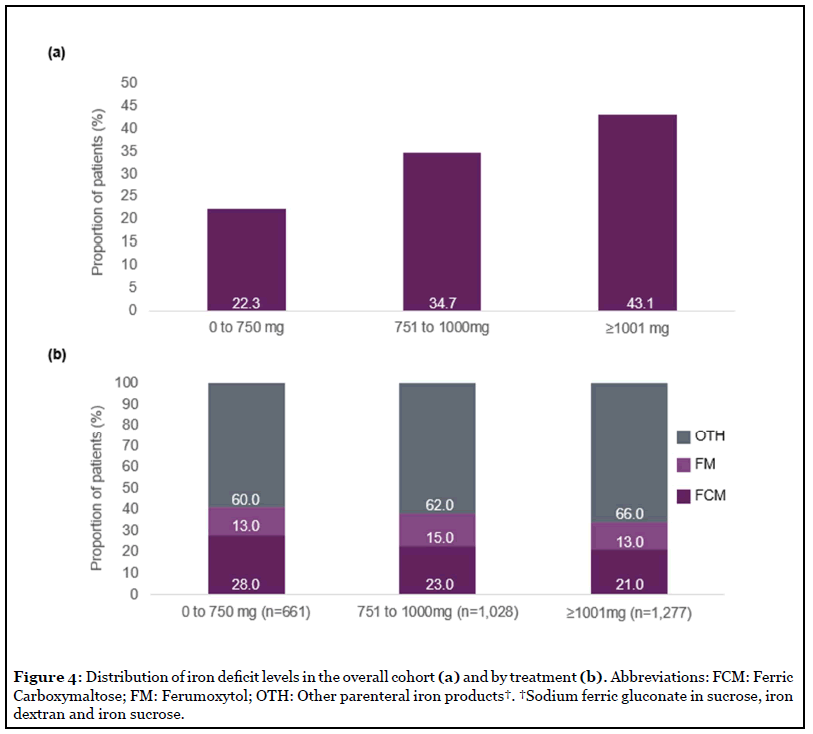
Iron deficit levels varied across patients (Figure 4). Overall, 77% of patients had an iron deficit of >750 mg, and 43.1% had an iron deficit of >1,000 mg. Product utilization did not differ by iron deficit levels, with 60.0% of patients with iron deficit of 0 to 750 mg and 66% of patients with iron deficit of >1,000 mg using products with lower mg of iron per dose (Figure 4b). Mean (SD) iron deficit was similar between FCM (966 mg [303]) and FM (1,007 mg [317]) (p=0.104), and higher for OTH (1,011 mg [313]) when compared to FCM (p=0.004). Mean (standard deviation [SD]) number of injections received within 3 weeks of index injection was 1.83 (0.64) for the FCM group, 1.93 (0.67) for the FM group and 1.87 (0.65) for the OTH group.
Overall, 33.9% of patients (n=1,006) received the full required parenteral iron dose within 3 weeks after index injection. Mean (SD) total iron dose received by patients treated with FCM, FM and OTH patients within 3 weeks of index date was 1,277.1 mg (446.9), 842.5 mg (357.6), and 534.2 mg (584.8), respectively.
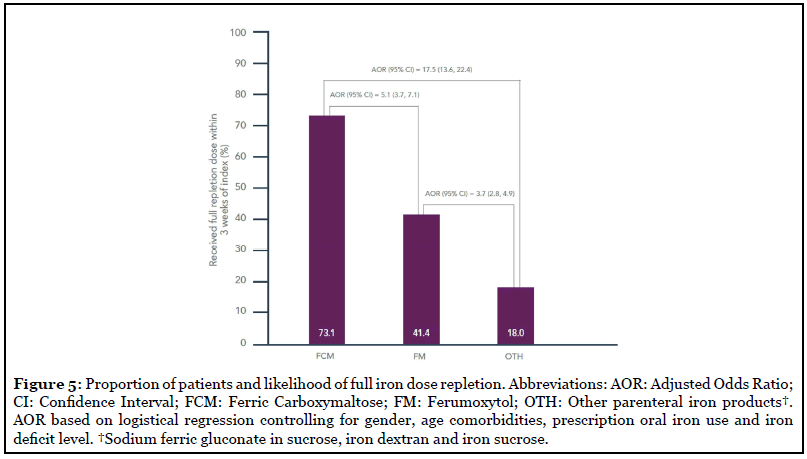
The highest proportion of patients receiving the full iron repletion dose within the 3 weeks after index date was in the FCM group (73.1%), followed by FM (41.4%) and OTH (18.0%) groups (Figure 5). After controlling for differences in iron deficit level and other covariates, patients treated with FCM were significantly more likely to have received their full repletion dose within 3 weeks of index date than patients treated with FM (adjusted odds ratio [AOR]=5.10, p<0.001) and OTH (AOR=17.5, p<0.001) (Figure 5). Patients treated with FM also had a higher likelihood of receiving full dose than patients treated with OTH (AOR=3.71, p<0.001) (Figure 5).
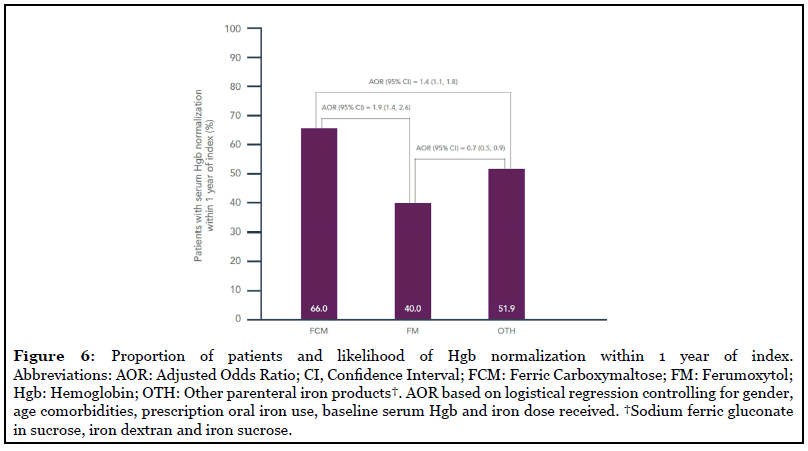
In total, 1,599 (53.9%) patients had normalized Hgb levels within 1 year of treatment initiation (median study follow-up=108 weeks). Across treatment cohorts, 66.6% of FCM patients, 40.0% of FM patients and 51.9% of OTH patients had normalized Hgb within 1 year of index date (Figure 6). After controlling for differences in baseline Hgb level, receipt of full repletion dose and other covariates, patients treated with FCM were significantly more likely to have normalized serum Hgb within 1 year of index than patients treated with FM (AOR=1.88, p<0.001) or OTH (AOR=1.40, p=0.004) (Figure 6). Patients treated with FM had lower likelihood of having normalized serum Hgb than patients treated with OTH (AOR=0.69, p=0.004) (Figure 6).
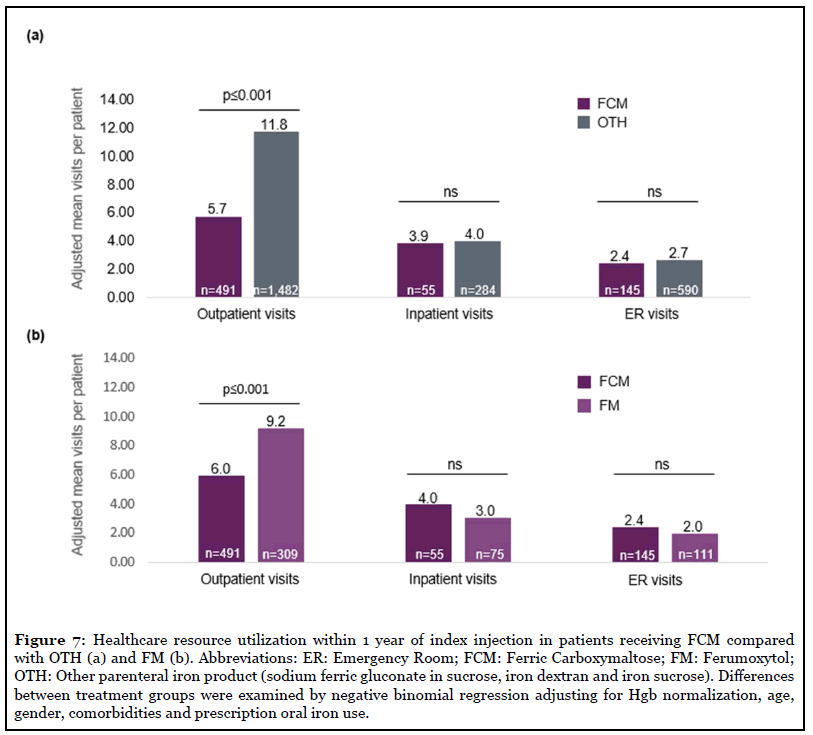
The choice of iron therapy impacted the number of outpatient visits. Adjusted mean number of outpatient visits per patient within 1 year after index injection was lower in the FCM group compared with OTH (5.7 vs 11.8, p ≤ 0.001) and FM groups (5.9 vs 9.2, p ≤ 0.001) after controlling for differences in baseline Hgb level and other covariates (Figure 7). Inpatient and emergency room (ER) visits were not significantly different between treatment groups.
Discussion
Anemia is a global public health problem that is associated with an increased risk of morbidity and mortality [3,18,19]. A previous study in US patients highlighted the importance of initial complete parenteral iron repletion for improving clinical outcomes [20], but there are limited data on realworld effectiveness of currently available parenteral iron products. This study assessed Hgb normalization with various parenteral iron products in a real-world setting using a large US database.
The goal of iron replacement therapy is to replenish iron stores and restore Hgb to a normal level. Therefore, understanding the underlying iron requirement is important for full iron repletion. The modified Ganzoni equation has been developed to estimate iron deficit level but it is not routinely adopted for use in clinical practice [1]. A simplified dosing method based on serum Hgb level and body weight has also been used [21]. When choosing an iron product, clinicians should consider the amount of iron delivered per dose, dosing frequency, and how well the iron is absorbed and tolerated for the individual patient. Patients with a high iron deficit may benefit from a product that administers more iron per dose to achieve iron repletion with less frequent dosing and fewer treatment visits, reducing burden to the patient and health care system. A more convenient dosing regimen may also improve patient compliance and treatment effectiveness.
This study highlights the importance of choice of iron therapy product for achieving normalized Hgb levels in patients with iron deficiency, and the potential of products with a higher iron dose per administration meeting the needs of many IDA patients. The results of this study show that many patients with high iron deficit do not receive products with more iron per dose in real-world practice. Mean iron deficit level was higher among patients receiving lower dose intravenous (IV) iron products compared with those receiving FCM, which administers more iron per dose. Almost half (45.0%) of patients in the OTH group had a calculated iron deficit >1,000 mg, and only 34.0% of patients with iron deficit >1,000mg used FCM or FM. Patients receiving products with less iron per dose would require more treatment visits to replenish their iron stores. Despite a higher number of outpatient visits in OTHtreated patients than FCM-treated patients observed in this analysis, the likelihood of full iron dose repletion and serum Hgb normalization remained lower with patients receiving OTH vs FCM. These findings suggest that the more frequent outpatient visits may not be enough to provide sufficient iron repletion for OTH patients with high iron deficit levels. It is also possible that patients may not be fully compliant with visits to clinic to receive iron dose on a regular basis. There is an unmet need among patients with high iron deficit for treatment optimization that deserves further investigation.
The FCM group had the highest likelihood to have normalized serum Hgb within 1 year of index than patients treated with FM or OTH. Surprisingly, FM patients had a lower likelihood of achieving Hgb normalization compared with OTH. This result should be interpreted with caution. The proportion of patients with chronic kidney disease in the FM group was higher than OTH group (79.8% vs. 61.3%). Although treating IDA in non-dialysis-dependent patients with chronic kidney disease is beneficial, previous studies suggested the target Hgb of >13 g/dl might increase the risk of cardiovascular disease in these patients [22]. In the current analysis, serum Hgb normalization was defined as 12 g/dL for females and 13.5 g/dL for males. It is possible that clinicians might have targeted a lower serum Hgb level for CKD patients to reduce the risk of cardiovascular disease in clinical practice [23], resulting in a lower proportion of patients achieving serum Hgb normalization in the FM group. Further research is warranted to confirm the study findings.
This study has several limitations. Due to the open network nature of the databases used in the current analysis, patients’ continuous eligibility cannot be ascertained. Health care services provided by out-of-network providers may not be captured by the databases. This can lead to under-reporting of health care resource utilization data in the study. Real-world data collected from routine clinical practice are subject to coding errors, missing data, and variations in reporting across clinical practices, and the use of concomitant over-the-counter iron replacement is not well-documented. Patients who did not have complete Hgb data recorded in electronic medical records were excluded from the study, which resulted in 2,580 patients being excluded from the analysis, potentially limiting the generalizability of current study findings. Finally, although multivariable regression analyses have controlled for potential confounding due to the presence of CKD and other comorbidities between treatment groups, the severity of comorbid conditions were not assessed. Tolerability of iron products may vary by individuals and it might have affected therapy choice for patients in the study. This information was not available in structured electronic medical records. Case-mix adjustment remains a limitation of comparative effectiveness research based on retrospective analysis of claims and electronic medical records.
Conclusion
The results of this analysis suggest that choice of iron replacement may affect the likelihood of Hgb normalization for patients in real-world practice, especially those with high iron deficit levels. These findings highlight the importance of choosing appropriate iron replacement product to optimize serum Hgb normalization in patients with IDA.
Acknowledgements
The authors received writing support from Ashley Newcombe and Sophie Doran, employees of DRG Abacus, part of Clarivate.
Disclosers and Conflicts of Interest
This study was supported by Daiichi Sankyo, Inc. (DSI). Jackie Kwong is an employee of DSI. Chris LaVallee, Patrick Cronin and Isha Bansal are employees of Decision Resources Group (DRG), a healthcare research and consulting company. Ralph Boccia was a consultant to DSI, Amgen, Bristol Myers Squibb, AMAG, SecuraBio, and was on the speaker’s bureau of Rigel and Celgene during the time of this study. Work by DRG was funded by DSI. The authors declare that there are no further conflicts of interest.
References
2. Miller JL. Iron deficiency anemia: a common and curable disease. Cold Spring Harbor Perspectives in Medicine. 2013 Jul 1;3(7):a011866.
3. WHO. The global prevalence of anaemia in 2011. Geneva: World Health Organization. 2015.
4. Poggiali E, De Amicis MM, Motta I. Anemia of chronic disease: a unique defect of iron recycling for many different chronic diseases. European Journal of Internal Medicine. 2014 Jan 1;25(1):12-7.
5. Lopez A, Cacoub P, Macdougall IC, Peyrin-Biroulet L. Iron deficiency anaemia. The Lancet. 2016 Feb 27;387(10021):907-16.
6. Beard J, Tobin B. Iron status and exercise. The American Journal of Clinical Nutrition. 2000 Aug 1;72(2):594S-7S.
7. Ebner N, Jankowska EA, Ponikowski P, Lainscak M, Elsner S, Sliziuk V, et al. The impact of iron deficiency and anaemia on exercise capacity and outcomes in patients with chronic heart failure. Results from the studies investigating co-morbidities aggravating heart failure. International Journal of Cardiology. 2016 Feb 15;205:6-12.
8. Jáuregui-Lobera I. Iron deficiency and cognitive functions. Neuropsychiatric Disease and Treatment. 2014;10:2087-95.
9. Murray-Kolb LE, Beard JL. Iron treatment normalizes cognitive functioning in young women. The American Journal of Clinical Nutrition. 2007 Mar 1;85(3):778-87.
10. Cappellini MD, Comin-Colet J, de Francisco A, Dignass A, Doehner W, Lam CS, et al. Iron deficiency across chronic inflammatory conditions: International expert opinion on definition, diagnosis, and management. American Journal of Hematology. 2017 Oct;92(10):1068-78.
11. Strauss WE, Auerbach M. Health-related quality of life in patients with iron deficiency anemia: impact of treatment with intravenous iron. Patient related outcome measures. 2018;9:285-98.
12. Thein M, Ershler WB, Artz AS, Tecson J, Robinson BE, Rothstein G, et al. Diminished quality of life and physical function in community-dwelling elderly with anemia. Medicine. 2009 Mar;88(2):107-14.
13. Ershler WB, Chen K, Reyes EB, Dubois R. Economic burden of patients with anemia in selected diseases. Value in Health. 2005 Nov 1;8(6):629-38.
14. Johnson-Wimbley TD, Graham DY. Diagnosis and management of iron deficiency anemia in the 21st century. Therapeutic Advances in Gastroenterology. 2011 May;4(3):177-84.
15. Goddard AF, James MW, McIntyre AS, Scott BB. Guidelines for the management of iron deficiency anaemia. Gut. 2011 Oct 1;60(10):1309-16.
16. Auerbach M, Macdougall I. The available intravenous iron formulations: history, efficacy, and Toxicology. Hemodialysis International. 2017 Apr;21:S83-92.
17. DRG. Decision Resources Group (DRG) Real World Evidence US Data Repository. Available at: https:// decisionresourcesgroup.com/solutions/real-world-data/ Accessed: March 2015
18. Zakai NA, Katz R, Hirsch C, Shlipak MG, Chaves PH, Newman AB, et al. A prospective study of anemia status, hemoglobin concentration, and mortality in an elderly cohort: the Cardiovascular Health Study. Archives of Internal Medicine. 2005 Oct 24;165(19):2214-20.
19. Culleton BF, Manns BJ, Zhang J, Tonelli M, Klarenbach S, Hemmelgarn BR. Impact of anemia on hospitalization and mortality in older adults. Blood. 2006 May 15;107(10):3841-6.
20. LaVallee C, Cronin P, Bansal I, Kwong WJ, Boccia R. Importance of Initial Complete Parenteral Iron Repletion on Hemoglobin Level Normalization and Health Care Resource Utilization: A Retrospective Analysis. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy. 2019 Oct;39(10):983-93.
21. Australia Nba. Iron product choice and dose calculation for adults. Available at: https://www.blood. gov.au/system/files/documents/Iron%20product%20 choice%20and%20dose%20calculation20052016.pdf. 2016.
22. Clement FM, Klarenbach S, Tonelli M, Johnson JA, Manns BJ. The impact of selecting a high hemoglobin target level on health-related quality of life for patients with chronic kidney disease: a systematic review and meta-analysis. Archives of Internal Medicine. 2009 Jun 22;169(12):1104-12.
23. Mimura I, Tanaka T, Nangaku M. How the target hemoglobin of renal anemia should be?. Nephron. 2015;131(3):202-9.
