Abstract
Reverse triggering is the ventilator-triggered muscular effort, related to entrainment, particularly seen among sedated patient. The reverse triggering frequently caused double triggering/double cycling, with increased tidal volume and transpulmonary pressure. In our monitored patients we have observed different types of double triggering caused by asynchrony like to reverse triggering, for which we adopted the term “pseudo-reverse-triggering”. To our knowledge, we described this asynchrony for the first time both in assist-controlled ventilation as well as during pressure support ventilation. These asynchronies must be recognized in clinical practice to be corrected in a personalized way.
Keywords
Reverse triggering, Pseudo-reverse-triggering, Double triggering, Double cycling, Advanced respiratory monitoring
Case report
The double trigger (DT) is the second asynchrony in frequency, after the failed cycles [1]. Compared to the latter, it can be more injurious since it usually increases tidal volume (Vt), transmural and transvascular pressure to levels that can cause ventilator-induced lung damage [2,3]; and self-inflicted by the patient (P_SILI) [4], as well diaphragmatic injury. DT and Reverse triggering (RT) may be related, although it is not always the case. However, the relationship between DT and a vigorous effort by the patient is a common feature.
We present three cases of critically ill patients admitted to the ICU who presented this asynchrony in the onset of the weaning period of the mechanical ventilation (MV). All the patients were sedated, RASS score 4.
The first case corresponds to a 54-year-old male patient, with a history of kidney transplantation and COVID-19 pneumonia, with 10 days MV, in volume-controlled assisted mode with a constant flow (ACV) (Figure 1). The second case was a 62-year-old male patient who was admitted for subarachnoid hemorrhage, complicated with an infraction due to vasospasm. After reduced sedation, monitoring was performed after four days during ACV mode (Figure 2). The third case is a 57-year-old male patient on the first postoperative day of double lung transplantation due to histiocytosis X; in this case, monitoring was performed during pressure support ventilation (PSV), (Figure 3). As a representative example of true RT, although without DT, we show the case four, the monitoring of a 69-year-old woman with COVID-19 pneumonia, during PSV, and after 19 days in MV (Figure 4).
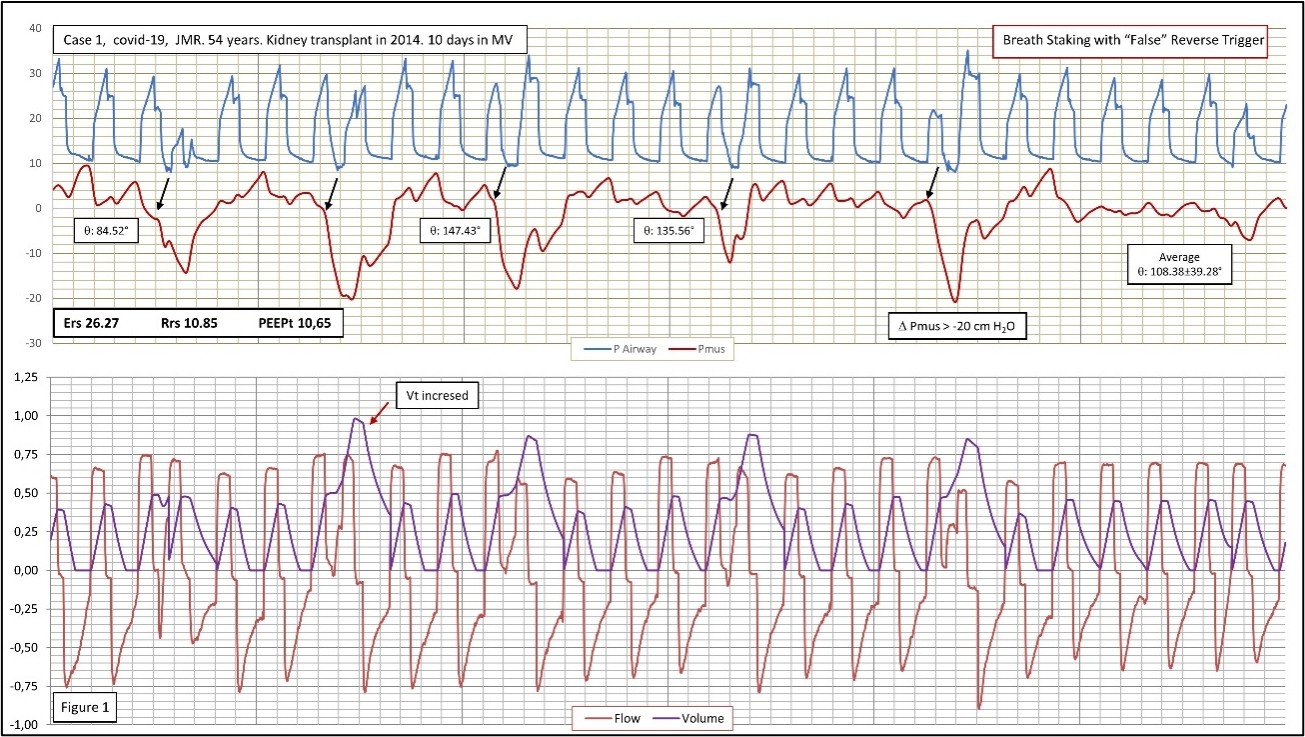
Figure 1. Patient 1 with pneumonia caused by COVID-19. 23 consecutive cycles corresponding to 60 sec are represented (5 with breath staking). ACV mode with continuous flow; Breath Staking is due to the overlapping of the patient’s respiratory driver in the cycles programmed in the ventilator. We have used the last 5 completely passive cycles to obtain, by using Linear Multiple Regression, the variables necessary for the equation of movement of the respiratory system: Elastance (Ers), Resistances (Rrs) and Total PEEP (PEEPt). Pmus, in red, is obtained according to the formula: Pmus=Paw-(Ers *Volume +Rrs *Flow +(PEEPtotal -PEEPapparent). The onset of the patient's neural driver is represented by a black arrow. The delay between the neural cycles to the cycle of the ventilator is expressed as angular phase. The angular dispersion is highly variable. The neural impulse appears rhythmic, vigorous (? -20 cm H2O), autonomous, and not entrainment by the ventilator' cycles. We labeled this case as VDT (Ventilator Double Trigger) and it corresponds to the original description of Pseudo-double Triggering [5]; while some will call it Reverse Triggering with Breath Staking.
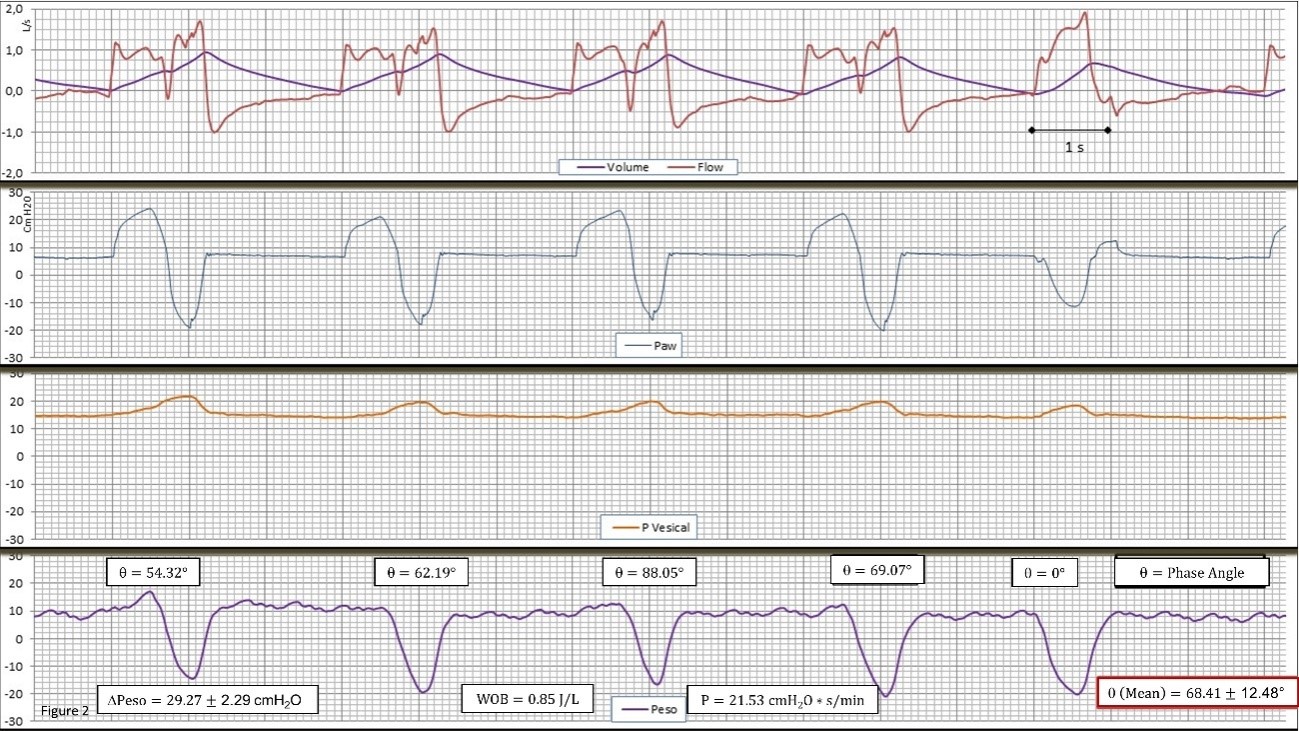
Figure 2. Record of flow (Flow) and volume (Volume), airway pressure (Paw), bladder pressure (Pvesical), and esophageal pressure (Peso) in volume-assisted ventilation and continuous flow in the case 2. Note in the first four consecutive double cycling cycles of the DTV type. In all cycles there is a vigorous driver of the patient, which is evident in the delta of the Peso, although at a lower frequency than that prescribed in the ventilator. It can be seen how in cycle 5, the patient triggers the inspiration of the ventilator in the form of a typical assisted without DT or RT. As can be seen in the figure, there are two well-defined drivers, one by the machine and the other by the patient. Both are similar in intensity (patient's WOB at 0.85 J/L and PTP of 222.53 cm H2O*s/L). The patient's inspiratory effort is similar in all cycles, except in the 5th cycle that is assisted, which suggests an autonomous patient intense driver (delta Peso approximately 30 cm H2O), less frequent and regular than the one of the machine. The measurement of the angular phase (θ=68.41 ± 12.48°) between the patient and ventilator cycles is widely variable, which also goes against a reflex mechanism. In this case the DT-RT 1:1 ratio appeared in 60% of the registry, the rest being variable with a ratio of 1:2 and 1:3. In this case the Vt is appreciate increase coincident with DTV, almost the double. At the same time, the I/E has been reduced to 1:1, a presumably undesired effect. These findings highlight the need for closely monitoring for protective ventilation purpose.
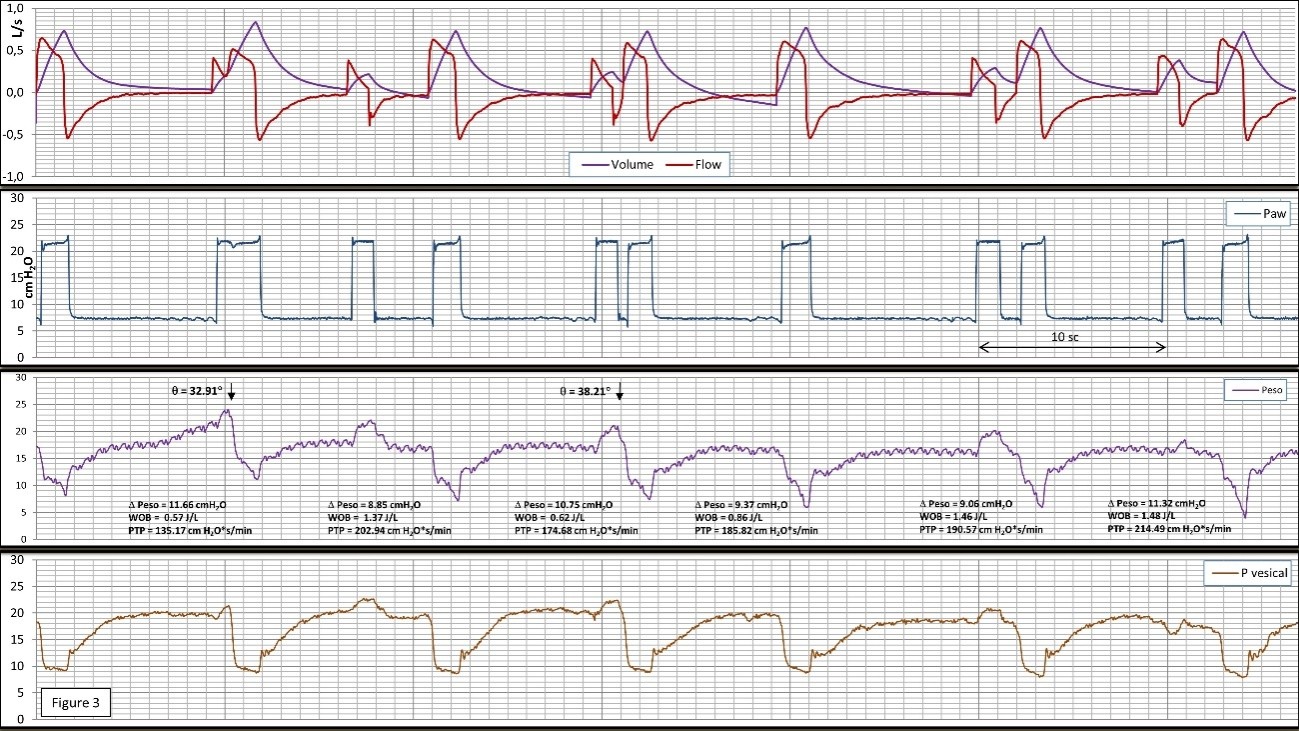
Figure 3. Recording of the signals (Flow and volume), Paw, Peso, and bladder pressure (Pvesical) during ventilation with support pressure in case 3. In this case, we show a double cycling defined as DTA, by auto-cycling of the ventilator; but the patient driver (Reverse Trigger in the broad sense is due to the release of intense active expiration, which is evident in the bladder signal. The contraction of the diaphragm is subsequent to the relaxation of the active expiration. To think that this driver, which includes release of forced expiration and subsequent inspiratory effort of the patient, is the cause of reflex phenomenon is highly unlikely. The relationship between the mechanical and the neural cycle is variable in this case. The parameters related to the inspiratory effort: Delta Peso: 11.55 ± 1.22 cm H2O, and the increase in the double trigger cycles can be noticed; WOB 1.06 ± 0.42 J/L and PTP 183.95 ± 27.59 cm H2O*s/min. In this case, WOB has been related to the volume generated by the stacked cycle corresponding to double cycling. The appearance of double cycling showed a variable relationship.
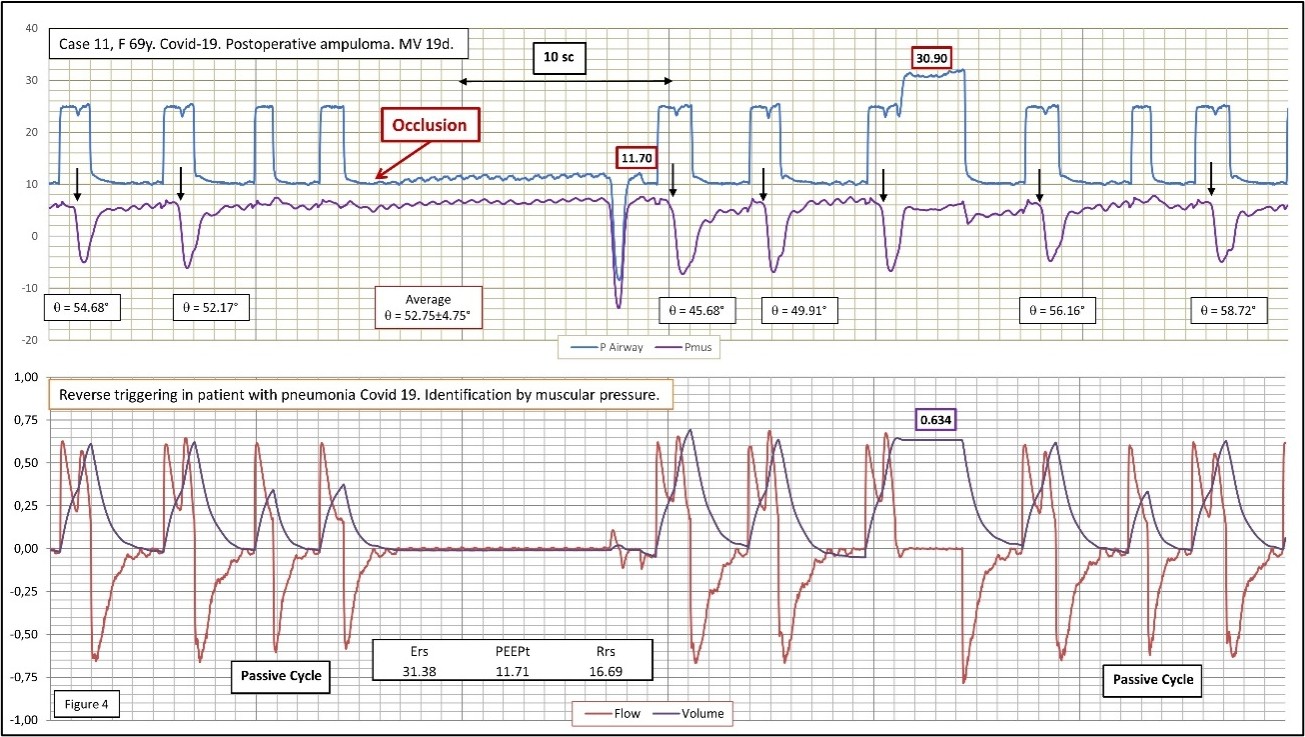
Figure 4. Representative case of "true" RT without double cycle in this case. The patient also show pneumonia caused by COVID-19, as indicate in text, ventilated in PSV mode, Inspiratory pressure 15/10 cm H2O PEEP. Main findings: Auto-triggering followed by RT. In our patients, it is the most frequent asynchrony owing to RT during PSV. Pmus has been lowered by 5 cm for a better visualization of Paw. An expiratory occlusion demonstrates a much slower but equally intense spontaneous respiratory driver than the RT. The neural impulse of RT appears after the onset of the mechanical cycle with enough uniform delay (measured as angular phase). The beginning of each cycle with the RT is indicated in the figure with a black arrow. Cycles 3 and 9 are completely passive. Pmus is obtained by the equation of motion as described in Figure 1. How the volume generated by RT is almost double that the volume set can be noticed, which causes an increase in plateau pressure to more than 30 cm H2O.
Data Acquisition
In second and third patients; the flow signal was obtained using a Hans Rudolph 4700® pneumotachograph connected to a Sensortechnics® differential pressure transducer, LBAS025B. Airway pressure (Paw), Esophageal (Peso) and Gastric (Pgas) were obtained using a Sensortechnics® SQ7500 pressure transducer. Vesical pressure (Pvesical) was obtained using a vascular sensor (Edwards TruWave®). Peso and Pgas measurements were obtained using a specific probe, Nutrivent, Sindam®. All signals were acquired by Teensy 3.5® analogue-to-digital converter sampled at 1045 Hz/channel for 60 min.
For the first and fourth patients, both patients with COVID-19, the respiratory signals were obtained with connection to the Medibus protocol of the Evita ventilator using personal software, by the acquisition of Paw, flow and volume signals sampled at 125 Hz/channel. This methodology allowed us to preserve our usual monitoring system of contamination. We have replaced the parameters derived from esophageal monitoring with the muscular pressure deduced from the equation of motion (Figures 1 and 4).
In both monitoring systems, the parameters related to effort and respiratory works have been analyzed, as well as the delay between the commencement of the ventilator and the neural breath (dP). The dP divided by duration de mechanical cycle (Ttot) and multiplied by 360° provide the phase angle (θ). The frequency and ratio of the association of the two phenomena DT and RT, were also recording.
In the first and the second case, after assessing asynchrony, the ventilatory mode was changed to PSV, improving interaction with the ventilator. In the third case, it was necessary to increase the sedation. In case four, during PSV, inspiratory pressure was increased.
DT was initially described as a consequence of an intense and wide inspiratory muscular effort that caused double cycling of the ventilator [1].
False double triggering (Pseudo-double-triggering) was described as a double cycle that occurred after a cycle initially initiated by the ventilator followed by a subsequent inspiratory neuromuscular impulse that caused the second cycle of the ventilator [5].
At the same time, a very similar phenomenon was described in patients with respiratory distress consisting of the ventilator-triggered muscular efforts. This term was described as RT, and which could promote DT (6). This muscular effort, apparently triggered by the ventilator, has been attributed to a reflex phenomenon. In principle, the respiratory driver of the patient can be neuro-coupled or entrainment by this reflex mechanism [6].
Subsequently, DT has been classified into three variants [7]: DT with the initial impulse generated by the patient (DTP), which is equivalent to the original DT, and Double trigger with the initial impulse generated by the set rate in the ventilator followed by the neuromuscular impulse of the patient (DTV) that is equivalent to the false double trigger. That last has a variant: due to auto-triggering of the ventilator (DTA). The latter two variants are referred by other authors as DTs due to RT.
Note that RT may promote or not double cycling (DC) and that in both cases it will be accompanied by an increase in elevated transpulmonary pressure and volume, a good example is provided in the figures, and therefore potentially harmful both pulmonary (VILI, P-SILI and diaphragmatic due to polymeric contraction.
The existence of RT in the presence of a vigorous patient driver not related to a reflex or entrainment phenomenon has not been sufficiently elucidated so far, except for the description of the term of DTV [5], where a phenomenon equivalent to RT is not related to a reflex mechanism.
This type of double driver (one from the ventilator that precedes, the second one from the patient) that does not induce DT can be as frequent and potentially dangerous as the one that does induce it.
In all the previous studies they refer to patients ventilated in ACV mode with both volume control and pressure control. We also provide for the first time, to our knowledge, cases with pressure support ventilation (PSV), associated with auto-triggering, which resemble Pressure Control Ventilation (Figures 3-4).
Our opinion is that in the first three cases the double trigger and the Reverse are due to the simple mismatch of two different drivers, the neural being powerful enough to generate an increase in Vt. To demonstrate true RT we propose to cancel the mechanical cycle of the ventilator using an occlusion maneuver or to change to a brief cycle of interleaved CPAP, which would reveal whether or not the patient’s impulse is related to the mechanical cycle-, as shown in (Figure 4).
We would prefer to call these asynchronies as Reverse Trigger in the broad sense or Dual DTV-type driver, where the mechanical cycle precedes the neural. In our experience, the most frequent double trigger in ACV mode is DTV and the DTA in support ventilation. We have rarely encountered classic DTP [8].
Conflicts of Interest
The authors declare that have no conflicts of interest.
Author Contributions
BL JA, conducted and designed the study. SS JM collected and analyzed the patient data. Both authors have interpreted all data, written, and revised the manuscript.
Ethics Approval and Consent to Participate
Study approved by the Research Ethics Board, University Hospital Reina Sofía of Cordoba, Spain (RT-PSV 2017). Written Informed consent for publication was obtained from the patient’s family.
Acknowledgments
We thank Almudena Serrano Benítez, for their valuable assistance in reviewing the manuscript.
References
2. Pohlman MC, McCallister KE, Schweickert WD, Pohlman AS, Nigos CP, Krishnan JA, et al. Excessive tidal volume from breath stacking during lung-protective ventilation for acute lung injury. Critical Care Medicine. 2008 Nov 1;36(11):3019-23.
3. Beitler JR, Sands SA, Loring SH, Owens RL, Malhotra A, Spragg RG, et al. Quantifying unintended exposure to high tidal volumes from breath stacking dyssynchrony in ARDS: the BREATHE criteria. Intensive Care Medicine. 2016 Sep 1;42(9):1427-36.
4. Brochard L, Slutsky A, Pesenti A. Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. American Journal of Respiratory and Critical Care Medicine. 2017 Feb 15;195(4):438-42.
5. Chang-Wen C, Wei-Chieh L, Hsu CH. Pseudo-doubletriggering.Intensive Care Medicine. 2007 Apr 1;33(4):742.
6. Akoumianaki E, Lyazidi A, Rey N, Matamis D, Perez-Martinez N, Giraud R, et al. Mechanical ventilationinduced reverse-triggered breaths: a frequently unrecognized form of neuromechanical coupling. Chest. 2013 Apr 1;143(4):927-38.
7. Liao KM, Ou CY, Chen CW. Classifying different types of double triggering based on airway pressure and flow deflection in mechanically ventilated patients. Respiratory Care. 2011 Apr 1;56(4):460-6.
8. Lozano JB, de la Fuente Martos C, Simón JS. Double Trigger and Pseudo-reverse-trigger?. Medicina Intensiva. S0210-5691.
