Abstract
Background: Subretinal drusenoid deposits (SDDs), a hallmark of age-related macular degeneration (AMD), have been linked to cardiovascular disease, but the underlying mechanism remains unclear. We investigated the association between SDDs and severe aortic stenosis (AS).
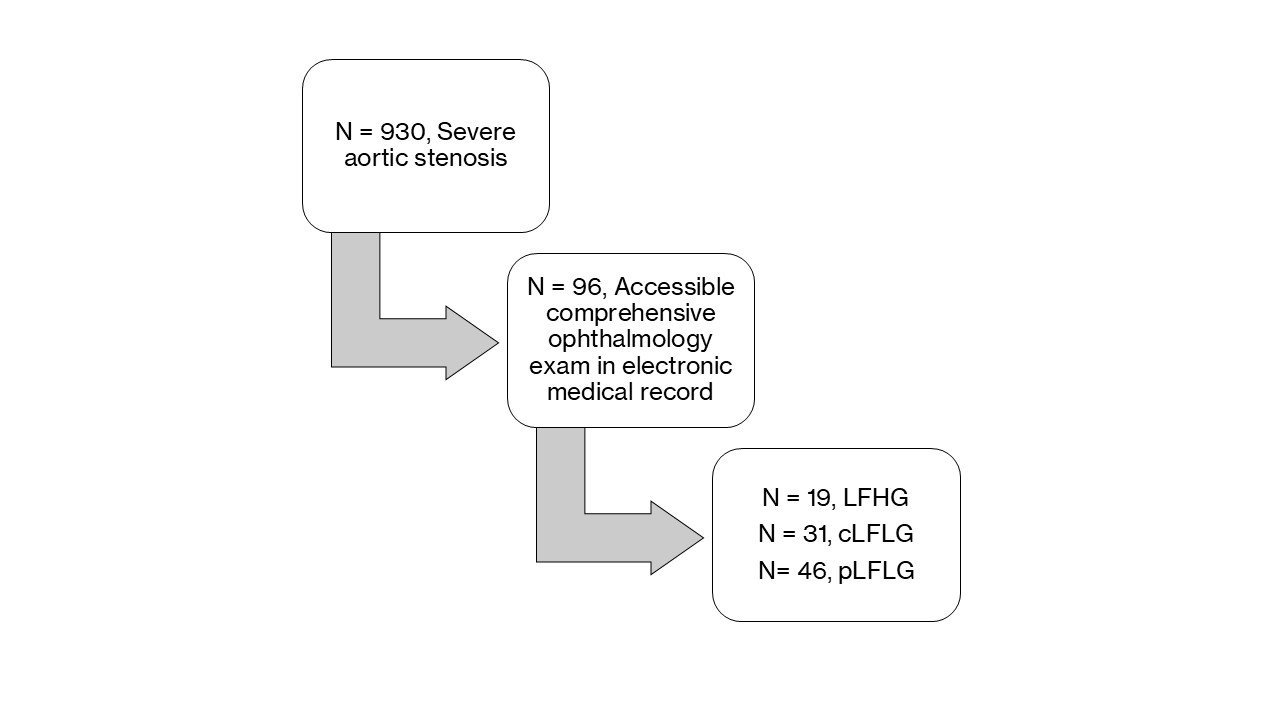
Methods: A total of 96 patients with low-flow (LF) severe AS underwent ophthalmologic evaluation to determine SDD status. All patients underwent transthoracic echocardiography and were classified into classical low-flow low-gradient (cLFLG), paradoxical low-flow low-gradient (pLFLG), or high-gradient low-flow (HGLF) AS. Echocardiographic measurements included peak aortic jet velocity (Vmax), mean aortic gradient (MG), cardiac index (CI), and Society of Thoracic Surgeons (STS) score. Group comparisons were performed using ANOVA, t-tests, and Fisher’s exact test.
Results: Among 96 patients with severe AS who underwent comprehensive ophthalmologic evaluation, SDDs were present in 54 (56%). Compared with patients without SDD, those with SDD were older (P=0.04), had a higher prevalence of AMD (P<0.001), and had higher STS scores (P=0.04). Patients with SDD demonstrated lower peak aortic velocity, peak and mean transvalvular gradients, and cardiac index (all P≤0.04). The distribution of AS hemodynamic subtypes did not differ between groups.
Conclusions: In patients with severe AS, SDDs were associated with lower transvalvular flow and gradient indices, supporting low-flow physiology as a potential mechanism underlying SDD development.
Keywords
Subretinal drusenoid deposit, Aortic stenosis, Transcatheter aortic valve replacement, Age related macular degeneration, Retina, Choroid
Introduction
Subretinal drusenoid deposits (SDD), also known as reticular pseudodrusen, have garnered increasing attention for their potential associations with systemic cardiovascular disease, including coronary artery disease, heart failure with reduced ejection fraction, atrial fibrillation, and carotid artery stenosis [1–4]. SDDs are extracellular lipid-rich accumulations located above the retinal pigment epithelium (RPE) and beneath the photoreceptor layer [5]. In contrast to soft drusen, which accumulate beneath the RPE, SDDs are increasingly recognized as a distinct morphological and pathogenic entity within the spectrum of age-related macular degeneration (AMD) [5,6]. The reported prevalence of SDD in AMD varies widely, ranging from 9–70%, depending on the stage and phenotype of the disease, and SDD have also been observed in 6–23% of individuals without AMD [2,7–9]. SDD are associated with a higher risk of progression to advanced AMD, particularly geographic atrophy, and correlate with greater photoreceptor disruption, impaired dark adaptation, and poorer visual outcomes compared to eyes with soft drusen alone [10,11]. SDD and drusen have been considered to be markers for two distinct diseases within AMD and SDD-associated AMD has been specifically associated with cardiovascular disease [12].
The proposed mechanism linking SDD to cardiovascular disease centers on the choroid, which is the vascular layer of the eye that supplies the photoreceptors and outer retina. The proposed mechanism is choroidal insufficiency secondary to reduced ocular perfusion consequent to cardiovascular disease. Reduced systemic perfusion may thus compromise the oxygen supply and metabolic support of the outer retina, leading to damage to photoreceptors that accumulate as SDDs. Because the choroid is one of the most densely perfused vascular beds in the human body, it may serve as a sensitive indicator of systemic vascular health, particularly in individuals with advanced cardiovascular disease. Severe aortic stenosis (AS) is among the most clinically significant valvular heart diseases [13]. Severe AS results in reduced cardiac output, systemic hypoperfusion, and subsequent end-organ dysfunction [14]. Considering the outer retina’s high metabolic demand and reliance on choroidal perfusion, patients with severe AS may be particularly susceptible to microvascular compromise that could manifest as SDD. We sought to investigate whether SDDs may serve as a marker of chronic systemic hypoperfusion, and in particular, if their presence may help identify decreased cardiac performance in patients with severe AS.
Methods
This single-center, retrospective study included all adult patients (≥18 years) who underwent evaluation for transcatheter aortic valve replacement (TAVR) at a large quaternary-care hospital in New York City between January 2019 and December 2024. The study was approved by the Institutional Review Board of the Icahn School of Medicine at Mount Sinai, with informed consent waived due to its retrospective design. Baseline demographic, clinical, and echocardiographic data were obtained through a comprehensive review of the electronic medical records. Patients with severe low-flow aortic stenosis, defined echocardiographically as an aortic valve area ≤1.0 cm² and a stroke volume index ≤35 mL/m², were included in the analysis. Patients with low-flow AS were stratified into three cohorts: LFHG (mean pressure gradient [MPG] ≥40 mmHg), pLFLG (MPG <40 mmHg and left ventricular ejection fraction [LVEF] ≥50%), and cLFLG (MPG <40 mmHg and LVEF <50%). All echocardiograms were interpreted by an expert structural echocardiographer (SL), with all measurements performed blinded to participants’ clinical data. The start of observation was defined as the date of the echocardiogram at the time of intervention, and follow-up ended at either the date of last contact or death.
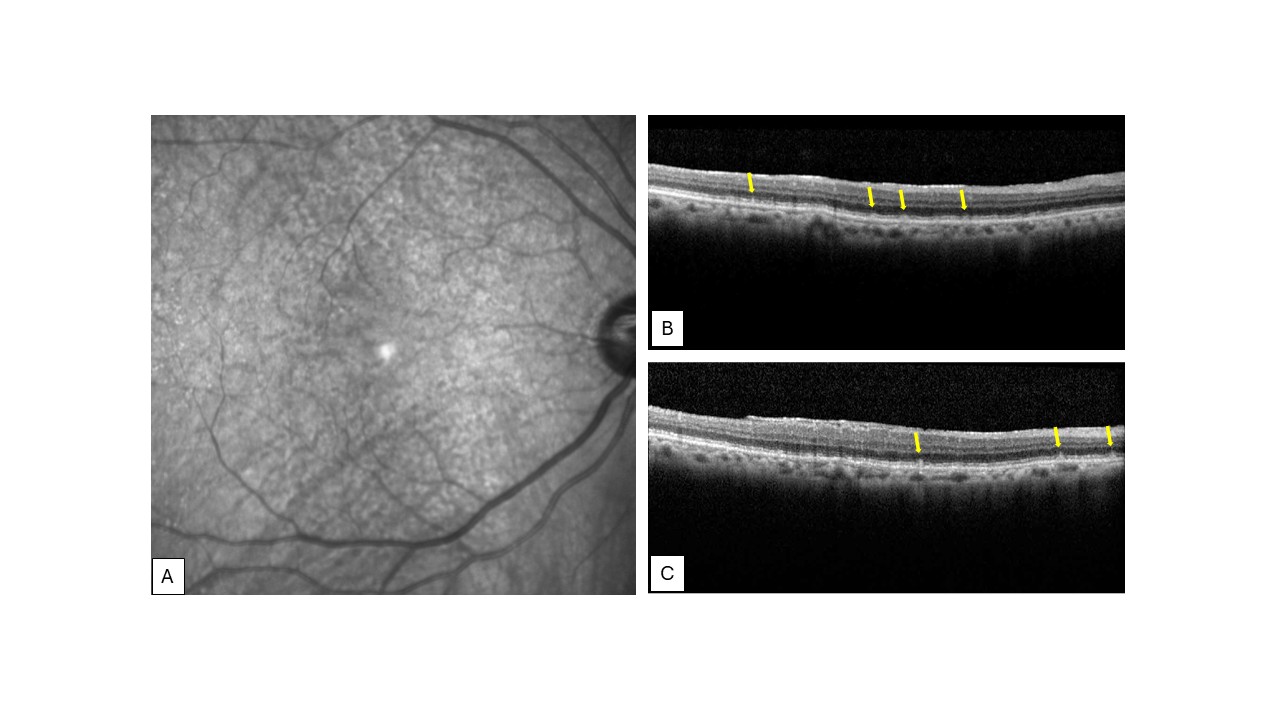
Ophthalmology examinations, including a dilated fundus exam, were found through retrospective chart review in 96 patients from the aortic stenosis original cohort (Figure 1). Baseline characteristics were collected, and patients were subsequently classified into subretinal drusenoid deposit (SDD) and non-SDD, groups through a consensus review process. This classification was based on chart review including clinical documentation and/or multimodal imaging including spectral domain optical coherence tomography (OCT) via commercially available systems in inpatient and outpatient clinics (Figure 2). When applicable image analysis for SDD were completed following a published protocol [15]. All imaging and documentation were independently recorded by treating physicians, with data extracted retrospectively from the electronic medical records. The study was approved by the Institutional Review Board at the Icahn School of Medicine at Mount Sinai. IRB approval no. 24-01456, which adhered to the tenets of the Declaration of Helsinki. Informed consent was waived because of the retrospective nature of the study.
Continuous variables were expressed as mean and standard deviation. Categorical variables were presented as numbers and percentages. Comparisons of baseline and echocardiographic characteristics were assessed using analysis of covariance (ANCOVA), Pearson’s Chi-square statistics, and Fisher’s exact test, followed by post-hoc analysis of between-group comparisons. Two-tailed P<0.05 was considered statistically significant. P values were adjusted for multiple comparisons within families of related outcomes with Holm-Bonferroni correction. Statistical significance was defined as a Holm–Bonferroni–adjusted p value <0.05. All statistical calculations and analyses were performed with SPSS, version 29.0.2.0 (IBM, Chicago, IL, USA) and STATA, version 17 (StataCorp, College Station, Texas).
Results
Ninety-six patients within the severe AS cohort had documented ophthalmology examinations with a dilated fundus exam and/or ocular imaging. Of these, 54 had SDDs present (56%). Baseline clinical variables are found in Table 1. Compared with patients without SDD, those with SDD were older (82 ± 8 vs 78 ± 10 years; P=0.04) and more frequently White (75.9% vs 57.1%; P=0.03). Clinically, patients with SDD had more frequent AMD (59% vs 19%; P<0.001) and higher STS scores (4.15 ± 3.5 vs 2.8 ± 2.4; P=0.04).
|
|
SDD - (n=42) |
SDD + (n=54) |
P value |
|
Age, years |
78±10 |
82±8 |
0.04 |
|
Female |
15 (35.7) |
22 (40.7) |
0.62 |
|
Race and ethnicity White Black Asian Other |
24 (57.1) 10 (23.8) 3 (7.1) 5 (11.9) |
41 (75.9) 3 (5.6) 1 (1.9) 9 (16.7) |
0.03 |
|
AMD |
8 (19.0) |
32 (59.3) |
< 0.001 |
|
STS score % |
2.8±2.4 |
4.15±3.5 |
0.04 |
|
Coronary Artery Disease |
25 (59.5) |
35 (64.8) |
0.77 |
|
Myocardial Infarction |
8 (19.0) |
12 (22.2) |
0.70 |
|
Congestive Heart Failure |
34 (81.0) |
43 (79.6) |
0.87 |
|
Hypertension |
36 (85.7) |
47 (87.0) |
0.85 |
|
Hyperlipidemia |
37 (88.1) |
47 (87.0) |
0.88 |
|
Diabetes Mellitus |
27 (64.3) |
25 (46.3) |
0.08 |
|
Stroke |
7 (16.7) |
16 (29.6) |
0.14 |
|
PAD |
7 (16.7) |
12 (22.2) |
0.47 |
|
Atrial fibrillation |
10 (23.8) |
22 (40.7) |
0.08 |
|
COPD |
10 (23.8) |
15 (27.8) |
0.66 |
|
Chronic Kidney Disease |
16 (38.1) |
21 (38.9) |
0.94 |
|
Liver Disease |
1 (2.4) |
5 (9.3) |
0.17 |
|
Malignancy |
14 (33.3) |
13 (24.1) |
0.32 |
|
Anemia |
23 (54.8) |
31 (57.4) |
0.80 |
|
Current Smoker |
0 (0) |
2 (3.7) |
0.21 |
|
BNP, pg/mL |
603.0±1457.7 |
949.7±2211.3 |
0.41 |
|
Serum creatinine, mg/dL |
1.3±1.0 |
1.4±1.6 |
0.68 |
|
Antiplatelet agents |
30 (71.4) |
37 (68.5) |
0.76 |
|
Anticoagulation |
9 (21.4) |
18 (33.3) |
0.20 |
|
Beta-blockers |
24 (57.1) |
28 (51.9) |
0.61 |
|
CCB |
14 (33.3) |
15 (27.8) |
0.56 |
|
ACE-I/ARBs |
19 (45.2) |
25 (46.3) |
0.92 |
|
ARNI |
4 (9.5) |
4 (7.4) |
0.72 |
|
SGLT-2i |
4 (9.5) |
3 (5.6) |
0.70 |
|
Loop diuretics |
16 (38.1) |
16 (29.6) |
0.38 |
|
MRA |
1 (2.4) |
3 (5.6) |
0.63 |
|
Values are presented as mean±standard deviation or n (%). SDD: Subretinal Drusenoid Deposit. AMD: Age-related Macular Degeneration. PAD: Peripheral Artery Disease. COPD: Chronic Obstructive Pulmonary Disease. STS: Society of Thoracic Surgeons. BNP: Brain Natriuretic Peptide. CCB: Calcium Channel Blocker. ACE-I: Angiotensin-Converting Enzyme Inhibitors. ARBs: Angiotensin Receptor Blockers. ARNI: Angiotensin Receptor/Neprilysin Inhibitor. SGLT-2I: Sodium-Glucose Cotransporter-2 Inhibitors. MRA: Mineralocorticoid Receptor Antagonist. |
|||
Transthoracic echocardiographic measures stratified by SDD status are summarized in Table 2. Patients with SDD presented lower peak aortic velocity (Vmax) (3.3±0.6 vs. 3.7±0.6, P = 0.003), peak pressure gradient (PPG) (45.6±17.4 vs. 56.7±19.1, P=0.004), mean pressure gradient (MPG) (26.5±10.7 vs. 33.4±13.2, P = 0.006) and lower cardiac index (1.93 vs. 2.13 L/min/m²; P=0.04) than those without SDD.
|
|
SDD - (n=42) |
SDD + (n=54) |
P value |
|
Aortic Stenosis Subtype LFHG cLFLG pLFLG |
11 (26.2) 16 (38.1) 15 (35.7) |
8 (14.8) 15 (27.8) 31 (57.4) |
0.10 |
|
LVESD, mm/m² |
3.5±0.9 |
3.3±1.1 |
0.47 |
|
LVEF, % |
53.8±13.1 |
54.4±12.5 |
0.84 |
|
IVS, cm |
1.3±0.4 |
1.2±0.2 |
0.05 |
|
LVPWT, cm |
1.2±0.3 |
1.2±0.2 |
0.23 |
|
LA, mm |
4.2±1.5 |
4.2±0.9 |
0.94 |
|
E/A, cm/s |
1.0±0.5 |
1.2±1.4 |
0.55 |
|
E/E’ |
16.4±6.6 |
16.2±7.9 |
0.91 |
|
LV mass index, g/cm2 |
100.4±35.5 |
94.0±28.1 |
0.35 |
|
≥ Moderate MR, % |
1 (2.4) |
0 (0) |
0.55 |
|
≥ Moderate TR, % |
3 (7.1) |
7 (13.0) |
0.70 |
|
≥ Moderate AR, % |
4 (9.5) |
10 (18.5) |
0.63 |
|
RVSP, mmHg |
40.2±13.6 |
36.9±15.1 |
0.38 |
|
S’ wave, cm/s |
11.9±2.0 |
11.0±2.9 |
0.53 |
|
AVAi, cm2 |
0.40±0.1 |
0.40±0.1 |
0.18 |
|
SVi, mL |
27.5±4.9 |
27.4±6.2 |
0.90 |
|
Vmax, m/s |
3.7±0.6 |
3.3±0.6 |
0.003 |
|
PPG, mmHg |
56.7±19.1 |
45.6±17.4 |
0.004 |
|
MPG, mmHg |
33.4±13.2 |
26.5±10.7 |
0.006 |
|
Cardiac Index L/min/m² |
2.14 |
1.93 |
0.039 |
|
Aortic valve calcium score Low Moderate High |
6 (14.3) 11 (26.2) 18 (42.9) |
9 (16.7) 17 (31.5) 20 (37.0) |
0.78 |
|
Values are presented as mean±standard deviation or n (%). LFHG: Low Flow High Gradient; cLFLG: Classic Low Flow Low Gradient; pLFLG: Paradoxical Low Flow Low Gradient; LVEDVi: Left Ventricular End-Diastolic Volume Index; LVESVi: Left Ventricular End-Systolic Volume Index; LVEF: Left Ventricular Ejection Fraction; IVS: Interventricular Septum Thickness; LVPWT: Left ventricular posterior wall thickness; LA: Left Atrial Diameter; MR: Mitral Regurgitation; TR: Tricuspid Regurgitation; AR: Aortic Regurgitation; RVSP: Right Ventricular Systolic Pressure; AVAi: Aortic Valve Area Index; SVi: Stroke Volume Index; Vmax: Peak Aortic Velocity; PPG: Peak Pressure Gradient; MPG: Mean Pressure Gradient; CI: Cardiac Index. |
|||
Discussion
In this retrospective study of patients with severe AS, we observed a significant association between SDDs and lower indices of transvalvular flow and gradient. Patients with SDD demonstrated lower peak aortic velocity, mean gradient, and cardiac index compared with those without SDD. These findings support the hypothesis that SDDs may reflect systemic low-flow physiology rather than representing an isolated ocular process, and suggest a potential link between impaired hemodynamics and SDD development. This is the first study to evaluate the presence and significance of SDDs in patients with known severe AS requiring TAVR evaluation, identifying SDDs as potential ocular markers of systemic hypoperfusion.
Our results align with and extend prior studies linking SDDs to systemic cardiovascular disease. Ledesma-Gil et al. demonstrated that AMD patients with SDDs had a significantly higher prevalence of high-risk vascular disease (HRVD), including valvular heart disease, myocardial infarction, and cerebrovascular events, with an adjusted odds ratio of 9.6 [1]. Their work established that SDDs are systemic markers of vascular compromise. Our findings advance this by showing that within a homogeneous severe AS cohort, SDDs track quantitatively with echocardiographic markers of reduced systemic perfusion, particularly in cLFLG AS. Fei et al. recently reported that SDDs in intermediate AMD were associated with reduced cardiac index and coexisting valvular heart disease, especially mitral and aortic regurgitation, as well as precursors such as aortic sclerosis and mitral annular calcification [16]. Notably, no strong association with stenotic valvular disease was found, perhaps due to the low numbers of such cases in the study. Our study of severe AS patients demonstrates that when stenosis is advanced enough to impair systemic flow, SDDs correlate with significantly lower gradients, velocities, and cardiac index. The most pronounced hemodynamic differences were confined to the cLFLG group. Together, these results suggest that SDDs may reflect a shared final pathway of systemic and ocular hypoperfusion, irrespective of whether the initiating lesion is regurgitant or stenotic valvular pathology. Lishinsky-Fischer et al. analyzed 597 AMD patients and found that those with SDDs had significantly more cardiovascular diagnoses and procedures, including percutaneous coronary intervention (PTCA), with adjusted odds ratios of around 2.7, further supporting SDDs as a sign of systemic vascular deficiency [2].
Mordechaev et al. demonstrated an association between ipsilateral internal carotid artery stenosis and SDDs, which supports downstream ophthalmic artery and choroidal hypoperfusion as a mechanism for SDD formation. SDD, thus, may serve as a sensitive biomarker for diseases relating to decreased cardiovascular flow through an ocular hypoperfusion mechanism [3]. Other studies have linked impaired cardiac function to impairment of the superficial retinal vasculature, without studying the deeper choroidal vasculature, which is more difficult. The EYE-MI pilot study linked impaired left ventricular ejection fraction to reduced superficial retinal capillary plexus density on OCTA in patients with acute coronary syndrome [17]. Patients with myocardial disease exhibit OCTA alterations in retinal microvasculature, correlating with cardiac dysfunction [18]. SDD exhibit impaired choriocapillaris perfusion on OCT angiography (OCTA) associated with outer retinal disruption [19]. Regarding choroidal perfusion, SDD are also independently associated with attenuation of all choroidal parameters including choroidal thinning and reduced vascularity, suggesting local vascular insufficiency [20]. These studies indicate that systemic hypoperfusion is reflected in SDD even in eyes with little to no signs of AMD consistent with our findings that SDDs are linked to reduced cardiac output in severe AS.
The presence of SDDs in patients with severe AS may serve as a non-invasive biomarker of systemic hypoperfusion. This could be particularly useful in the cLFLG subtype, a group with notoriously poor prognosis and challenging diagnostic criteria. Ophthalmic imaging for SDDs could complement echocardiographic and hemodynamic assessments in identifying patients at highest risk, providing additional insight into disease severity beyond conventional cardiac imaging.
This study has several strengths. It represents the largest cohort of severe AS patients with ophthalmic data to date and the first to stratify associations by AS hemodynamic subtype. Comprehensive echocardiographic evaluations were performed and interpreted by an expert structural echocardiographer, ensuring reliable cardiac measurements. Adjustments for demographics such as age, gender, and race further strengthen the validity of our findings. By focusing on a homogeneous, high-risk cardiovascular population, this study helps bridge cardiology and ophthalmology, contributing new evidence that retinal imaging may hold systemic diagnostic value.
Several limitations should be acknowledged. The retrospective, single-center design, and reliance on available ophthalmic exams introduces potential selection bias. SDD identification was based on chart review and available multimodal imaging, introducing some heterogeneity in classification. The cross-sectional nature of the study does not allow inference of causality. The small sample size may lead to bias and subgroup analyses were underpowered, particularly in the pLFLG and HFLG groups. While an elevated hazard ratio for mortality was observed among SDD patients, the study was underpowered to establish prognostic significance.
Future research should include prospective multicenter studies with larger sample sizes, different types of aortic stenosis, and standardized multimodal retinal imaging and echocardiography. Longitudinal assessment of SDDs before and after aortic valve replacement could clarify whether SDDs regress with improved systemic perfusion. Incorporation of advanced ocular perfusion metrics such as OCTA, choroidal thickness, and choroidal vascularity index would allow better understanding of the underlying mechanisms connecting the systemic and retinal vasculature. In conclusion, our findings demonstrate that SDDs are closely associated with severe AS, particularly in the cLFLG subtype. These results support the hypoperfusion hypothesis of SDD pathogenesis and suggest that SDDs may serve as a valuable non-invasive biomarker of systemic cardiovascular disease, with potential applications in risk stratification and interdisciplinary management of patients with advanced valvular heart disease.
Author Contribution Statement
C.M., L. W., S.L., and R.T.S contributed to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
References
2. Lishinsky-Fischer N, Misgav K, Chowers I, Tiosano L, Shwartz Y, Levy J. Association of subretinal drusenoid deposits and cardiovascular disease. Sci Rep. 2024 Oct 26;14(1):25569.
3. Mordechaev E, Jo JJ, Mordechaev S, Govindaiah A, Fei Y, Tai K, et al. Internal Carotid Artery Stenosis and Ipsilateral Subretinal Drusenoid Deposits. Invest Ophthalmol Vis Sci. 2024 Feb 1;65(2):37.
4. Smith RT, Olsen TW, Chong V, Kim J, Hammer M, Lema G, et al. Subretinal drusenoid deposits, age-related macular degeneration, and cardiovascular disease. Asia Pac J Ophthalmol (Phila). 2024 Jan-Feb;13(1):100036.
5. Zweifel SA, Spaide RF, Curcio CA, Malek G, Imamura Y. Reticular pseudodrusen are subretinal drusenoid deposits. Ophthalmology. 2010 Feb;117(2):303–12.e1.
6. Spaide RF, Ooto S, Curcio CA. Subretinal drusenoid deposits AKA pseudodrusen. Surv Ophthalmol. 2018 Nov-Dec;63(6):782–815.
7. Zweifel SA, Imamura Y, Spaide TC, Fujiwara T, Spaide RF. Prevalence and significance of subretinal drusenoid deposits (reticular pseudodrusen) in age-related macular degeneration. Ophthalmology. 2010 Sep;117(9):1775–81.
8. Zarubina AV, Neely DC, Clark ME, Huisingh CE, Samuels BC, Zhang Y, et al. Prevalence of Subretinal Drusenoid Deposits in Older Persons with and without Age-Related Macular Degeneration, by Multimodal Imaging. Ophthalmology. 2016 May;123(5):1090–100.
9. Domalpally A, Agrón E, Pak JW, Keenan TD, Ferris FL 3rd, Clemons TE, et al. Prevalence, Risk, and Genetic Association of Reticular Pseudodrusen in Age-related Macular Degeneration: Age-Related Eye Disease Study 2 Report 21. Ophthalmology. 2019 Dec;126(12):1659–66.
10. Marsiglia M, Boddu S, Bearelly S, Xu L, Breaux BE Jr, Freund KB, et al. Association between geographic atrophy progression and reticular pseudodrusen in eyes with dry age-related macular degeneration. Invest Ophthalmol Vis Sci. 2013 Nov 8;54(12):7362–9.
11. Reiter GS, Told R, Schranz M, Baumann L, Mylonas G, Sacu S, et al. Subretinal Drusenoid Deposits and Photoreceptor Loss Detecting Global and Local Progression of Geographic Atrophy by SD-OCT Imaging. Invest Ophthalmol Vis Sci. 2020 Jun 3;61(6):11.
12. Thomson RJ, Chazaro J, Otero-Marquez O, Ledesma-Gil G, Tong Y, Coughlin AC, et al. SUBRETINAL DRUSENOID DEPOSITS AND SOFT DRUSEN: Are They Markers for Distinct Retinal Diseases? Retina. 2022 Jul 1;42(7):1311–8.
13. Grigorios T, Stefanos D, Athanasios M, Ioanna K, Stylianos A, Periklis D, et al. Transcatheter versus surgical aortic valve replacement in severe, symptomatic aortic stenosis. J Geriatr Cardiol. 2018 Jan;15(1):76–85.
14. Harjola VP, Mullens W, Banaszewski M, Bauersachs J, Brunner-La Rocca HP, Chioncel O, et al. Organ dysfunction, injury and failure in acute heart failure: from pathophysiology to diagnosis and management. A review on behalf of the Acute Heart Failure Committee of the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). Eur J Heart Fail. 2017 Jul;19(7):821–36.
15. Cymerman RM, Skolnick AH, Cole WJ, Nabati C, Curcio CA, Smith RT. Coronary Artery Disease and Reticular Macular Disease, a Subphenotype of Early Age-Related Macular Degeneration. Curr Eye Res. 2016 Nov;41(11):1482–8.
16. Fei Y, Jo JJ, Chen S, Ledesma-Gil G, Otero-Marquez O, Mordechaev E, et al. Quantifying cardiac dysfunction and valvular heart disease associated with subretinal drusenoid deposits in age-related macular degeneration. Eur J Ophthalmol. 2024 Nov;34(6):2038–44.
17. Arnould L, Guenancia C, Azemar A, Alan G, Pitois S, Bichat F, et al. The EYE-MI Pilot Study: A Prospective Acute Coronary Syndrome Cohort Evaluated With Retinal Optical Coherence Tomography Angiography. Invest Ophthalmol Vis Sci. 2018 Aug 1;59(10):4299–306.
18. Wu JY, Hu JY, Ge QM, Xu SH, Zou J, Kang M, et al. Ocular microvascular alteration in patients with myocardial infarction-a new OCTA study. Sci Rep. 2024 Feb 24;14(1):4552.
19. Abdolrahimzadeh S, Zweifel SA, Di Pippo M, Bajka A, Scuderi G, Lotery AJ. Central macular choriocapillaris impairment as a manifestation of microvascular disease in eyes with subretinal drusenoid deposits. Eye (Lond). 2024 Jan;38(1):173–8.
20. Keenan TD, Klein B, Agrón E, Chew EY, Cukras CA, Wong WT. Choroidal thickness and vascularity vary with disease severity and subretinal drusenoid deposit presence in nonadvanced age-related macular degeneration. Retina. 2020 Apr;40(4):632–42.
