Loading
Archives of Pharmacology and Therapeutics
ISSN: 2688-9609
Featured Articles
Why Can Modulation of α6-Containing GABAA Receptors Reduce the Symptoms of Multiple Neuropsychiatric Disorders?
Werner Sieghart
α6-containing GABAA receptors (α6GABAARs) are strongly expressed in cerebellar granule cells, where they mediate a correctly timed and precise coordination of all muscle groups that execute behavior and protect the brain from information overflow. Recently, it was demonstrated that positive modulators with a high selectivity for α6GABAARs (α6-modulators) can reduce the symptoms of multiple neuropsychiatric disorders in respective animal models to an extent comparable with established clinical therapeutics.
Arch Pharmacol Ther, 2024, Volume 6, Issue 1, p1-7 | DOI: 10.33696/Pharmacol.6.047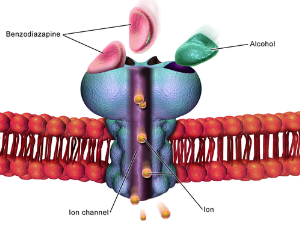
COVID-19 Disease and SARS-CoV-2 Vaccination in Patients with Cancer
Daniel A. King, Jeffrey Chi, Shreya Prasad Goyal, Muhammad Wasif Saif
Since the declaration of COVID-19 as a pandemic in March 2020, there have been more than 100 million reported cases of COVID-19 worldwide and more than 2.1 million deaths. The purpose of this editorial is to review recent updates regarding COVID-19 disease and SARS-CoV-2 vaccination in cancer patients.
Arch Pharmacol Ther, 2021, Volume 3, Issue 1, p5-9 | DOI: https://doi.org/10.33696/Pharmacol.3.020
Pharmacogenetic Variants in the DPYD and TYMS Genes are Clinically Significant Predictors of Fluoropyrimidine Toxicity: Are We Ready for Use in our Clinical Practice
Muhammad Wasif Saif, Hilal Hachem, Sneha Purvey, Ruchi Hamal, Lulu Zhang, Nauman Saleem Siddiqui, Amandeep Godara, Robert B. Diasio
Fluoropyrimidines have been extensively used for almost 6 decades to treat a variety of solid cancers, especially colon, gastric, anal, rectal, head & neck and breast. However, 31–34% of patients encountered grade 3–4 adverse events (AEs) with 0.5% mortality oftennecessitating dose reduction or discontinuation. A significant proportion of these AEs are likely to be the result of inter-individual genetic variation, in particularly such as dihydropyrimidine dehydrogenase (DPYD). DPYD gene encodes DPD, the rate-limiting enzyme responsible for catabolism of 5-FU and is responsible for >85% of 5-FU elimination.
Arch Pharmacol Ther, 2020, Volume 2, Issue 1, p6-8 | DOI: 10.33696/Pharmacol.2.012
Pharmacologic Therapies for Non-Muscle Invasive Bladder Cancer: Current and Future Treatments
Ilana P. Goldberg, Benjamin Lichtbroun, Eric A. Singer, Saum Ghodoussipour
Bladder cancer is the sixth most common malignancy in the United States and 70% of cases are non-muscle invasive at the time of diagnosis. Effective treatment is crucial to prevent progression, which occurs in about 30% of patients. The American Urological Association (AUA) guidelines recommend treatment of non-muscle invasive bladder cancer (NMIBC) with intravesical Bacille Calmette-Guerin (BCG) and chemotherapy.
Arch Pharmacol Ther, 2022, Volume 4, Issue 1, p13-22 | DOI: 10.33696/Pharmacol.4.030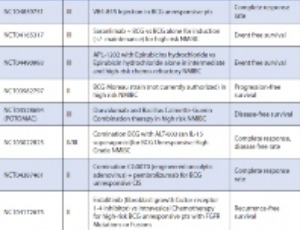
Relative COVID-19 Vaccine Booster Effectiveness and ClinicalEpidemiological Characteristics Before and After 29 Days of Shot
Jose Luis Turabian
Background: When the highest vaccine COVID-19 booster effectiveness (VBE) is obtained is not clearly known. Objective: To compare the cases of COVID-19 in booster vaccinated people with a time of <29 days vs. ≥ 29 days from booster to infection diagnosis and assess their relative VBE. Methodology: An observational, longitudinal and prospective case series study of adult patients with COVID-19 breakthrough infections in booster vaccinated people, in general medicine and for the period December 2021 to February 2022, during the omicron variant contagion wave.
Arch Pharmacol Ther, 2022, Volume 4, Issue 1, p23-34 | DOI: 10.33696/Pharmacol.4.031
ProLung™-budesonide Inhibits SARS-CoV-2 Replication and Reduces Lung Inflammation
Kameswari S. Konduri, Ram Pattisapu, Jogi Pattisapu, Girija G. Konduri, John Zwetchkenbaum, Bidhan Roy, Monalisa Barman, Adria Frazier, Brett L. Hurst, Nejat Duzgunes
Inhaled budesonide benefits patients with COVID-19. ProLung™-budesonide enables the sustained, low dose administration of budesonide within a delivery vehicle similar to lung surfactant. ProLung™-budesonide may offer anti-inflammatory and protective effects to the lung in COVID-19, yet it’s effect on SARS-CoV-2 replication is unknown.
Arch Pharmacol Ther, 2021, Volume 3, Issue 2, p52-65 | DOI: 10.33696/Pharmacol.3.028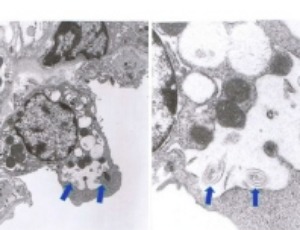
About Scientific Archives
Scientific Archives is a global publisher initiated with the mission of ensuring equal opportunity for accessing science to research community all over the world. Spreading research findings with great relevance to all channels without any barrier is our goal. We want to overcome the challenges of Open Access with ensured quality and transparency.
