Abstract
Cervical small cell carcinoma (SCC) is a rare disease, with a high degree of malignancy and a poor prognosis. Para-aortic lymph nodes (PALN) are frequent sites of recurrence after pelvic chemoradiotherapy. In the present study, we report the case of a 26-year-old female patient with cervical SCC who suffered for two times PALN relapses. Our research results have shown that PALN lesions can be effectively controlled with permanent radiation therapy. Prophylactic PALN radiotherapy may be an effective option for patients with cervical SCC who require radical pelvic radiation therapy.
Keywords
Small cell cervical carcinoma, Cervical neurone endocrine carcinoma, Cervix cancer radiotherapy
Introduction
Female reproductive system neuroendocrine tumors represent 2% of all female reproductive system malignancies [1]. SCC of the female reproductive system is a high-grade neuroendocrine tumor, which occurs in the cervix and accounts for 1% to 2% of all cervical cancers [2]. Cervical SCC has a high degree of malignancy, poor prognosis, and a high probability of early lymph node and distant metastasis. The clinical manifestations of cervical SCC are relatively rare, currently there is no exact diagnosis and standard treatment. The common recommendation is to apply individualized comprehensive treatment based on radical surgery, chemotherapy, radiotherapy and other treatment methods [3]. Radiotherapy plays an important role in the treatment of cervical SCC. Nowadays, it is still controversial whether pelvic radiotherapy for cervical SCC is routinely performed with para-aortic prophylactic extended field radiotherapy. This study retrospectively analyzed the clinical and pathological data of a patient with cervical SCC and combined with relevant literature analysis to deepen the understanding of cervical SCC.
Clinical Information
A 26-year-old female patient was presented with abnormal vaginal bleeding and was diagnosed with FIGO 2018 Stage IB3 SCC in March 2018. Chest and abdomen CT showed a macroscopic cervical tumor. No significant morphological abnormalities were found in the lungs, liver, pancreas, spleen, and kidney. The neuron specific- enolase (NSE) index value was 24.51 ng/mL. She had 3 courses of neoadjuvant chemotherapy (NAC) based on etoposide and platinum (EP) regimen, followed by radical hysterectomy and pelvic lymphadenectomy in May 2018. The postoperative pathological report showed: AE1/AE3(+), Vimentin (+), P40(-), CK5/6(-), Syn (+), CgA(-), CD56(+), Ki67(80?+), S100(-), CEA(-), P63(-). Cervical SCC invaded muscle layer >1/2, Endocervical canal (-); Vaginal cuff (-). Bilateral parametrium (-) (Figure 1). No pelvic lymph node metastasis was found. After two courses of adjuvant chemotherapy with the EP regimen, the pelvic prophylactic radiotherapy started in July 2018. The upper boundary of planning target volume (PTV) was located in the middle of the 4th lumbar vertebra. The prescribed dose of radiotherapy was 45Gy/25 fractions (Figure 2). Brachytherapy was given at 0.5 cm under the mucosa, 14Gy/2 fractions. An additional one cycle of adjuvant chemotherapy with EP regimen was concurrently administered with radiotherapy. After the completion of radiotherapy, the NSE index value decreased to 10.25 ng/mL in August 2018. This value was regularly monitoring.
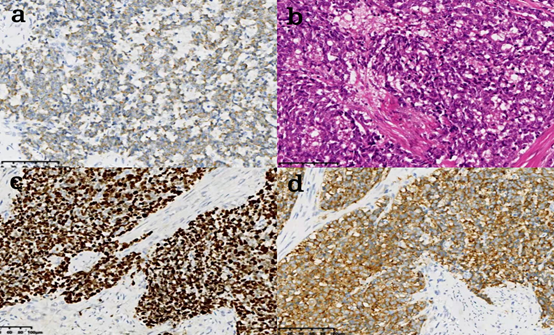
Figure 1: a. AE1/AE3 (+), b. HE20x, c. Ki67 (80%+), d. Syn (+).
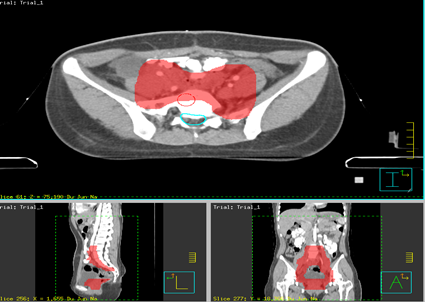
Figure 2. Simulated diagram of cumulative dose distribution of the pelvic radiotherapy. PTV range in red.
A complete re-examination workup (from chest-to-abdomen CT) was performed about 7 months after completion of radiotherapy, and revealed enlarged PALN (Figure 3a), with no obvious abnormalities. The NSE index value was 36.81 ng/mL in April 2019. The lower border of the enlarged lymph node was located in the middle of 4th lumbar vertebra, adjacent to the upper margin of pelvic radiotherapy field. Based on the new Response Evaluation Criteria in Solid Tumors (revised RECIST guideline, version 1.1) [4], we considered this newly diagnosed PALN as a progressive disease (PD). Besides, as patient did not receive previous irradiation in PALN region, the irradiation field encompassed the entire gross tumor volume (GTV: enlarged PALN), the CTV range was: - upper border: middle of the 1st lumbar vertebra (renal vein level), - lower border: middle of the 4th lumbar vertebra. The total dose delivered was 45 Gy/ 25 fractions, and GTV was boosted up to 60 Gy/ 25 fractions (Figure 4). During radiotherapy, patient developed a second-degree myelosuppression and no chemotherapy was administered. An assessment of the effectiveness by the abdominal CT one month after the completion of the radiotherapy indicated a complete remission (CR) of the lesion. The NSE index value also decreased and was 10.83 ng/mL. The patient was re-examined 3 months after the completion of PALN irradiation, the CT images showed that the primary enlarged lymph nodes had disappeared and a new enlarged lymph node appeared in the retroperitoneal region (Figures 3b and 3c) in August 2019. The NSE index value increased once again up to 27.80 ng/mL. The inferior border of the newly enlarged lymph node is located in the middle of the 1st lumbar vertebra (Figures 5a and 5b), immediately adjacent to the upper margin of the PALN radiotherapy field. The effectiveness evaluation: PD. Considering the recurrence in the radiotherapy field, the patient refused to continue radiotherapy course because of the adverse side effects of radiation therapy, and instead of radiotherapy, she was given 6 cycles of chemotherapy with EP regimen, followed by an abdominal CT review, which showed no significant change in retroperitoneal lymph node metastases (Figure 3d), and the NSE index value increased up to 26.11 ng/mL in December 2019, the efficacy evaluation indicates a stable disease (SD).
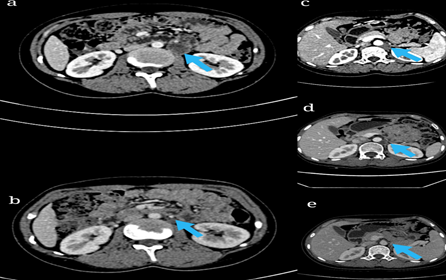
Figure 3. Imaging findings before and after treatment a: Enlarged lymph nodes adjacent to the abdominal aorta at the first relapse after pelvic field radiotherapy, b: The original enlarged lymph nodes disappeared after radiotherapy in the PALN area, c: New metastases after radiotherapy in the PALN area, d: The enlarged lymph nodes remained after 6 courses of chemotherapy. e: Local lesions decreased after radiotherapy.
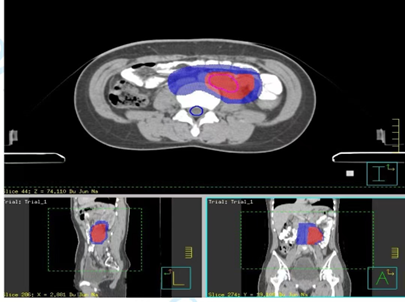
Figure 4. Simulated diagram of cumulative dose distribution of the PALN radiotherapy. CTV range in blue, GTV range in red.
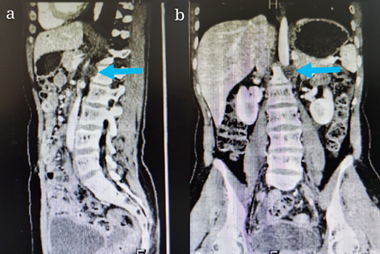
Figure 5. Imaging findings of recurrence after radiotherapy in PALN region. a: retroperitoneal enlarged lymph nodes (sagittal position), lower margin located in the middle of 1st lumbar vertebra. b: enlarged retroperitoneal lymph nodes (coronal).
Since there is no significant change in the enlarged lymph node after 6 cycles of chemotherapy, the patient received once again radiotherapy in another cancer treatment center, the irradiation field encompassed the entire gross tumor volume (GTV), and 95% of P- GTV receive 60 Gy/ 25 fractions, the enlarged lymph nodes disappeared after radiotherapy, the efficacy evaluation indicates a complete remission (CR) (Figure 3e) and the NSE index value decreased to 10.33 ng/mL in February 2020. Changes in NSE index value and treatment timeline are shown in (Figures 6 and 7).
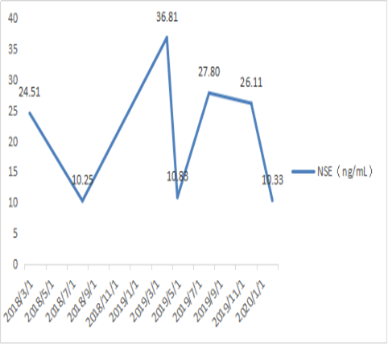
Figure 6. Changes in NSE index value.
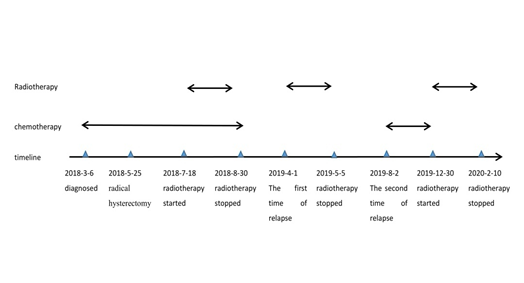
Figure 7. Timeline of different treatments.
Discussion
The cervical SCC is clinically rare, highly malignant, highly aggressive, with a poor prognosis, and is prone to early distant metastasis and local recurrence. Pelvic, paravertebral, supraclavicular lymph node metastasis are common lymph node metastasis sites [5]. Some studies showed a median overall survival of 22-24.8 months for cervical SCC [6]. It is generally accepted that most patients relapse within 1 year of diagnosis, accounting for about 80% of all relapses. In our study, the out-field failure occurred 10 months after diagnosis, which is consistent with the above findings. Chemotherapy plays an indispensable role as an adjuvant treatment option. Regarding the clinical manifestations, cervical SCC is similar to small cell lung cancer, EP schemes are often used for chemotherapy in clinical practice [7]. Study from Pei et al. [8] showed that patients who had more than 5 cycles of chemotherapy with the EP regimen were more likely to demonstrate a better survival rate; the difference was statistically significant. In our study, comparing the two relapses treatments, the first relapse reached complete remission of the lesion after a single course of radiotherapy; the second relapse did not show any reduction of the lesion after six courses of chemotherapy and disappeared after re-irradiation. The results indicate that patients with solitary recurrence in PALN would be candidates for definitive radiotherapy.
To date, there is a controversy about whether pelvic radiotherapy for cervical SCC should be performed routinely with prophylactic PALN extension field radiotherapy. According to the literature review, out of 15 patients with cervical SCC who underwent pelvic radiotherapy, 7 patients developed recurrences outside of the radiation field, including 5 patients with recurrence in the PALN region and 2 patients with vaginal recurrence [9]. Literature suggests that because of the high degree of cervical SCC malignancy, although sensitive to local and systemic treatment, most patients have a poor prognosis, there is still a high probability of local recurrence and distant metastasis, prophylactic PALN extension field radiotherapy can reduce the recurrence rate in the PALN area, and improve the overall survival rate [10]. Therefore, prophylactic PALN extension field radiotherapy can improve the prognosis of patients [10,11]. In the present case, the patient underwent two times of PALN relapses, no recurrence was found within the radiation field, and radiotherapy can effectively control the PALN recurrence. It is worth investigating whether PALN recurrence can be avoided when prophylactic PALN radiotherapy is given at the same time with the pelvic radiotherapy. Based on the characteristics of recurrence, we analyzed the following reasons: 1. Radiotherapy plays an important role in the treatment of cervical SCC, and the lesions in the radiation field can be effectively controlled; 2. The lymph node metastasis of cervical SCC is mainly regional, and skip lymph node metastasis is rare, and recurrences of this patient were consistent with this characteristic; 3. It is also questionable, whether metastatic tumor cells existed in the PALN area before the positive imaging results; 4. Besides, the microenvironment in the radiation field is not suitable for the growth of tumor cells, which induced the skip of tumor cells outside the radiation field.
In conclusion, Cervical SCC is highly malignant, prone to distant metastasis and local recurrence, and the abdominal para-aortic lymph node is preferential common recurrence sites. Prophylactic PALN radiotherapy may be an effective option for patients with cervical small cell carcinoma who need radical pelvic radiotherapy.
Authors’ Contributions
Hong LIU identified the patient, Hong LIU and Xi CHEN managed treatment of the patient, CHEN Xi and KEITA Mamady drafted the manuscript, Zilin Li provided radiology images, FANG Zhaohui and TRAOPE Bangaly provided pathology slides, Hong LIU revised the paper.
Data Statement
The data generated and analyzed during the current case report are not publicly available to protect patient privacy.
Competing Interests
All authors in this case report declare that they have no competing interests.
References
2. Xing D, Zheng G, Schoolmeester JK, Li Z, Pallavajjala A, Haley L, et al. Next-generation sequencing reveals recurrent somatic mutations in small cell neuroendocrine carcinoma of the uterine cervix. The American Journal of Surgical Pathology. 2018 Jun;42(6):750.
3. Satoh T, Takei Y, Treilleux I, Devouassoux-Shisheboran M, Ledermann J, Viswanathan AN, et al. Gynecologic Cancer InterGroup (GCIG) consensus review for small cell carcinoma of the cervix. International Journal of Gynecologic Cancer. 2014 Nov 1;24(Supp 3):S102-S108.
4. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). European Journal of Cancer. 2009 Jan 1;45(2):228-47.
5. Yuan L, Jiang H, Lu Y, Guo SW, Liu X. Prognostic factors of surgically treated early-stage small cell neuroendocrine carcinoma of the cervix. International Journal of Gynecologic Cancer. 2015 Sep 1;25(7): 228-47.
6. Scutiero G, Loizzi V, Macarini L, Landriscina M, Greco P. Small cell carcinoma of the uterine cervix metastasising to the cerebellum. Journal of Obstetrics and Gynaecology. 2013 Aug 1;33(6):639-41.
7. Tempfer CB, Tischoff I, Dogan A, Hilal Z, Schultheis B, Kern P, Rezniczek GA. Neuroendocrine carcinoma of the cervix: a systematic review of the literature. BMC cancer. 2018 Dec;18(1):530.
8. Pei X, Xiang L, Ye S, He T, Cheng Y, Yang W, et al. Cycles of cisplatin and etoposide affect treatment outcomes in patients with FIGO stage I-II small cell neuroendocrine carcinoma of the cervix. Gynecologic Oncology. 2017 Dec 1;147(3):589-96.
9. Viswanathan AN, Deavers MT, Jhingran A, Ramirez PT, Levenback C, Eifel PJ. Small cell neuroendocrine carcinoma of the cervix: outcome and patterns of recurrence. Gynecologic Oncology. 2004 Apr 1;93(1):27-33.
10. Hoskins PJ, Swenerton KD, Pike JA, Lim P, Aquino-Parsons C, Wong F, Lee N. Small-cell carcinoma of the cervix: fourteen years of experience at a single institution using a combined-modality regimen of involved-field irradiation and platinum-based combination chemotherapy. Journal of Clinical Oncology. 2003 Sep 15;21(18):3495-501.
11. Lee J, Lin JB, Chang CL, Jan YT, Sun FJ, Wu MH, et al. Prophylactic lower para-aortic irradiation using intensity-modulated radiotherapy mitigates the risk of para-aortic recurrence in locally advanced cervical cancer: A 10-year institutional experience. Gynecologic Oncology. 2017 Jul 1;146(1):20-6.
