Abstract
Traumatic brain injury (TBI) leads to motor deficits and impaired balance and gait, significantly impacting patients’ quality of life. Repetitive transcranial magnetic stimulation (rTMS) aims to increase recovery by modulating cortical plasticity. A male patient in his late 20s presented with tetraplegia and balance impairment for four years after TBI. Initial home-based exercises yielded no significant improvement. The patient subsequently underwent rTMS and neuromotor training over 2 weeks to improve motor control and balance. The Fugl-Meyer assessment (FMA) and Berg Balance scale (BBS) demonstrated improvements post-treatment: the FMA score increased by 25.76% and 20.5% for the upper and lower limbs, respectively, and the BBS score increased by 37.5%. Post-treatment, the patient was able to walk with minimal support. The Functional Independence Measure and Functional Assessment Measure scale (FIM-FAM) scale demonstrated a 17.6% improvement, indicating enhanced Activities of Daily Living (ADLs) and increased independence. rTMS combined with neuromotor rehabilitation may enhance motor and balance outcomes in patients with chronic TBI. Further studies are warranted in terms of randomized controlled trials.
Keywords
New rehabilitative technologies, Neurorehabilitation, Trauma and rehabilitation
Introduction
Approximately 30% of patients with traumatic brain injury (TBI) have motor dysfunctions and gait and balance problems, which vary depending on the injured area [1] and significantly affect physical function, independence, and quality of life. Moreover, injury may occur following a fall or a road traffic accident (RTA), and community participation may be limited [2–5], placing significant economic strain on society [6].
Cortical excitability generally decreases after TBI in the affected primary motor cortex relative to the unaffected motor cortex. This might result from a shift in interhemispheric interactions, with increased transcallosal inhibition from the intact to the damaged motor cortex [7,8]. Repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS) are applied over the intact hemisphere to allow safe cortical stimulation in humans, promote the restoration of activity across bihemispheric neural networks, and facilitate adaptive plasticity [9,10]. rTMS is a procedure that uses magnetic fields to stimulate nerve cells in the brain in various neurological conditions, including Cerebrovascular accident, Parkinson’s disease, TBI, and cerebellar diseases.
The principle of rTMS is based on Faraday’s law of electromagnetic induction, which states that when an electric current flows within a circular coil, a magnetic field is generated perpendicular to the plane of the coil and reaches the cerebral cortex after passing through soft tissues and the skull. The magnetic field that reaches the cerebral cortex generates an eddy current that is perpendicular to this magnetic field and thus parallel to the plane of the coil (skull). The direction of the eddy current that is generated is opposite to the direction of the electric current that flows through the coil. This eddy current acts on interneurons in the cerebral cortex and ultimately affects neurons throughout the brainstem and spinal cord that descend from the cerebral cortex. Thus, evidently, rTMS results in “transcranial” magnetic stimulation, but what actually affects the neurons is the eddy current that is generated in vivo by “magnetic stimulation” [11].
rTMS generates a transient magnetic field, which in turn induces a secondary current in the brain that is capable of depolarizing neurons. Depending on the frequency, duration of the stimulation, shape of the coil, and strength of the magnetic field, rTMS can activate or suppress activity in cortical regions [12].
Some studies are available on cognitive improvement in TBI patients treated with rTMS, but very few have shown improvements in locomotor activity, balance, and coordination after rTMS rehabilitation. Therefore, we attempted to rehabilitate a man who had been suffering from TBI for the last 4 years.
In another clinical trial [13], in patients with chronic hemiparetic stroke, after receiving high-frequency (10 Hz, repeated 8 times) or sham rTMS over the primary motor cortex (M1) of the damaged hemisphere, the authors reported that rTMS induced a significantly greater increase in the MEP amplitude than sham rTMS did; this corticomotor excitability change was associated with enhanced motor skill acquisition.
A study by Pomeroy et al. [14] assessed the efficacy of low-frequency rTMS and revealed that in patients who underwent rTMS combined with neurorehab, the motor-evoked potential frequency increased by 14% for the biceps and 20% for the triceps, whereas with placebo rTMS, the motor-evoked potential frequency decreased by 12% for the biceps and 6% for the triceps.
Some negative findings have also been reported. Malcolm et al. reported no improvement with the combination of r-TMS with physical therapy [15].
rTMS should not be used in patients with metallic implants within the body, such as prostheses or pacemakers, and a history of seizures within one year, as it can decrease the seizure threshold. Some adverse effects, such as headaches, which are self-limiting, and seizures, which may require the cessation of this therapy, have been reported.
Case Presentation
A married male patient in his late 20s, the only bread earner of the family, known to have traumatic brain injury (TBI), presented to the outpatient department with difficulty standing and walking following a TBI in 2020. The patient was initially admitted to the intensive care unit of a tertiary health center following a road traffic accident (RTA). After initial management, Non-Contrast Computed Tomography (NCCT) of the brain was performed, and the patient was diagnosed with a subdural hematoma with diffuse axonal injury. He was comatose for three months and was managed conservatively by a physician through medications, a Ryle’s tube, a catheterization, and was discharged after achieving hemodynamic stability.
In August 2023, he reported with his caregiver to the Physical Medicine & Rehabilitation outpatient department. He was tetraplegic due to TBI and was assessed by the Functional Independence Measure and Functional Assessment Measure (FIM-FAM Scale) used to measure activity of daily living independence (score-113/210) and Fugl-Meyer Assessment scale (FMA) (assessment of sensorimotor function) [16] and he had difficulty maintaining balance, as assessed by the Berg Balance Scale (BBS) [17] (score: 2/56), (Figure 1). He had no past history of seizures or was on any medications for seizure prevention.

Figure 1. Patient was not able to stand even with support.
Initially, the patient was advised to perform home-based exercise, which was demonstrated to his caregiver ( passive range of motion of all 4 limbs, active range of motion of bilateral upper limbs, breathing exercise, isometric quadriceps and hamstring exercise, active ankle foot exercise, all exercises to be done three times a day with at least 10 repetitions) and a follow-up was performed after 2 months, but no improvement was observed. After that, repetitive transcranial magnetic stimulation (rTMS) was planned for this patient.
rTMS was given in both the cerebral hemispheres via a figure of 8 coil with 100% motor evoked potentials (MEP) for 10 days (5 days/week, Monday to Friday for two weeks) at the primary motor cortex (M1) over the right hemisphere (Mode-1 Hz, 10 pulses over 10 seconds, total of 1200 pulses) and left hemisphere (Mode 5Hz, 10 pulses over 2 seconds total 150 pulses) with a 10-minute pause between 2 sessions. High frequency rTMS was given on the dominant hemisphere (i.e., left) to stimulate it, and low frequency rTMS was given to the right hemisphere to inhibit its inhibitory action on the left hemisphere, so that over-activation of the left frontal and temporal cortices may reduce the recovery potentials by inhibiting (perilesional) the right frontal and temporal areas [18]. Immediately after the rTMS session, the patient performed 45 minutes of neuromotor training.
The neuromotor training was designed to promote the recovery of voluntary motor functions and improve balance.
- Motor control exercises for the trunk, strengthening of muscles of the upper and lower limbs, and crutch muscle strengthening exercises (Figure 2).
- Progressive neuromuscular facilitation with proprioceptive discrimination training.
- Training for load modulation in the standing posture and during gait, e.g., Figure 3
- Aerobic exercises
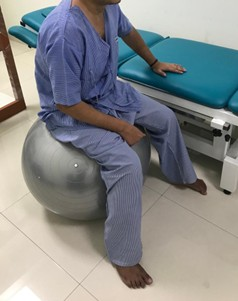
Figure 2. Balance training of the patient with gym ball.
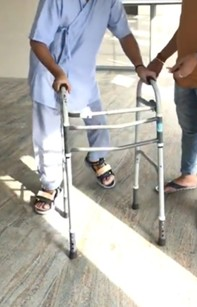
Figure 3. Gait training with walker.
Outcome and follow-up
The Functional Independence Measure and Functional Assessment Measure (FIM-FAM Scale), Fugl-Meyer Assessment Scale (FMA), and Berg Balance Scale (BBS) were used to assess the clinical effects of rTMS in combination with neuromotor rehabilitation therapy. Data were collected before rTMS and at 2, 4, and 8 weeks after therapy. We calculated the percentage change at each visit using the assessment scale. We calculated the percentage change between the 1st and 4th visits using the following formula: [(4th visit score - 1st visit score)/total score × 100].
As shown in Table 1, the FMA score increased in both the upper limb (T0-43/66, T3-60/66) and the lower limb (T0–15/34, T3–22/34). With neuromotor rehabilitation, the BBS also increased (T0–2/56, T3–23/56). The FIM-FAM score significantly improved (+17.6%) after this therapy.
|
Scale |
1st visit T0 |
2nd visit T1 |
3rd visit T2 |
4th visit T3 |
Max score |
Difference (MCID) |
% of change between 1st and 4th visit |
|
FMA(UE)motor |
43 |
58 |
60 |
60 |
66 |
17 (>5.3) |
+26 % |
|
FMA(LE)motor |
15 |
18 |
20 |
22 |
34 |
7 (>6) |
+21% |
|
BBS |
2 |
18 |
20 |
23 |
56 |
21 (>7) |
+38% |
|
FIM-FAM |
113 |
142 |
148 |
150 |
210 |
37 (>22) |
+18% |
|
Table 1 showing changes of FMA (for upper limb & lower limb), BBS & FIM- FAM over time. FMA (UE): Fugl-Meyer Assessment Scale Upper Limb; FMA (LE): Fugl-Meyer Assessment Scale Lower Limb; BBS: Berg Balance Scale; FIM-FAM Scale: Functional Independence Measure and Functional Assessment Measure |
|||||||
The FMA score for the upper limb initially was 43, reaching 66 by the end of the 8th week, representing a 25% increase. Similarly, the FMA for the lower limbs also increased by 20%. For BBS, a 37% improvement was observed. Regarding ADL independence, we observed a 17% improvement in FIM-FAM scores. It is also observed is an improvement in Minimal Clinically Important Difference (MCID) scores for all four scales.
Discussion
Our study revealed that 10 days of repetitive transcranial magnetic stimulation combined with neuromotor rehabilitation may lead to clinically meaningful improvements in upper and lower limb motor function and balance, even 4 years after traumatic brain injury (TBI). Post-treatment, the patient could walk with minimal support (with a single elbow crutch support). Modulation in the cerebral cortex (primary motor cortex) helps to facilitate further motor recovery and balance.
In the literature, motor and cognitive recovery is maximal in the first few weeks after injury until the tenth week, after which generally no improvement is observed [19]. Our patient demonstrated improvements in motor function and coordination, even 4 years after the injury, as shown in Figure 4. These findings suggest that neuromodulation of cerebral and cerebellar circuits via repetitive transcranial magnetic stimulation (rTMS) and neuromotor rehabilitation facilitates recovery of motor function, balance, and coordination. Improvements in upper and lower limb coordination can be attributed to the cerebellum’s role in movement execution and motor control, as well as its connections with M1 via the cerebellothalamocortical pathway [20,21]. At the end of therapy, there was significant improvement in gait, and the patient was able to walk with a cane. An improvement in the upper limb was observed, and his activities of daily living became independent to some extent in eating, grooming, bathing, and toileting; ultimately, vocational training was initiated.

Rosu et al. (2023) reported that “rTMS and Cerebrolysin may be effective interventions for improving cognitive and functional outcomes in TBI patients” [22]. Our findings also revealed a positive effect on motor activity and balance improvement following rTMS with neuromotor rehabilitation in a traumatic brain injury patient.
Khedr et al. (2005) explored the effects of rTMS in patients with acute ischemic stroke, which was applied over the M1 of the stroke hemisphere for 10 days [23] and consisted of 10 10-second trains of 3-Hz stimulation with 50 seconds between each train. In that study, patients were initially given passive limb manipulation, which progressed to more active movements by the end of the first week, provided they showed improvement in their function. Patients in the active rTMS group showed significantly greater improvement than those in the sham rTMS group, resulting in a higher percentage of independence and a greater proportion of patients with only mild disability at the follow-up assessment, 10 days after the end of treatment [23].
We did not observe any adverse effects during the therapy, such as seizures or headaches.
Unfortunately, very few studies have investigated gait and balance rehabilitation in patients with TBI, and very limited evidence exists regarding the use of r-TMS for TBI. Therefore, this case study investigated the effects of r-TMS combined with physical therapy on balance, gait, and motor functions in TBI patients. To establish the clinical effects of rTMS in rehabilitation, larger RCTs are needed to assess its effects on motor function, coordination, and balance, as well as its side effects, contraindications, and the duration of its effects after therapy.
Limitations
This is a single-case study, and there is a confounding bias regarding the effectiveness of repetitive transcranial magnetic stimulation (rTMS) alone, as it was combined with neurorehabilitation. A longer period of follow-up and RCT is required to observe the effect, persistence of the efficacy and any long-term side effects of rTMS.
Conclusion
Repetitive transcranial magnetic stimulation (rTMS) in traumatic brain injury (TBI) may help improve upper and lower limb function, activity of daily living, and balance and coordination and may help in improving the activity of daily living even after 4 years of brain trauma.
Conflict of Interest
No conflict of interest.
References
2. Fu TS, Jing R, McFaull SR, Cusimano MD. Recent trends in hospitalization and in-hospital mortality associated with traumatic brain injury in Canada: A nationwide, population-based study. J Trauma Acute Care Surg. 2015 Sep;79(3):449–54.
3. McCulloch KL, Buxton E, Hackney J, Lowers S. Balance, attention, and dual-task performance during walking after brain injury: associations with falls history. J Head Trauma Rehabil. 2010 May-Jun;25(3):155–63.
4. Winkler D, Unsworth C, Sloan S. Factors that lead to successful community integration following severe traumatic brain injury. J Head Trauma Rehabil. 2006 Jan-Feb;21(1):8–21.
5. Menon DK, Schwab K, Wright DW, Maas AI; Demographics and Clinical Assessment Working Group of the International and Interagency Initiative toward Common Data Elements for Research on Traumatic Brain Injury and Psychological Health. Position statement: definition of traumatic brain injury. Arch Phys Med Rehabil. 2010 Nov;91(11):1637–40.
6. Humphreys I, Wood RL, Phillips CJ, Macey S. The costs of traumatic brain injury: a literature review. Clinicoecon Outcomes Res. 2013 Jun 26;5:281–7.
7. Shimizu T, Hosaki A, Hino T, Sato M, Komori T, Hirai S, et al. Motor cortical disinhibition in the unaffected hemisphere after unilateral cortical stroke. Brain. 2002 Aug;125(Pt 8):1896–907.
8. Murase N, Duque J, Mazzocchio R, Cohen LG. Influence of interhemispheric interactions on motor function in chronic stroke. Ann Neurol. 2004 Mar;55(3):400–9.
9. Hummel FC, Cohen LG. Non-invasive brain stimulation: a new strategy to improve neurorehabilitation after stroke? Lancet Neurol. 2006 Aug;5(8):708–12.
10. Fregni F, Pascual-Leone A. Technology insight: noninvasive brain stimulation in neurology-perspectives on the therapeutic potential of rTMS and tDCS. Nat Clin Pract Neurol. 2007 Jul;3(7):383–93.
11. Masahiro Abo, Wataru Kakuda. Rehabilitation with rTMS. Cham: Springer International Publishing; 2015.
12. Pascual-Leone A, Davey N, Rothwell JC, Wassermann E, Puri BK. Handbook of Transcranial Magnetic Stimulation. London (UK): Arnold; 2002
13. Kim YH, You SH, Ko MH, Park JW, Lee KH, Jang SH, et al. Repetitive transcranial magnetic stimulation-induced corticomotor excitability and associated motor skill acquisition in chronic stroke. Stroke. 2006 Jun;37(6):1471–6.
14. Pomeroy VM, Cloud G, Tallis RC, Donaldson C, Nayak V, Miller S. Transcranial magnetic stimulation and muscle contraction to enhance stroke recovery: a randomized proof-of-principle and feasibility investigation. Neurorehabil Neural Repair. 2007 Nov-Dec;21(6):509–17.
15. Malcolm MP, Triggs WJ, Light KE, Gonzalez Rothi LJ, Wu S, Reid K, et al. Repetitive transcranial magnetic stimulation as an adjunct to constraint-induced therapy: an exploratory randomized controlled trial. Am J Phys Med Rehabil. 2007 Sep;86(9):707–15.
16. Fugl-Meyer AR, Jääskö L, Leyman I, Olsson S, Steglind S. The post-stroke hemiplegic patient. 1. a method for evaluation of physical performance. Scand J Rehabil Med. 1975;7(1):13–31.
17. Berg KO, Wood-Dauphinee SL, Williams JI, Maki B. Measuring balance in the elderly: validation of an instrument. Can J Public Health. 1992 Jul-Aug;83 Suppl 2:S7–11.
18. Chantsoulis M, Półrola P, Góral-Półrola J, Hajdukiewicz A, Supiński J, Kropotov JD, et al. Application of ERPs neuromarkers for assessment and treatment of a patient with chronic crossed aphasia after severe TBI and long-term coma - Case Report. Ann Agric Environ Med. 2017 Mar 31;24(1):141–7.
19. Hart T, Kozlowski AJ, Whyte J, Poulsen I, Kristensen K, Nordenbo A, et al. Functional recovery after severe traumatic brain injury: an individual growth curve approach. Arch Phys Med Rehabil. 2014 Nov;95(11):2103–10.
20. Groiss SJ, Ugawa Y. Cerebellum. Handb Clin Neurol. 2013;116:643–53.
21. Martino Cinnera A, Bonnì S, Iosa M, Ponzo V, Fusco A, Caltagirone C, et al. Clinical effects of non-invasive cerebellar magnetic stimulation treatment combined with neuromotor rehabilitation in traumatic brain injury. A single case study. Funct Neurol. 2016 Apr-Jun;31(2):117–20.
22. Verisezan Rosu O, Jemna N, Hapca E, Benedek I, Vadan I, Muresanu I, et al. Cerebrolysin and repetitive transcranial magnetic stimulation (rTMS) in patients with traumatic brain injury: a three-arm randomized trial. Front Neurosci. 2023 Jun 8;17:1186751.
23. Khedr EM, Ahmed MA, Fathy N, Rothwell JC. Therapeutic trial of repetitive transcranial magnetic stimulation after acute ischemic stroke. Neurology. 2005 Aug 9;65(3):466–8.
