Immunologic Memory for Cancer Vaccine Efficacy
Recent reports and ongoing clinical trials demonstrate potential roles for cancer vaccine therapy in a number of malignancies [1]. Adaptive cellular immune responses are required for protective immunity when vaccines are used against infectious diseases [2]. For therapeutic cancer vaccines used to treat established tumors, elaboration of durable T cell mediated responses are also understood to be necessary for efficacy [3–5]. Immune responses measured in peripheral blood lymphocytes correlate with clinical outcomes [6,7]. Persistent immunity in the form of memory T cells specific to cancer antigens has been shown to lead to effective anti-tumor immunity [8]. Antigen-specific T-cell responses after vaccination has correlated with survival in multiple cancers [9–14].
The importance of antigen specific memory T cells for cancer vaccine efficacy highlights the need for improved understanding of biomarkers to optimize vaccine therapies for cancer. Detecting pre-existing immunity to antigens included in a cancer vaccine may select candidates for vaccination. The timing and magnitude of an antigen-specific immune response to vaccination may identify patients who will develop productive and persistent anti-tumor immunity.
Pre-existing Immunity to Vaccine Antigens
Developing effective antigen specific T cells after vaccination depends on antigen-specific T-cell precursor frequencies [15,16]. Pre-existing CD8 T-cell memory will influence the magnitude of naïve CD8 T cell responses [17]. Acquisition of an effector memory cell phenotype will depend on the CD8 T-cell differentiation state, which is also determined by preexisting memory T cells [18]. Levels of preexisting antigen specific T cells could conceivably be either a positive or negative predictor for persistent antitumor immunity. Higher precursor frequency could lead to more rapid clonal expansion of effector and memory T cells when vaccines are introduced, accelerating antitumor responses. More precursors may not always provide an advantage as there may be a threshold beyond which there may be decreased responses due to intraclonal competition, although this may also be overcome with CD4 T cell help [19,20]. Conversely, high numbers of antigen specific T cells in the presence of known tumor burden or disease progression could represent the inability of an antigen to serve as an immunogenic target, possibly due to immune evasion through the loss of the target on tumor cells.
In our study of vaccines targeting HER2, we found that patients with higher pre-existent T-cell responses to HER2 were more likely to develop higher HER2 T-cell responses. We also found that these antigen specific T cell responses were more likely to last greater than 6 months after immunization in patients with pre-existent immunity than those that did not. Sixty-three percent of patients with baseline antigen specific T immunity against HER2 continued to have responses 1 year after completing HER2 targeted vaccines and the presence of preexisting immunity was associated with a 0.33 log increase [21].
Preexisting immunity to the vaccine vector, rather than the target antigen may be a particular consideration for some cancer vaccines. This may be particularly true for vaccines utilizing bacterial or viral vectors, as the subsequent immune response may be modified due to more rapid clearance of the vaccine [22]. Preexisting immunity to viral vectors such as adenovirus have been an obstacle to generating immune responses because they can reduce the time and exposure to the vaccinated antigens, but this has also been overcome to generate antitumor immunity [23,24].
Timing and Magnitude of Response to Vaccination
We have found that the magnitude of antigen-specific T cell elaborated one month after a vaccine series is more predictive of long term immunity than the existence of pre-existing immune responses [21]. Patients with HER2+ cancers were treated with HER2 directed vaccines and antigen specific T-cells were measured by interferon-gamma ELISPOT assays at baseline, one month after completing vaccine series, and long-term follow-up at 1 year. One month after vaccination, an increase in HER2+ interferon-gamma T-cell frequency was associated with retaining those immune responses long term. A one-log increase of month-1 T-cell response was associated with 0.47-log increase in long term T-cell responses. Patients who had immunity one month after completing the vaccination series had higher odds of having persistent HER2-specific immunity, OR=6.50 (95%CI 2.41-17.52, p=0.0002), regardless of whether they had pre-existing immunity.
Signaling through interferon-gamma, can control the magnitude of T-cell responses and memory differentiation [25]. However, although interferon-gamma is produced by cytotoxic T cells and is associated with a desired Th1 response for antitumor immunity, it is also associated with synthesis of immune checkpoint molecules and immunosuppressive mechanisms [26]. Therefore the magnitude and timing, in addition to the phenotype of the elaborated immune response to vaccine are all important considerations for long-lasting immunity.
Depending on the vaccine, T cell frequencies are generally expected to peak between days 7 to 14 before contracting with a very small percentage of T cells persisting as memory T cells [27]. Therefore, the 1 month timepoint may assess the earliest measures of initial clonal expansion that establishes a memory pool of T cells, capturing the transition from effector to early memory T cells representing a post contraction population that is potentially long-lived [28]. The magnitude of this response is related to the frequency of antigen specific T cells in the preexisting repertoire, how many are activated and start to divide with vaccination and how many cells survive the contraction phase. With each successive vaccination in a series, the precursor frequency would be expected to increase along with the subsequent population of memory T cells, which should represent anti-tumor T cell immunity. Meta-analysis of TCR engineered T cell trials suggest that clinical responses will correlate with higher numbers of antigen specific effector T cells [29]. Mathematical modeling taking into account T cell priming, migration, and interaction with tumors based on these data support the existence of a threshold of effector T cell needed to control tumor progression [29].
A third of patients in our study did not develop antigen specific immunity one month after vaccination [21]. For patients that do not develop immunity after vaccination, a number of factors may be at play. These may include the current immune microenvironment of the tumor, the immune phenotype of the T cells available for a vaccine, and the suitability of a patient’s tumor and immune system for a particular vaccine. The genotype and immune repertoire of patient’s individual immune system may favor a response to vaccines on the basis of HLA restriction of the vaccine epitopes or the presence of pre-existing immunity to the vectors or adjuvants.
Conclusions
Predictive markers for long term immunity from cancer vaccines require further study to optimally deploy vaccines. The journey from vaccination to generation of effector T cells capable of trafficking to tumors and overcoming a hostile tumor microenvironment may also be influenced by the immune properties of the specific cancer, the patient’s own immune repertoire, in addition to factors we have discussed. Increasing our understanding of these immune dynamics, and how they can be modulated will allow the development of rational strategies to incorporate cancer vaccines into cancer treatment paradigms. As the development of cancer vaccines accelerate, markers that predict long-lasting anti-tumor immunity become an ever more important consideration. Factors that span the timeline from before a patient receives a vaccine to clinical responses (Figure 1) are ripe for further study and could be explored further as a composite prediction model integrating pre-existing factors with early immune responses, and tumor microenvironment biomarkers. Identifying and monitoring the key immune predictors of effective antitumor immunity could permit optimal integration of vaccines with adjuvants, boosters, and other immunotherapies such as immune checkpoint blockers which has been noted in several clinical trials [7,30–32]. These agents could further synergize with vaccines and help overcome barriers to efficacy such as immunosuppressive tumor microenvironments and factors that influence T cell exhaustion, expansion and persistence.
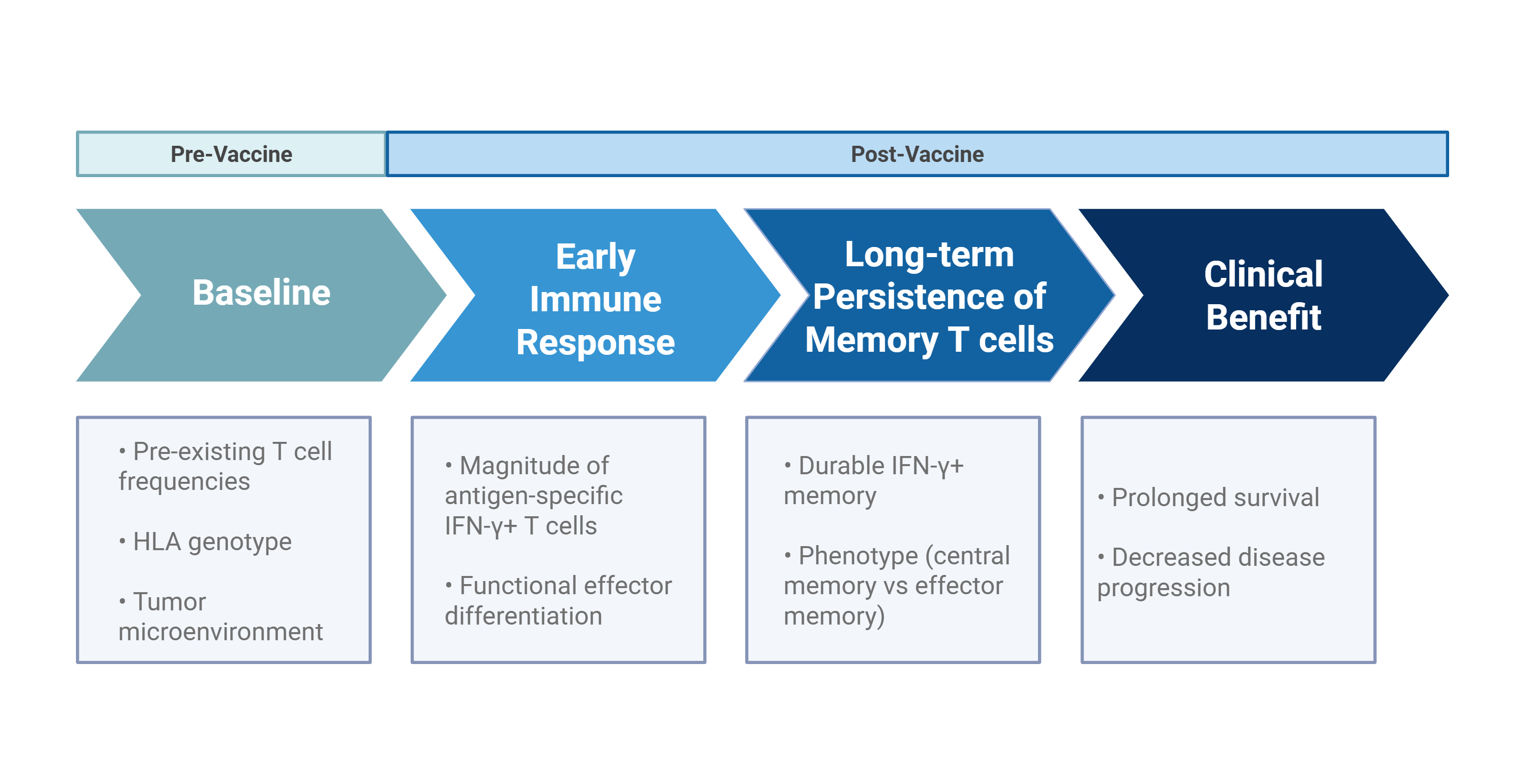
Figure 1. Proposed predictive timeline linking pre-vaccination determinants to clinical benefit following therapeutic vaccination.
Acknowledgements
This work was supported by R01 CA098761, U01 CA154967, and DAMD BC030289. MLD was supported by the Athena Distinguished Professor of Breast Cancer Research, an ACS Clinical Research Professorship and is a Komen Scholar.
Competing Interests
JBL has received grant funding from Merck, AstraZeneca, Precigen, ArsenalBio, Volastra, Nurix, Aminex Therapeutics through his institution and is a consultant for Verismo Therapeutics. MLD has grant funding from Precigen, Veanna, Bavarian Nordic, and Aston Sci. MLD also holds shares in Epithany and is an inventor on patents held by the University of Washington. The authors declare there are no other competing interests.
References
2. Ahmed R, Gray D. Immunological memory and protective immunity: understanding their relation. Science. 1996 Apr 5;272(5258):54–60.
3. Palucka AK, Coussens LM. The Basis of Oncoimmunology. Cell. 2016 Mar 10;164(6):1233–47.
4. Romero P, Banchereau J, Bhardwaj N, Cockett M, Disis ML, Dranoff G, et al. The Human Vaccines Project: A roadmap for cancer vaccine development. Sci Transl Med. 2016 Apr 13;8(334):334ps9.
5. Mittra S, Harding SM, Kaech SM. Memory T Cells in the Immunoprevention of Cancer: A Switch from Therapeutic to Prophylactic Approaches. J Immunol. 2023 Sep 15;211(6):907–16.
6. Slingluff CL Jr, Lee S, Zhao F, Chianese-Bullock KA, Olson WC, Butterfield LH, et al. A randomized phase II trial of multiepitope vaccination with melanoma peptides for cytotoxic T cells and helper T cells for patients with metastatic melanoma (E1602). Clin Cancer Res. 2013 Aug 1;19(15):4228–38.
7. McNeel DG, Eickhoff JC, Wargowski E, Zahm C, Staab MJ, Straus J, et al. Concurrent, but not sequential, PD-1 blockade with a DNA vaccine elicits anti-tumor responses in patients with metastatic, castration-resistant prostate cancer. Oncotarget. 2018 May 22;9(39):25586–96.
8. Mizukoshi E, Nakagawa H, Tamai T, Kitahara M, Fushimi K, Nio K, et al. Peptide vaccine-treated, long-term surviving cancer patients harbor self-renewing tumor-specific CD8+ T cells. Nat Commun. 2022 Jun 3;13(1):3123.
9. Aruga A, Takeshita N, Kotera Y, Okuyama R, Matsushita N, Ohta T, et al. Long-term Vaccination with Multiple Peptides Derived from Cancer-Testis Antigens Can Maintain a Specific T-cell Response and Achieve Disease Stability in Advanced Biliary Tract Cancer. Clin Cancer Res. 2013 Apr 15;19(8):2224–31.
10. Odunsi K, Matsuzaki J, Karbach J, Neumann A, Mhawech-Fauceglia P, Miller A, et al. Efficacy of vaccination with recombinant vaccinia and fowlpox vectors expressing NY-ESO-1 antigen in ovarian cancer and melanoma patients. Proc Natl Acad Sci U S A. 2012 Apr 10;109(15):5797–802.
11. van Baren N, Bonnet MC, Dréno B, Khammari A, Dorval T, Piperno-Neumann S, et al. Tumoral and immunologic response after vaccination of melanoma patients with an ALVAC virus encoding MAGE antigens recognized by T cells. J Clin Oncol. 2005 Dec 10;23(35):9008–21.
12. Kirkwood JM, Lee S, Moschos SJ, Albertini MR, Michalak JC, Sander C, et al. Immunogenicity and antitumor effects of vaccination with peptide vaccine+/-granulocyte-monocyte colony-stimulating factor and/or IFN-alpha2b in advanced metastatic melanoma: Eastern Cooperative Oncology Group Phase II Trial E1696. Clin Cancer Res. 2009 Feb 15;15(4):1443–51.
13. Disis ML, Wallace DR, Gooley TA, Dang Y, Slota M, Lu H, et al. Concurrent trastuzumab and HER2/neu-specific vaccination in patients with metastatic breast cancer. J Clin Oncol. 2009 Oct 1;27(28):4685–92.
14. Gulley JL, Arlen PM, Madan RA, Tsang KY, Pazdur MP, Skarupa L, et al. Immunologic and prognostic factors associated with overall survival employing a poxviral-based PSA vaccine in metastatic castrate-resistant prostate cancer. Cancer Immunol Immunother. 2010 May;59(5):663–74.
15. Moon JJ, Chu HH, Pepper M, McSorley SJ, Jameson SC, Kedl RM, et al. Naive CD4(+) T cell frequency varies for different epitopes and predicts repertoire diversity and response magnitude. Immunity. 2007 Aug;27(2):203–13.
16. Fraser KA, Schenkel JM, Jameson SC, Vezys V, Masopust D. Preexisting high frequencies of memory CD8+ T cells favor rapid memory differentiation and preservation of proliferative potential upon boosting. Immunity. 2013 Jul 25;39(1):171–83.
17. Martin MD, Wirth TC, Lauer P, Harty JT, Badovinac VP. The impact of pre-existing memory on differentiation of newly recruited naive CD8 T cells. J Immunol. 2011 Sep 15;187(6):2923-31.
18. Joshi NS, Cui W, Dominguez CX, Chen JH, Hand TW, Kaech SM. Increased numbers of preexisting memory CD8 T cells and decreased T-bet expression can restrain terminal differentiation of secondary effector and memory CD8 T cells. J Immunol. 2011 Oct 15;187(8):4068–76.
19. Rizzuto GA, Merghoub T, Hirschhorn-Cymerman D, Liu C, Lesokhin AM, Sahawneh D, et al. Self-antigen-specific CD8+ T cell precursor frequency determines the quality of the antitumor immune response. J Exp Med. 2009 Apr 13;206(4):849–66.
20. Malandro N, Budhu S, Kuhn NF, Liu C, Murphy JT, Cortez C, et al. Clonal Abundance of Tumor-Specific CD4(+) T Cells Potentiates Efficacy and Alters Susceptibility to Exhaustion. Immunity. 2016 Jan 19;44(1):179–193.
21. Liao JB, Dai JY, Reichow JL, Lim JB, Hitchcock-Bernhardt KM, Stanton SE, et al. Magnitude of antigen-specific T-cell immunity the month after completing vaccination series predicts the development of long-term persistence of antitumor immune response. J Immunother Cancer. 2024 Nov 9;12(11):e010251.
22. Saxena M, Van TTH, Baird FJ, Coloe PJ, Smooker PM. Pre-existing immunity against vaccine vectors--friend or foe? Microbiology (Reading). 2013 Jan;159(Pt 1):1–11.
23. Pine SO, Kublin JG, Hammer SM, Borgerding J, Huang Y, Casimiro DR, et al. Pre-existing adenovirus immunity modifies a complex mixed Th1 and Th2 cytokine response to an Ad5/HIV-1 vaccine candidate in humans. PLoS One. 2011 Apr 13;6(4):e18526.
24. Tseha ST. Role of Adenoviruses in Cancer Therapy. Front Oncol. 2022 Jun 9;12:772659.
25. Sercan O, Stoycheva D, Hämmerling GJ, Arnold B, Schüler T. IFN-gamma receptor signaling regulates memory CD8+ T cell differentiation. J Immunol. 2010 Mar 15;184(6):2855–62.
26. Jorgovanovic D, Song M, Wang L, Zhang Y. Roles of IFN-γ in tumor progression and regression: a review. Biomark Res. 2020 Sep 29;8:49.
27. Williams MA, Bevan MJ. Effector and memory CTL differentiation. Annu Rev Immunol. 2007;25:171–92.
28. Kim C, Fang F, Weyand CM, Goronzy JJ. The life cycle of a T cell after vaccination - where does immune ageing strike? Clin Exp Immunol. 2017 Jan;187(1):71–81.
29. Mallet M, Boulos RE, Alcazer V, Bonaventura P, Estornes Y, Chuvin N, et al. Tumour burden and antigen-specific T cell magnitude represent major parameters for clinical response to cancer vaccine and TCR-engineered T cell therapy. Eur J Cancer. 2022 Aug;171:96–105.
30. Massarelli E, William W, Johnson F, Kies M, Ferrarotto R, Guo M, et al. Combining Immune Checkpoint Blockade and Tumor-Specific Vaccine for Patients With Incurable Human Papillomavirus 16-Related Cancer: A Phase 2 Clinical Trial. JAMA Oncol. 2019 Jan 1;5(1):67–73.
31. Kos FJ, Frankel P, Cristea M, Eng M, Tinsley R, Dempsey S, et al. Immunologic Signatures of Peripheral Blood T Cells Reveal the Outcome of p53MVA Vaccine and Pembrolizumab Treatment in Patients with Advanced Ovarian Cancer. Cancer Res Commun. 2023 Dec 20;3(12):2585–95.
32. Youn JW, Hur SY, Woo JW, Kim YM, Lim MC, Park SY, et al. Pembrolizumab plus GX-188E therapeutic DNA vaccine in patients with HPV-16-positive or HPV-18-positive advanced cervical cancer: interim results of a single-arm, phase 2 trial. Lancet Oncol. 2020 Dec;21(12):1653–60.
