Abstract
Objective: This case aims to describe a rare case of complex regional pain syndrome type II (CRPS II) associated with tibial nerve entrapment and dystonia, and to highlight the role of electrodiagnostic (EDX) testing in identifying a potentially treatable peripheral nerve lesion.
Methods: We report a rare case of CRPS II involving the tibial nerve with associated dystonia, painful paresthesia’s, and autonomic changes in the right foot. EDX testing were performed and demonstrated a focal tibial neuropathy at the tarsal tunnel. Based on these findings, the patient underwent surgical decompression of the tarsal tunnel.
Results: Following surgical release, the patient experienced significant improvement in pain, complete resolution of dystonia, and normalization of sensory and functional deficits, with return to baseline functional status.
Conclusion: This case demonstrates that targeted evaluation with EDX testing can identify focal peripheral nerve pathology in select patients with CRPS II, leading to effective surgical intervention. Resolution of CRPS symptoms and associated dystonia after tarsal tunnel release supports further investigation into peripheral mechanisms contributing to dystonia and paresthesia’s in CRPS.
Keywords
Complex regional pain syndrome, Electrodiagnosis, Focal dystonia, Tarsal tunnel syndrome
Introduction
Focal dystonia is seen in about 25% of all complex regional pain syndrome (CRPS) cases and is characterized by involuntary predominantly flexor postures of the distal limbs and can spread to more proximal sites [1,2]. Dystonia linked to peripheral trauma is a rarely recognized clinical syndrome that goes by many names including traumatic dystonia, focal dystonia and/or CRPS dystonia. Dystonia associated with CRPS can present as dynamic, task-dependent movements or as fixed, sustained postures that persist regardless of activity [3]. Focal dystonia is characterized by dynamic, twisting, or repetitive movements, whereas fixed dystonia manifests as a sustained, immobile posture of a limb or body region that remains unchanged with activity [4]. These motor abnormalities are frequently disabling and are associated with poor functional outcomes and limited response to treatment [5].
CRPS is a chronic pain condition usually following trivial trauma presenting with persistent pain, autonomic and trophic features. CRPS is classified into two main types based on the absence of nerve injury (type I) or the presence of a nerve injury (type II) [6]. Diagnosing CRPS II can include objective testing such as a neurological examination and/or electrodiagnostic (EDX) studies to detect the presence of a discrete nerve injury [7]. While this distinction is well defined, most studies of CRPS–associated dystonia have limited diagnostic testing [7,8]. In one study evaluating patients with fixed dystonia, about 63% of cases had a peripheral injury prior to the onset of dystonia but only 2 out of 42 prospective patients had abnormal EDX findings [6].
Looking for a discrete nerve injury to diagnose CRPS II may be overlooked in the acute setting due to the presence prominent dysautonomia that characterizes CRPS. Autonomic manifestations, including temperature asymmetry, skin color changes, abnormal sweating, and edema, are a core feature of CRPS and represent a major challenge to rehabilitation in this patient population [9,10]. These features may exacerbate pain with activity, limit weightbearing and bracing tolerance, and reduce participation in therapeutic exercise, thereby contributing to prolonged disability [11,12]. As a result, identifying potentially modifiable contributors to pain and autonomic dysfunction is critical when evaluating patients with CRPS and associated dystonia [12].
Treatment interventions for CRPS-associated focal dystonia have included physical and occupational therapies [6], bracing/casting, oral antispasmodics, botulinum toxin injections [13,14], spinal cord stimulation [15], intrathecal baclofen [16], electrical nerve stimulation [17], and surgical interventions [18,19]. Focal dystonia carries a poor prognosis– one study showed relapsing dystonic symptoms in ~73% of participants [6]. However, most of these studies have been on a patient population of CRPS I and either excluded or included only a minority of patients with CRPS II [2,6,18].
Decompression of entrapped peripheral nerves is frequently used to improve sensory and motor symptoms (paresthesia’s, weakness, etc.) but has rarely been reported to improve dystonia. This case report describes the successful treatment of focal dystonia and pain in CRPS II with tibial nerve decompression. We also highlight the potential treatment options for CRPS and possible management differences between CRPS I and II.
Case Presentation
Patient description
A 28–year–old male presents with right foot pain, paresthesia’s, and muscle cramping following a traumatic injury to his right foot 3 years prior. He was previously diagnosed with CRPS I based on the Budapest criteria [8] as well as an initial workup demonstrating normal labs, normal bone scan, and normal EDX testing on three separate occasions. However, the right tibial nerve into the foot was not evaluated in any of these EDX studies. Right ankle MRI demonstrated marrow edema in the talus and calcaneus, consistent with CRPS. Prior to presentation at our clinic, the patient had failed multiple interventions, including bracing, platelet–rich plasma injections, outpatient physical therapy, several neuropathic pain medications, three separate lumbar epidural corticosteroid injections, and an inpatient rehabilitation program focused on chronic pain.
At the time of presentation to our clinic, the patient primarily reported focal neuropathic pain and paresthesia’s localized to the plantar aspect of the foot, with cramping and abnormal involuntary posturing of the right ankle. These symptoms resulted in the inability to work or drive a car due to dystonia. He did not report widespread pain, contralateral limb symptoms, or upper extremity involvement. He did not endorse prominent systemic features of dysautonomia such as orthostatic intolerance, fatigue, palpitation, cognitive dysfunction, bowel or bladder symptoms, or diffuse skin changes.
Physical exam revealed dynamic dystonic posturing of the long toe flexors/extensors and foot intrinsic muscles with walking and other activities. Full strength and normal patellar, Achilles, and Babinski reflexes were noted. His gait was impaired with evidence of absent heel strike and weight bearing on the lateral aspect of the foot during stance phase. Allodynia/hyperpathia was present in the medial plantar nerve distribution with sensory loss along the lateral plantar nerve. Tinel sign was positive along the tibial nerve at the tarsal tunnel with radiating symptoms in the medial and lateral plantar nerve areas of innervation.
Repeat EDX testing demonstrated mildly decreased amplitude of the tibial compound muscle action potential (CMAP) across the ankle recording at the abductor hallucis brevis and decreased mixed nerve action potential amplitude of the medial and lateral plantar nerves (Table 1). Needle EMG was normal in; first dorsal interossei pedis, anterior tibialis, gastrocnemius, short head of the biceps femoris, semimembranosus, vastus medialis oblique. We interpreted this as suggesting an incomplete tibial neuropathy in the tarsal tunnel without active denervation (no fibrillation potentials).
|
a. Motor NCS summary |
||||
|
Site (Right) |
Onset (ms) |
Norm Onset (ms) |
O-T Amp (mV) |
Norm O-T Amp (mV) |
|
Tibial Motor (Abd Hall Brev) |
||||
|
Ankle |
4.8 |
< 6.1 |
4.1 |
> 4.4 |
|
Knee |
14.9 |
|
3.7 |
|
|
b. Mixed NCS summary |
||||
|
Site (Right) |
Peak (ms) |
Norm Peak (ms) |
P-T Amp (mV) |
Norm P-T Amp (mV) |
|
Lateral Plantar Mixed (Medial Malleolus) |
||||
|
Lateral Foot |
4.0 |
< 4.5 |
5.7 |
> 8 |
|
Medial Plantar Mixed (Medial Malleolus) |
||||
|
Medial Foot |
4.0 |
< 4.5 |
5.6 |
> 8 |
|
Abbreviations: NCS: Nerve Conduction Studies |
||||
Intervention
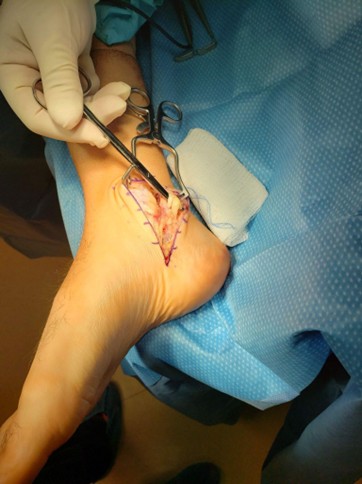
A tibial nerve block resulted in temporary resolution of dystonia and paresthesia’s (Supplementary Video 1). Based on this response, the patient subsequently underwent surgical tarsal tunnel release (Figure 1). The procedure was performed in accordance with the technique described by Loder et al. [20]. A curvilinear longitudinal incision was made midway between the central aspect of the medial malleolus and the anterior aspect of the Achilles tendon, extending approximately three centimeters proximally and distally into the porta pedis. The posterior tibial nerve was carefully decompressed with a hands-off technique. Intraoperatively, the laciniate ligament (flexor retinaculum) was noted to be taut and constricting the posterior tibial nerve. A complete proximal-to-distal decompression of the posterior tibial nerve was performed. Distally, the posterior tibial nerve was followed to the level of its terminal branches, including the medial and lateral plantar nerves. The investing fascia of the abductor hallucis brevis muscle belly was identified and carefully released to allow for decompression of the distal nerve branches within the porta pedis, as this structure represents a known site of distal entrapment and potential contributor to persistent or recurrent tarsal tunnel symptoms. The laciniate ligament was released and was not repaired prior to closure, allowing for unimpeded decompression.
Figure 1. Intraoperative exposure of the posterior tibial nerve during right tarsal tunnel release.
Postoperatively, the patient remained non-weight bearing on crutches for two weeks. At the two-week postoperative visit, sutures were removed, and the patient was transitioned to full weight bearing in a normal shoe. The patient was followed postoperatively and demonstrated progressive and sustained resolution of preoperative symptoms (pain and movement abnormalities), consistent with effective nerve decompression.
Outcome
Following surgical decompression, the patient was seen in clinic one month later, and allodynia and hyperpathia in the foot resolved. No numbness, tingling, or burning was reported anywhere on the foot. Light touch and pin-wheel examination was normal in the superficial fibular, deep fibular, sural, medial plantar and lateral plantar nerve distributions. He had full range of motion and normal gait without dystonia. Supplementary Video 1 includes videos of gait impacted by dystonia, dystonia resolved with nerve block and again following surgical intervention. The patient was able to return to work and drive a car without difficulty. Improvement was assessed clinically based on patient reports and physical examination. At his four-month postoperative follow-up with the surgeon, the patient continued to demonstrate complete resolution of foot pain and dystonia and had returned to full-time work without restrictions.
In an ideal world, long term follow up would be preferred. The patient did not return for scheduled follow-up visits after apparent successful surgery, precluding long-term outcome assessment. Prior to the surgery, he followed up consistently for treatment for over three years for this problem. Although this could be incorrect, we think the lack of follow up was most consistent with symptom resolution.
Discussion
This case illustrates a rare but important presentation of CRPS II–associated dystonia secondary to tibial nerve entrapment. Peripheral nerve injury may be underrecognized in CRPS, particularly when autonomic features dominate the clinical presentation [7,8]. Failure to identify a discrete nerve lesion can result in misclassification as CRPS type I and delay consideration of targeted treatments directed at the underlying nerve injury. In recent studies, CRPS II specific treatment approaches have been proposed. MacRae et al. 2023 showed improved outcomes using oral prednisone and surgical interventions in patients with CRPS II with positive EDX findings [21]. In our case, focused EDX testing identified a tibial neuropathy, reclassifying the diagnosis as CRPS II and broadening possible treatments.
A weakness of this case report is that no validated CRPS–specific outcome measures (e.g., pain scales, functional scores, or autonomic symptom inventories) were obtained at baseline or post-intervention, limiting objective quantification of symptom change. However, we do include a supplementary video with objective visual improvement in dystonia following surgical intervention.
The pathophysiology of improvement in this case is unknown. The central nervous system may be involved, but sorting out changes in pain pathways, motor pathways, psychological reactions or placebo is beyond the scope of our data collection and this paper. This case illustrates a rare, excellent treatment outcome after decompression of the tibial nerve in the tarsal tunnel of focal dystonia, paresthesia’s, and CRPS.
Conclusion
This case demonstrates that identifying and treating a focal tibial neuropathy may lead to significant improvement in CRPS II-associated dystonia, paresthesia’s, neuropathic pain, and function. Comprehensive neurologic examination and targeted EDX testing are critical in CRPS patients with movement abnormalities. Further studies are needed to define optimal outcome measures and to identify which CRPS patients are most likely to benefit from nerve–directed interventions.
Conflicts of Interest
No financial interest or any conflict of interest exists.
Funding Statement
This research received no external funding.
Supplementary Video 1 Description
The video shows the movements at initial evaluation before surgery, during diagnosis with tibial nerve block, pictures during surgery, immediate post operative video, then rehabilitation and recovery videos.
References
2. Munts AG, Mugge W, Meurs TS, Schouten AC, Marinus J, Moseley GL, et al. Fixed dystonia in complex regional pain syndrome: a descriptive and computational modeling approach. BMC Neurol. 2011 May 24; 11:53.
3. Schwartzman RJ, Kerrigan J. The movement disorder of reflex sympathetic dystrophy. Neurology. 1990 Jan;40(1):57–61.
4. LeDoux MS. Dystonia: phenomenology. Parkinsonism Relat Disord. 2012 Jan;18 Suppl 1(Suppl 1): S162–4.
5. Ibrahim NM, Martino D, van de Warrenburg BP, Quinn NP, Bhatia KP, Brown RJ, et al. The prognosis of fixed dystonia: a follow-up study. Parkinsonism Relat Disord. 2009 Sep;15(8):592–7.
6. Schrag A, Trimble M, Quinn N, Bhatia K. The syndrome of fixed dystonia: an evaluation of 103 patients. Brain. 2004 Oct;127(Pt 10):2360–72.
7. Birklein F, Riedl B, Sieweke N, Weber M, Neundörfer B. Neurological findings in complex regional pain syndromes--analysis of 145 cases. Acta Neurol Scand. 2000 Apr;101(4):262–9.
8. Goebel A, Birklein F, Brunner F, Clark JD, Gierthmühlen J, Harden N, et al. The Valencia consensus-based adaptation of the IASP complex regional pain syndrome diagnostic criteria. Pain. 2021 Sep 1;162(9):2346–8.
9. Eldufani J, Elahmer N, Blaise G. A medical mystery of complex regional pain syndrome. Heliyon. 2020 Feb 19;6(2): e03329.
10. Taylor SS, Noor N, Urits I, Paladini A, Sadhu MS, Gibb C, et al. Complex Regional Pain Syndrome: A Comprehensive Review. Pain Ther. 2021 Dec;10(2):875–92.
11. Wasner G, Schattschneider J, Binder A, Baron R. Complex regional pain syndrome--diagnostic, mechanisms, CNS involvement and therapy. Spinal Cord. 2003 Feb;41(2):61–75.
12. Barnhoorn KJ, van de Meent H, van Dongen RT, Klomp FP, Groenewoud H, Samwel H, et al. Pain exposure physical therapy (PEPT) compared to conventional treatment in complex regional pain syndrome type 1: a randomised controlled trial. BMJ Open. 2015 Dec 1;5(12): e008283.
13. Schilder JC, van Dijk JG, Dressler D, Koelman JH, Marinus J, van Hilten JJ. Responsiveness to botulinum toxin type A in muscles of complex regional pain patients with tonic dystonia. J Neural Transm (Vienna). 2014 Jul;121(7):761–7.
14. Siongco PRL, Rosales RL, Moore AP, Freynhagen R, Arimura K, Kanovsky P, et al. Botulinum neurotoxin injections for muscle-based (dystonia and spasticity) and non-muscle-based (neuropathic pain) pain disorders: a meta-analytic study. J Neural Transm (Vienna). 2020 Jun;127(6):935–51.
15. Voet C, le Polain de Waroux B, Forget P, Deumens R, Masquelier E. Spinal cord stimulation for complex regional pain syndrome type 1 with dystonia: a case report and discussion of the literature. F1000Res. 2014 Apr 30; 3:9.
16. Van der Plas AA, van Rijn MA, Marinus J, Putter H, van Hilten JJ. Efficacy of intrathecal baclofen on different pain qualities in complex regional pain syndrome. Anesth Analg. 2013 Jan;116(1):211–5.
17. Buwembo J, Munson R, Rizvi SA, Ijaz A, Gupta S. Direct Sciatic Nerve Electrical Stimulation for Complex Regional Pain Syndrome Type 1. Neuromodulation. 2021 Aug;24(6):1075–82.
18. Bhatia KP, Bhatt MH, Marsden CD. The causalgia-dystonia syndrome. Brain. 1993 Aug;116 (Pt 4):843–51.
19. Kuah A, Arroyo C, Rivera L. Complex regional pain syndrome dystonia induced rigid adductovarus rearfoot: A surgical case study. Foot & Ankle Surgery: Techniques, Reports & Cases. 2022 Sep 1;2(3):100237.
20. Loder B, Mastay A, Morrison K, Goldstein S, Seidel G. A seven-year retrospective outcome study of a medial extensile surgical tarsal tunnel release in the treatment of tarsal tunnel syndrome. J Ortho Sur Reh. 2023;7(5):161.
21. MacRae FA, Boissonnault E, Winston P. A retrospective review of the management and outcomes of patients diagnosed with complex regional pain syndrome type II using electrodiagnostic findings. Can J Pain. 2023 Aug 1;7(1):2242892.