Abstract
Case Summary: This case highlights a rare and diagnostically challenging presentation of liver failure from Autoimmune Hepatitis (AIH) in a 35-year-old male with Human Immunodeficiency Virus (HIV). The patient, initially on antiretroviral therapy (ART) and tuberculosis prophylaxis, presented with a one-month history of abdominal pain, jaundice, acholic stool, tea-colored urine, and nausea. Initial workup suggested obstructive jaundice due to choledocholithiasis and acute calculous cholecystitis, managed by endoscopic retrograde cholangiopancreatography (ERCP) and cholecystectomy. However, the patient’s jaundice and liver enzyme abnormalities persisted postoperatively, prompting further investigation.
Subsequent testing revealed positive anti-smooth muscle antibodies (ASMA) and liver biopsy showed chronic portal and lobular inflammation with cholestasis, confirming the diagnosis of Autoimmune Hepatitis. Common causes of liver dysfunction in HIV such as viral hepatitis and drug-induced liver injury (DILI) were ruled out. Immunosuppressive therapy with Prednisone and Mycophenolate Mofetil stabilized liver function transaminases, but did not normalize bilirubin levels, necessitating a cycle of DPMAS (Double Plasma Molecular Adsorption System), which effectively reduced bilirubin and ammonia levels.
This case underscores the clinical complexity of diagnosing AIH in immunocompromised patients, especially when structural and infectious causes coexist. The successful use of conventional immunosuppression and DPMAS in this context suggests the need for future studies on their safety and efficacy in HIV-positive patients.
Keywords
Case report, Autoimmune hepatitis, HIV, DPMAS
Introduction
Autoimmune hepatitis (AIH) is an uncommon cause of chronic liver disease, with a global pooled prevalence of approximately 15.65 per 100,000 population. It is characterized by immune-mediated hepatocellular injury resulting from loss of tolerance to hepatic autoantigens, leading to chronic inflammation, hepatocellular necrosis, and potential progression to cirrhosis. Over recent decades, the incidence of AIH has been increasing worldwide [1].
In contrast, Human Immunodeficiency Virus (HIV) infection is characterized by progressive immune dysfunction due to quantitative and qualitative depletion of CD4+ T lymphocytes, resulting in immunosuppression [2].
Given these opposing immunologic mechanisms—autoimmunity versus immunodeficiency—the coexistence of AIH and HIV infection is exceedingly rare and represents a clinical paradox. We present a case of a 35-year-old male with HIV infection who developed persistent cholestatic jaundice and was ultimately diagnosed with autoimmune hepatitis despite an initially apparent obstructive etiology.
Case Presentation
A 35-year-old male, diagnosed with HIV, was maintained on antiretroviral therapy consisting of Tenofovir, Lamivudine, and Dolutegravir. He was also receiving Isoniazid prophylaxis. His history was otherwise unremarkable except for a family history of psoriasis and allergy to co-amoxiclav.
He presented with a one-month history of intermittent right upper quadrant pain, triggered by fatty food and alcohol intake, associated with nausea, vomiting, jaundice, acholic stools, and tea-colored urine. Initial evaluation at outside institutions revealed obstructive jaundice secondary to choledocholithiasis with acute calculous cholecystitis. He underwent endoscopic retrograde cholangiopancreatography (ERCP) with successful biliary clearance on final cholangiogram, followed by cholecystectomy. However, despite adequate source control, his jaundice persisted and progressively worsened.
On initial examination, the patient was hemodynamically stable with a body mass index (BMI) of 25.6 kg/m². He had icteric sclerae and right upper quadrant tenderness with a positive Murphy’s sign, without stigmata of chronic liver disease.
Serial laboratory evaluation demonstrated markedly elevated total bilirubin (311.8 to 561.34 µmol/L) with only mild transaminase elevation (AST 31–52 U/L, ALT 21–51 U/L), suggesting a predominantly cholestatic pattern. Viral hepatitis A/B/C workup were negative.
Given the persistence of cholestasis despite resolution of mechanical obstruction, further evaluation was pursued. Autoimmune markers revealed a negative anti-nuclear antibody (ANA), negative anti mitochondrial antibody (AMA), but a positive anti-smooth muscle antibody (ASMA). Likewise, the serum immunoglobulin IgG was thrice elevated. Intraoperative liver biopsy demonstrated chronic portal inflammation with interface hepatitis, focal lobular hepatitis, and cholestasis, without significant fibrosis or steatohepatitis (Figure 1).
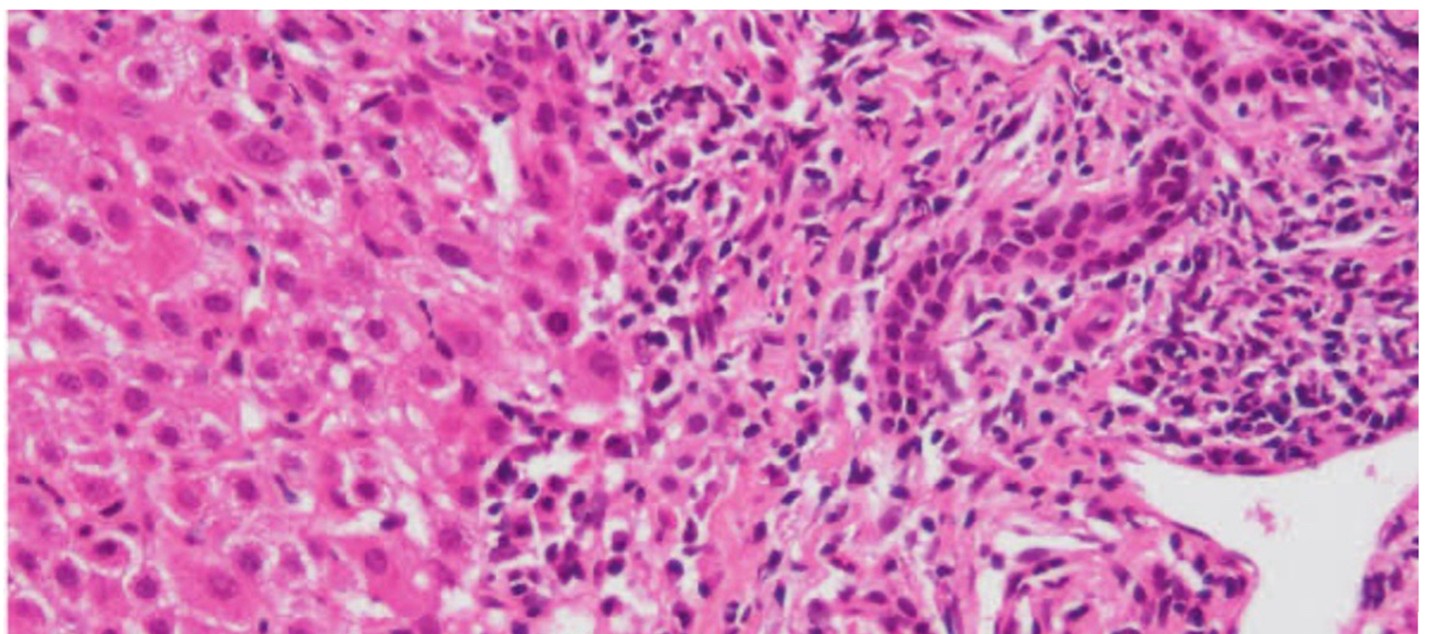
Figure 1. Liver biopsy showed chronic portal and lobular inflammation with cholestasis, confirming the diagnosis of autoimmune hepatitis.
Potential hepatotoxic medications, including Isoniazid were discontinued; however, liver function abnormalities persisted. Likewise, it was learned that his ART were in fact discontinued even 1 month prior to the said admission. A diagnosis of autoimmune hepatitis was made and the patient was subsequently initiated on immunosuppressive therapy with Prednisone and Mycophenolate Mofetil (MMF). Other meds given were ursodeoxycholic acid, and silymarin as a hepatotoprotectants.
While ALT, and AST stabilization were achieved, the hyperbilirubinemia remained persistent, and clinically there was nonresolution of jaundice. Serum ammonia was also monitored and appeared slightly elevated. Patient also became restless. The patient subsequently underwent extracorporeal liver support using double plasma molecular adsorption system (DPMAS) with a bilirubin adsorption cartridge (BS330) combined with ammonia (HA330) hemoperfusion cartridge, resulting in a significant reduction in bilirubin levels (Table1) and improvement in sensorium.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The temporal relationship between clinical events, therapeutic interventions, and biochemical trends is summarized in Figure 2, which demonstrates the progressive rise in bilirubin despite adequate biliary decompression, partial response to immunosuppression, and marked decline following DPMAS therapy. The patient was discharged clinically stable and continued on outpatient immunosuppressive therapy with close follow-up.
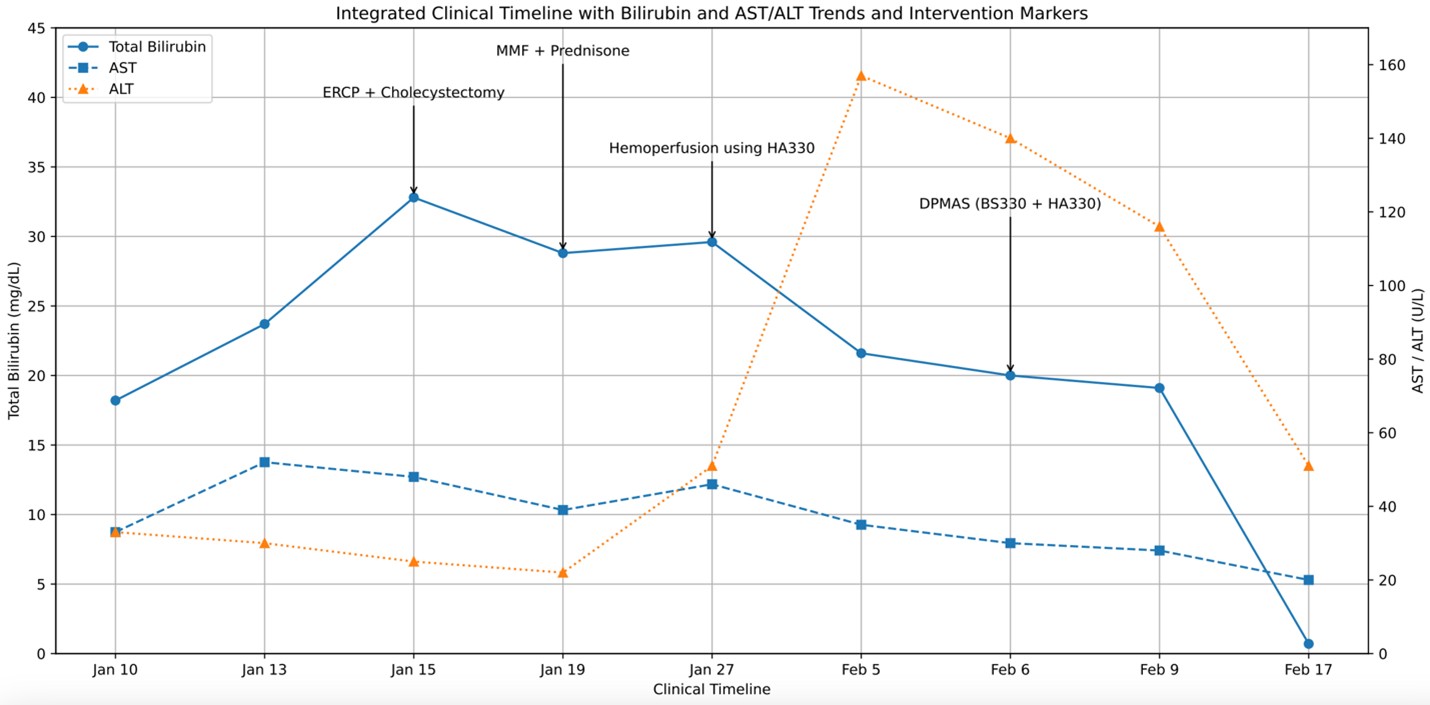
Figure 2. Integrated clinical timeline demonstrating diagnostic evolution, therapeutic interventions, and bilirubin response.
Discussion
This case illustrates a rare and biologically paradoxical coexistence of autoimmune hepatitis (AIH) in a patient with human immunodeficiency virus (HIV) infection. These two conditions are traditionally viewed as occupying opposite ends of the immunologic spectrum. AIH is characterized by loss of immune tolerance with expansion of autoreactive CD4+ and CD8+ lymphocytes, whereas HIV infection leads to progressive immune depletion and dysfunction, particularly affecting CD4+ T cells [8,11]. The concurrence of these conditions therefore challenges conventional paradigms of immune regulation and raises important diagnostic and therapeutic considerations.
Although autoimmune phenomena have been increasingly described in the era of antiretroviral therapy (ART), their overall prevalence in people living with HIV remains low [3]. AIH itself is a rare disease with a global prevalence of approximately 15.65 per 100,000 [1], and its occurrence in HIV-infected individuals is even less frequent, underscoring the uncommon nature of this overlap [4]. This rarity highlights the importance of recognizing such cases, particularly in regions with a rising burden of HIV infection [2].
The pathophysiologic basis for this coexistence remains incompletely understood. One proposed mechanism involves immune reconstitution following initiation of ART, which may unmask latent autoimmune processes. However, accumulating evidence suggests that persistent immune dysregulation despite virologic suppression plays a significant role. This includes aberrant B-cell activation, cytokine imbalance, and impaired regulatory T-cell function, which together may create a permissive environment for autoimmunity even in the setting of chronic immunosuppression [5,10,13]. This duality underscores the complexity of immune homeostasis in HIV and provides a plausible explanation for the paradoxical emergence of AIH.
From a diagnostic standpoint, this case emphasizes the importance of reassessing initial clinical assumptions when the disease course deviates from expected outcomes. The presence of a structural etiology in the form of choledocholithiasis initially provided a unifying explanation for the patient’s presentation. However, the persistence and progression of hyperbilirubinemia despite adequate biliary decompression served as a key turning point that prompted further evaluation. In patients with HIV, the differential diagnosis of liver dysfunction is broad and includes drug-induced liver injury, opportunistic infections, and viral hepatitis coinfections [9]. Consequently, autoimmune etiologies may be under-recognized or diagnosed late.
The diagnosis of AIH in this patient was supported by positive anti-smooth muscle antibodies and characteristic histologic findings of interface hepatitis with portal and lobular inflammation. These findings are consistent with established diagnostic criteria and guidelines for AIH [6–8]. Notably, the biochemical profile demonstrated disproportionately elevated bilirubin levels with relatively mild transaminase elevation, suggesting a cholestatic-predominant presentation. Although less typical, this pattern has been described in AIH and should not preclude consideration of the diagnosis in appropriate clinical contexts.
Therapeutically, management of AIH in patients with HIV remains largely extrapolated from experience in immunocompetent populations due to the absence of disease-specific guidelines. Available reports suggest that standard immunosuppressive therapy for AIH, including corticosteroids and azathioprine, can be effective and generally well tolerated in patients receiving ART [11,12]. Alternatively, for AIH, with or without HIV, evidence suggests that MMF and Tacrolimus can be used as either initial add-on therapy to steroids, or for those with refractory or partial response to azathioprine [14–16]. In this case, immunosuppressive therapy resulted in stabilization of liver enzyme trends without apparent compromise of HIV control, supporting its use with careful monitoring.
A notable feature of this case is the adjunctive use of extracorporeal liver support via double plasma molecular adsorption system (DPMAS) using a bilirubin and ammonia adsorption cartridge. This liver support system is usually used for acute liver failure, or acute on chronic liver failure, regardless of etiology. DPMAS causes rapid removal of bilirubin, ammonia, cytokine and other inflammatory mediators, without requiring exogenous plasma, and continuous intensification of treatment, if necessary, by switching to a new set of adsorption columns [17–19]. While not routinely employed in AIH, its use in this setting highlights a potential role in managing refractory hyperbilirubinemia, particularly in patients with severe cholestasis where conventional therapy alone may be insufficient to produce a fast liver recovery. The observed biochemical improvement suggests that extracorporeal toxin removal may provide a bridging benefit while immunosuppressive therapy addresses the underlying inflammatory process. This can also prevent further liver deterioration culminating into a fulminant liver failure. However, current evidence remains limited, and further studies are needed to strengthen its role in AIH, especially in immunocompromised populations like HIV.
In summary, this case underscores the importance of maintaining a high index of suspicion for autoimmune hepatitis in patients with HIV who present with persistent or unexplained liver dysfunction. The coexistence of these conditions, although rare, reflects a complex interplay between immune deficiency and immune dysregulation. Early recognition and timely initiation of appropriate therapy are essential to improving clinical outcomes.
References
2. Gangcuangco LMA. HIV crisis in the Philippines: urgent actions needed. Lancet Public Health. 2019 Feb;4(2):e84.
3. Ramos-Ruperto L, Busca C, Díez-Vidal A, Robles-Marhuenda Á, Díaz-Almirón M, Mican R, et al. Prevalence and Temporal Trends of Autoimmune Diseases in People Living with HIV. AIDS Res Hum Retroviruses. 2023 Mar;39(3):130–35.
4. Saleem S, Inayat F, Khan AA, Awan JR, Goraya MHN, Hussain A, et al. The demographics of autoimmune hepatitis in human immunodeficiency virus-infected patients: a United States cross-sectional study. Prz Gastroenterol. 2023;18(1):93–9.
5. Chaiteerakij R, Sanpawat A, Avihingsanon A, Treeprasertsuk S. Autoimmune hepatitis in human immunodeficiency virus-infected patients: A case series and review of the literature. World J Gastroenterol. 2019 Sep 21;25(35):5388–402.
6. Czaja AJ. Diagnosis and Management of Autoimmune Hepatitis: Current Status and Future Directions. Gut Liver. 2016 Mar;10(2):177–203.
7. Hennes EM, Zeniya M, Czaja AJ, Parés A, Dalekos GN, Krawitt EL, et al. International Autoimmune Hepatitis Group. Simplified criteria for the diagnosis of autoimmune hepatitis. Hepatology. 2008 Jul;48(1):169–76.
8. Mack CL, Adams D, Assis DN, Kerkar N, Manns MP, Mayo MJ, et al. Diagnosis and Management of Autoimmune Hepatitis in Adults and Children: 2019 Practice Guidance and Guidelines From the American Association for the Study of Liver Diseases. Hepatology. 2020 Aug;72(2):671-722.
9. Puius YA, Dove LM, Brust DG, Shah DP, Lefkowitch JH. Three cases of autoimmune hepatitis in HIV-infected patients. J Clin Gastroenterol. 2008 Apr;42(4):425–9.
10. Rodrigues EM Filho, Fernandes R, Susin R, Fior B. Immune reconstitution inflammatory syndrome as a cause of autoimmune hepatitis and acute liver failure. Rev Bras Ter Intensiva. 2017 Jul-Sep;29(3):382–5.
11. Zoboli F, Ripamonti D, Benatti SV, Comi L, Rizzi M. Autoimmune hepatitis and HIV infection: two case reports and review of the literature. AIDS. 2017 Sep 24;31(15):2172–5.
12. Mubder M, Azab M, Jayaraj M, Cross C, Lankarani D, Dhindsa B, et al. Autoimmune hepatitis in patients with human immunodeficiency virus infection: A systematic review of the published literature. Medicine (Baltimore). 2019 Sep;98(37):e17094.
13. Moy BT, Rezaizadeh H. Immune Reconstitution Inflammatory Syndrome Causing Autoimmune Hepatitis in an HIV-infected Patient Started on Highly Active Antiretroviral Therapy: 819. ACG. 2015 Oct 1;110:S357.
14. Manns MP, Lohse AW, Vergani D. Autoimmune hepatitis--Update 2015. J Hepatol. 2015 Apr;62(1 Suppl):S100–11.
15. Inductivo-Yu I, Adams A, Gish RG, Wakil A, Bzowej NH, Frederick RT, et al. Mycophenolate mofetil in autoimmune hepatitis patients not responsive or intolerant to standard immunosuppressive therapy. Clin Gastroenterol Hepatol. 2007 Jul;5(7):799–802.
16. Ferre-Aracil C, Riveiro-Barciela M, Trapero-Marugán M, Rodríguez-Perálvarez M, Llovet LP, Téllez L, et al. Tacrolimus as an Effective and Durable Second-Line Treatment for Chronic Autoimmune Hepatitis: A Multicentric Study. Dig Dis Sci. 2021 Aug;66(8):2826–32.
17. Tang R, Ji Y, Yao H, Zhou X, Feng D, Cheung EC, et al. Efficacy of DPMAS combined with PE in improving survival outcomes of patients with acute hepatitis E-induced liver failure: A retrospective cohort analysis. ILIVER. 2025 Nov 5;4(4):100203.
18. Nicolas JOM, Inductivo-Yu I, Panlilio MT. Experience on use of Double Plasma Molecular Adsorption System (DPMAS) Among Patients with Liver Failure and Decompensated Liver Cirrhosis in Tertiary Specialty Centers-A Case Series. Acta Scientific Gastrointestinal Disorders. 2024 Sep;7(9):29–34.
19. Chen Y, Han T, Duan Z; Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Clinical application of artificial liver and blood purification: expert consensus recommendations. Hepatol Int. 2023 Feb;17(1):4–17.
