Abstract
Background: Adolescents and young adults living with HIV often face adherence challenges due to psychosocial, developmental, and structural barriers. This study examined whether the mode of HIV transmission—vertical vs. horizontal—impacts adherence or viral suppression in youth receiving long-acting cabotegravir–rilpivirine within a specialty pharmacy-supported pediatric/adolescent HIV outpatient clinic.
Methods: A retrospective, cohort analysis was conducted on patients aged 13–23 who received at least six bimonthly cabotegravir-rilpivirine injections from October 1, 2022, to October 1, 2024. Patients were classified by transmission mode and monitored for adherence and viral load. Adherence was defined as receiving each injection within ±7 days of the 56-day treatment interval. Patients who switched to monthly dosing, changed therapy due to drug interactions, or became pregnant were excluded.
Results: A total of 45 patients met inclusion criteria (14 vertically acquired, 31 horizontally acquired). Adherence was 100% in the vertical group and 99% in the horizontal group (p = 0.31). All patients established and/or maintained viral suppression (HIV-1 RNA <200 copies/mL). However, mean viral load differed significantly: 128.1 copies/mL in the vertical group vs. 1.8 copies/mL in the horizontal group (p = 0.01), despite both being within suppressed ranges.
Conclusions: High adherence and virologic suppression were observed in both groups, with no clinical difference in transmission mode. Specialty pharmacy care may play a critical role in supporting long-acting injectable antiretroviral therapy (LAI – ART) success in youth living with HIV.
Keywords
HIV, Adherence, Youth, Specialty pharmacy
Introduction
The treatment and long-term management of HIV differ significantly depending on whether the infection is acquired vertically—transmitted from mother to child during pregnancy, childbirth, or breastfeeding, or horizontally—through sexual contact or exposure to infected blood. Vertically acquired HIV presents unique clinical, immunological, and psychosocial challenges that are distinct from those encountered in horizontally acquired cases [1]. Individuals living with vertically transmitted HIV often begin ART in infancy or early childhood, leading to prolonged drug exposure, higher risks of resistance, and cumulative toxicities over time. Furthermore, the developing immune system in children responds differently to both HIV and ART, complicating treatment strategies [2]. These individuals also face the burden of navigating chronic illness from birth, which can impact adherence, mental health, and quality of life throughout adolescence and adulthood. In contrast, horizontally acquired HIV typically occurs after the immune system has matured and often in individuals with greater autonomy and access to health education, influencing both the clinical progression and management of the disease. Addressing the disparities in outcomes between these two populations is critical for optimizing care models and informing tailored therapeutic strategies.
In recent years, LAI-ART has emerged as a promising alternative to daily oral regimens for people living with HIV. This formulation, typically administered monthly or bimonthly, offers several potential advantages, including improved adherence, reduced pill fatigue, and decreased stigma associated with daily medication use. As a result, LAI-ART is increasingly being adopted in both clinical practice and global treatment guidelines, particularly for individuals who struggle with consistent oral adherence However, despite growing enthusiasm and uptake, current research on the effectiveness of LAI-ART has primarily focused on adults living with horizontally acquired HIV, often excluding adolescent and youth populations, particularly individuals living with vertically acquired HIV infection. To date, no studies have comprehensively examined whether mode of HIV acquisition impacts the pharmacologic, immunologic, or virologic outcomes associated with LAI-ART. This gap in the literature underscores the need to investigate how the unique clinical trajectories of vertically infected individuals may influence the safety and efficacy of long-acting therapies.
Previous studies have demonstrated the effectiveness of specialty pharmacy interventions in improving medication adherence among individuals living with chronic and complex conditions such as HIV [4]. These interventions often include coordinated care models involving medication therapy management, adherence monitoring, refill synchronization, patient education, and ongoing clinical support—services that have shown particular benefit in enhancing adherence to self-administered medications, whether oral or injectable. However, the existing body of literature is largely limited to medications that patients administer independently in the home setting. There remains a significant gap in research examining the role of specialty pharmacies in supporting adherence to regimens such as LAI-ART like cabotegravir-rilpivirine (Cabenuva) (ViiV Healthcare, London, UK), which are administered in clinical settings by healthcare professionals. As LAI-ART options become more widely available and integrated into HIV treatment paradigms, it is essential to explore whether and how specialty pharmacy services can be effectively leveraged to support adherence and persistence in this context. Understanding this role is particularly important given the logistical, structural, and behavioral adherence challenges unique to clinic-based administration schedules, and it may represent an important evolution in specialty pharmacy practice.
Building upon the established evidence supporting specialty pharmacy interventions in enhancing adherence to self-administered ARTs, this study aims to address significant gaps in understanding adherence dynamics and clinical outcomes associated with clinic-administered LAI-ART. The primary objective is to assess the influence of HIV mode of transmission on both medication adherence and viral suppression among patients receiving Cabenuva, thereby identifying potential demographic or behavioral factors that may differentially impact treatment success within these two cohorts. The secondary objective focuses on exploring the applicability and potential adaptation of specialty pharmacy adherence support mechanisms in the context of maintaining patient engagement and retention in clinic-based injection appointments. Through this dual focus, the study seeks to generate actionable insights that could guide the evolution of specialty pharmacy practice, optimizing adherence support frameworks to meet the unique challenges posed by clinic-administered HIV therapies and ultimately improving long-term patient outcomes.
Methods
Study design
This retrospective cohort study was conducted at a pediatric and young adult hospital-based outpatient infectious disease clinic with specialty pharmacy support, evaluating patients between October 1, 2022 and October 1, 2024. The study included patients aged 13 to 23 years living with HIV who were receiving injectable cabotegravir–rilpivirine. Patients were stratified into two groups based on mode of HIV acquisition: vertically acquired HIV and horizontally acquired HIV. Inclusion criteria required patients to have received at least six injections during the study period, aligning with the long-acting dosing schedule and representing a minimum of 50% adherence. Although Cabenuva is approved for both monthly and every-two-month administration, this study focused exclusively on the every-two-month injection cycle. Patients who switched to monthly dosing at any point during the study period were excluded. Additional exclusion criteria were any patient that switched to a different ART regimen due to the initiation of a drug interaction with Cabenuva, as well as pregnancy, due to insufficient human data on the use of Cabenuva in pregnant patients.
Specialty pharmacy services overview
All patients in this study were managed through a comprehensive health system specialty pharmacy care model designed to support the clinical and logistical complexities associated with LAI-ART. This multidisciplinary model integrated clinical pharmacy oversight with operational support from pharmacy technicians and close coordination with HIV care providers.
One key feature of the model was proactive adherence support. Pharmacy staff conducted pre-visit outreach as standard of care within the specialty pharmacy model to remind patients of upcoming injection appointments, verify clinic availability, and confirm medication readiness. Insurance authorizations and medication procurement were handled in advance to avoid delays. Pharmacy personnel also worked directly with providers to reschedule missed appointments promptly and ensure dosing remained within the clinically appropriate window (±7 days from the scheduled 56-day interval).
Documentation of injection administration in the electronic medical record (EMR) automatically triggered a follow-up workflow task for pharmacy technicians. Within 24 hours of each injection, technicians contacted every patient to assess for any adverse reactions. If a patient reported symptoms such as injection site pain, flu-like illness, or other concerns, the technician escalated the call to a clinical pharmacist. Pharmacists then provided tailored counseling, side effect management, and clinical triage as needed, ensuring timely intervention and preventing unnecessary treatment discontinuation. A summary of pharmacist and technician duties is shown in Table 1.
In addition to adverse event monitoring, pharmacists routinely reviewed adherence data and viral load trends and reported the information to the clinic providers. They collaborated with the healthcare team to address clinical flags such as elevated viral load, potential drug interactions, or changes in life circumstances (e.g., pregnancy, housing instability) that could impact medication adherence or safety. For patients requiring a switch to monthly dosing or those facing temporary barriers to injectable therapy, pharmacists assisted in navigating formulary options and ensuring uninterrupted antiretroviral coverage.
|
Role |
Intervention Type |
Description |
Purpose |
|
Pharmacist |
Pre-injection Clinical Review |
Assessed for drug interactions, contraindications (e.g., pregnancy), and dosing appropriateness |
Ensure safe and effective use of Cabenuva |
|
Pharmacist |
Adherence Monitoring |
Tracked injection dates and ensured doses were received within the ±7-day window |
Maintain therapeutic levels and prevent viral rebound |
|
Pharmacist |
Side Effect Management |
Provided counseling and clinical recommendations for patients reporting post-injection symptoms |
Address adverse events and reduce risk of therapy discontinuation |
|
Pharmacist |
Therapy Modification Support |
Assisted with transitions to monthly dosing or alternate regimens when clinically indicated |
Maintain continuity of ART in response to changing needs |
|
Pharmacist |
Insurance Navigation & Access Facilitation |
Worked with payers to obtain prior authorizations and resolve delays in medication availability |
Prevent lapses in medication access |
|
Pharmacist |
Provider Collaboration |
Communicated with prescribers regarding adherence concerns, clinical flags, or therapy changes |
Enable coordinated care and timely clinical decision-making |
|
Pharmacist |
Patient Education |
Offered counseling on injection timing, expectations, storage, and travel considerations |
Promote informed and confident participation in care |
|
Pharmacy Technician |
24-Hour Post-Injection Follow-Up |
Contacted all patients within 24 hours of injection to assess for side effects |
Early identification of adverse events; escalation to pharmacist |
|
Pharmacy Technician |
Workflow Task Management |
Completed EMR-generated follow-up tasks triggered by documentation of injection |
Ensure timely and consistent patient outreach |
|
Pharmacy Technician |
Scheduling Coordination |
Assisted with aligning pharmacy and clinic schedules for next injection |
Reduce risk of missed or delayed doses |
|
Pharmacy Technician |
Appointment Reminders |
Conducted reminder calls or texts for upcoming injection visits |
Improve adherence and reduce no-show rates |
|
Pharmacy Technician |
Benefits Verification Support |
Gathered insurance information and assisted pharmacists with authorization prep |
Streamline access to medication |
|
Pharmacy Technician |
Escalation Protocol Execution |
Transferred calls involving clinical concerns to the pharmacist |
Ensure patients receive appropriate clinical guidance |
|
Pharmacy Technician |
Documentation & Reporting |
Recorded follow-up outcomes and adverse event reports in the EMR |
Maintain accurate records and support pharmacist decision-making |
Outcome measures
The primary outcomes assessed were medication adherence and viral suppression. Adherence was measured by evaluating the timing of each injection in relation to the Target Treatment Date (TTD), defined as every 56 days following the second dose of Cabenuva. Patients were considered adherent for an injection if it was administered within a 14-day window surrounding the TTD—specifically, 7 days before or 7 days after the scheduled date. Injections administered more than 7 days after the TTD were classified as missed and considered non-adherent and non-compliant. Patients were required to have received at least six documented injections to be included in the analysis. Viral suppression was defined as a plasma HIV-1 RNA level <200 copies/mL, consistent with U.S. Department of Health and Human Services (DHHS) guidelines [5]. In addition to suppression status, mean viral load was analyzed to assess differences in residual viremia between vertically and horizontally infected patients.
Data collection and analysis
Clinical and pharmacy data were extracted from the electronic medical record system. Data included age, sex, HIV transmission mode, injection dates, pharmacist interventions, and laboratory results. Group comparisons for adherence and viral load were performed using appropriate statistical tests. A p-value <0.05 was considered statistically significant. All analyses were conducted using R (Version 4.4.2).
Results
A total of 45 patients (Table 2) met inclusion criteria, comprising 14 with vertically acquired HIV and 31 with horizontally acquired HIV. All participants received Cabenuva injections every two months and engaged with specialty pharmacy services throughout the study period.
|
Characteristic |
All Patients (n=45) |
Mode of HIV Transmission |
|
|
Vertical (n=14) |
Horizontal (n=31) |
||
|
Age |
|||
|
Mean, y (SD) |
19.20 (2.4) |
18.96 (2.8) |
19.98 (2.0) |
|
Median, y |
20 |
20 |
20 |
|
Minimum, y |
13 |
13 |
15 |
|
Maximum, y |
23 |
21 |
23 |
|
Sex |
|||
|
Male at Birth, % |
77.8 |
43.0 |
93.5 |
|
Race |
|||
|
Black, % |
88.9 |
85.7 |
98.97 |
|
White, % |
11.1 |
14.3 |
1.0 |
|
Other, % |
0.02 |
0 |
0.03 |
|
SD: Standard Deviation |
|||
Adherence to the injection schedule was high across both groups. Among those with vertically acquired HIV, 100% of injections (n=141 injections) were administered within the defined adherence window (±7 days of the 56-day Target Treatment Date) (Figure 1). In the horizontally acquired group, 99% of injections (n=334 injections) met adherence criteria (Figure 2). There was no statistically significant difference in adherence rates between the two groups (p = 0.31).
Viral suppression was established and/or maintained in 100% of patients in both groups, defined as HIV-1 RNA <200 copies/mL. While all patients ultimately reached undetectable viral loads, a statistically significant difference in mean viral load was observed between groups: the vertically infected group had a mean of 128.1 copies/mL, compared to 1.8 copies/mL in the horizontally infected group (p = 0.01).
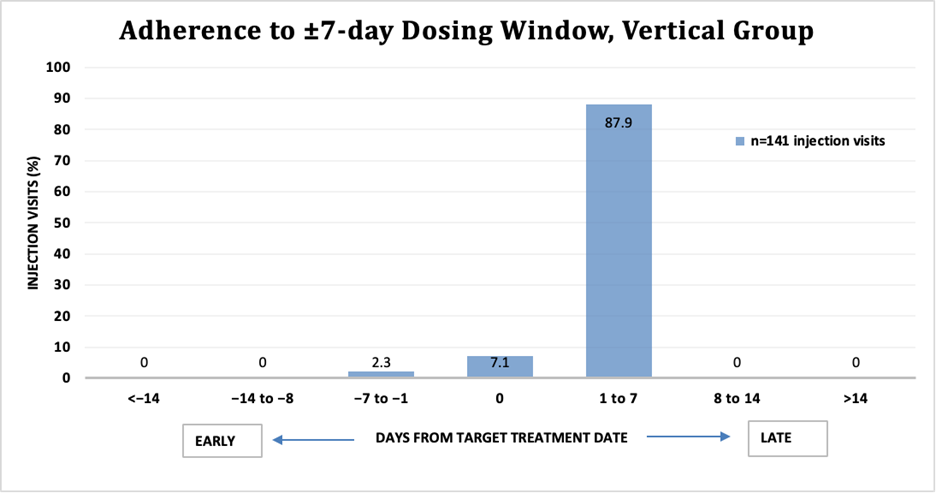
Figure 1. Adherence to ±7-day dosing window, vertical group.
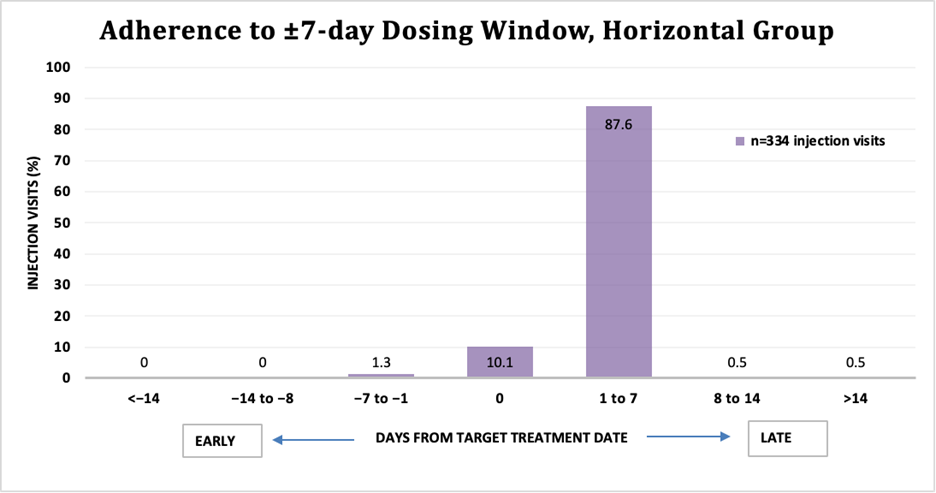
Figure 2. Adherence to ±7-day dosing window, horizontal group.
Discussion
This retrospective review evaluated whether mode of HIV transmission—vertical versus horizontal—influences adherence and viral suppression outcomes among youth receiving Cabenuva injections in the context of pharmacist-led specialty care. The findings demonstrate that high adherence and complete viral suppression were sustained in both groups over a two-year period, suggesting that transmission mode does not negatively impact treatment engagement or virologic outcomes when robust pharmacy support systems are in place.
As part of standard of care for this clinic, all patients were discussed in a multidisciplinary team meeting to review historical genotypes, viral load suppression history and adherence to appointments prior to switching from oral antiretrovirals to a long-acting injectable regimen. Although adherence rates were similarly high, mean viral load differed significantly by mode of transmission. Patients with vertically acquired HIV demonstrated higher mean viral loads despite remaining below the threshold for virologic failure. This discrepancy may reflect long-term virologic and immunologic consequences of early-life HIV infection, including immune exhaustion, persistent viral reservoirs, and prior ART exposure or resistance patterns not detected by historical genotypes. However, the clinical relevance of this difference is uncertain given that all patients ultimately attained undetectable viral loads. The FDA-approved indication for Cabenuva is intended for patients who are already virologically suppressed prior to initiating therapy. In our study, the majority of patients met this criterion; however, there were a subset of individuals who were not virologically suppressed at baseline due to poor adherence to their oral ART. In these cases, clinicians opted to initiate Cabenuva despite the lack of viral suppression, based on the clinical judgment that LAI-ART would offer improved adherence and ultimately better virologic outcomes. While this may have slightly skewed the mean viral load data, these instances reflect real-world decision making and illustrate how Cabenuva may be used in clinical practice beyond its current FDA-approved indication. This divergence underscores the importance of evaluating the drug’s performance not only in controlled trial settings but also in pragmatic, real-world contexts.
The integration of specialty pharmacy services likely contributed to the uniformly high adherence observed. Pharmacist-led interventions, such as coordination of injection appointments, proactive adherence monitoring, and patient-specific counseling, may help mitigate barriers traditionally faced by youth living with HIV, including stigma, fluctuating motivation, and logistical challenges. These findings support the expanding role of clinical pharmacists in HIV care, particularly in managing LAI-ART regimens among adolescent and young adult populations.
There are several limitations to this study. The sample size was modest, particularly in the vertically infected subgroup, which may limit generalizability. Next, the retrospective design may introduce selection bias, and exclusion of patients who switched to monthly dosing or alternative regimens may underestimate challenges faced in broader clinical practice. Additionally, while adherence was assessed using well-defined criteria based on injection timing, other dimensions of adherence, such as psychosocial factors or patient-reported outcomes, were not captured. Finally, our specialty pharmacy is embedded within a hospital system, allowing for closer collaboration and communication with providers. This integrated model may have contributed to improved outcomes. Independent specialty pharmacies, operating separately from clinical care teams, may find it more difficult to replicate these results. Therefore, the unique structure of our institution represents a limitation in terms of broader applicability.
Finally, there is notable variability in how virologic suppression is defined across scientific literature and clinical trials. While this study utilized the DHHS guideline of <200 copies/mL to define virologic suppression, other clinical trials often use more stringent thresholds. Notably, Cabenuva’s own prescribing information defines virologic suppression as <50 copies/mL [6]. This discrepancy in definitions presents a potential limitation of our study, particularly when interpreting clinical outcomes. For example, the mean viral load among patients with vertically acquired HIV in our cohort was 128.1 copies/mL—technically considered suppressed under the <200 threshold but not under the more stringent <50 definition. Had we applied the <50 copies/mL cutoff, as used in Cabenuva’s clinical trials and product labeling, the interpretation of our results may have differed significantly from a clinical perspective. This highlights the importance of consistent and clinically relevant viral load thresholds when evaluating treatment efficacy.
Despite these limitations, this study provides real-world evidence that transmission mode does not adversely affect adherence or suppression outcomes in youth receiving LAI-ART under structured pharmacy support. Future studies should investigate the long-term durability of these outcomes and explore interventions for patients who may require greater flexibility in ART delivery.
Conclusion
In this study of youth living with HIV receiving long-acting cabotegravir–rilpivirine, mode of HIV transmission—vertical versus horizontal—was not associated with differences in adherence or viral suppression when pharmacist-led specialty care was provided. Despite a statistically significant difference in mean viral load between groups, all patients maintained virologic suppression throughout the study period. These findings highlight the effectiveness of long-acting ART in achieving consistent treatment outcomes across diverse patient backgrounds and underscore the value of pharmacist-driven support in optimizing adherence among adolescents and young adults. Continued integration of specialty pharmacy services may be key to sustaining long-term success with injectable HIV therapy in this population.
Conflicts of Interest and Source of Funding
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. This work was supported in part by the American Lebanese Syrian Associated Charities (ALSAC). No additional external funding was received for the study.
Acknowledgement
We thank Cherise Guess, PhD, ELS for her helpful comments and suggestions in the preparation of this manuscript.
References
2. Ásbjörnsdóttir KH, Hughes JP, Wamalwa D, Langat A, Slyker JA, Okinyi HM, et al. Differences in virologic and immunologic response to antiretroviral therapy among HIV-1-infected infants and children. AIDS. 2016 Nov 28;30(18):2835–43.
3. Nguyen N, Lane B, Golub SA, Chastain C, Zucker J, King K, et al. Long-acting injectable ART to advance health equity: a descriptive analysis of US clinic perspectives on barriers, needed support and programme goals for implementation from applications to the ALAI UP Project. J Int AIDS Soc. 2024 Jul;27 Suppl 1(Suppl 1):e26282.
4. St James GJ, Duckworth DL, Bochenek SH, Rhudy C, Zeltner M, Tagavi AB, et al. Effect on medication adherence of applying a specialty pharmacy care model to nonspecialty medications: A quasi-experimental cohort study. Am J Health Syst Pharm. 2023 Nov 23;80(Suppl 4):S135–42.
5. Guidelines for the Use of Antiretroviral Therapy Agents in Adults and Adolescents with HIV. [Clinicalinfo.HIV.Gov website]. September 12, 2024 (Accessed August 23, 2025). Available at: https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-arv/whats-new.
6. Almazyad AF, Haq N, Alsarra IA, Alshehri S, Alam P, Imran M, et al. Novel Drug Delivery Systems Approved by USFDA from 2019 to 2023: A Comprehensive Review. Curr. Drug Deliv. 2025 Sep 1.
