Abstract
Introduction: Hemodialysis patients as well as healthcare workers are considered to be in a high-risk category for SARS-CoV-2 infection and a priority for vaccination.
Methods: In a single-center out-patient hemodialysis unit, 46 healthcare workers and 216 patients were vaccinated simultaneously with BNT162b2 (BioNTech-Pfizer) vaccine. They received two doses, 21 days apart. The primary objectives were to evaluate the safety and efficacy of the vaccine.
Findings: There were no major adverse events in either group. Lymphadenopathy was reported by some health workers. All (100%) individuals in the healthcare workers group developed a positive antibody response (anti-S IgG) after the second dose compared with 91.7% of patients. Among patients there was a significant negative correlation between anti-S levels and age after both, the first dose (R= -0.176, p=0.01) and the second dose (R= -0.193, p=0.005); there was also a significant negative correlation between anti-S and Charlson Comorbidity Index adjusted for age after both, the first dose (R= -0.150, p=0.028) and the second dose (R= -0.163, p=0.018). Finally, a negative correlation between anti-S and Body Mass Index was found after the first dose (R= -0.140, p=0.04). No correlations were found with dialysis vintage, Kt/V, or diabetes.
Conclusions: Following vaccination, patients had a significantly lower anti-S response than healthcare workers. Age, Charlson Comorbidity Index and Body Mass Index negatively impacted the humoral response. However, with more than 91% response we believe that vaccination can be recommended strongly in the hemodialysis population.
Introduction
In January 2020, the World Health Organization (WHO) classified COVID-19 to be a Public Health Emergency of International Concern and declared it a pandemic on March 11, 2020 [1]. Over one hundred and eighty-five million people have been infected by SARS-CoV-2 and roughly four million have died worldwide so far [2]. In addition, the economic and social impact of the pandemic has been extensive [3,4].
Although the range of effective therapeutics is broadening, vaccination has emerged as the best solution to ameliorate the severity of the disease and to achieve community immunity. A successfully implemented global vaccination program appears to offer the best chance for society to return to its pre-pandemic state. Under normal circumstances, the development of a new vaccine, takes years. mRNA vaccines targeting SARS-CoV-2 became available with unprecedented speed [5], within a few months of the genomic identification of the virus [6].
Patients receiving in-center hemodialysis (ICHD) require regular travel to clinics in order to have their treatment. This exposure leaves them at increased risk of SARS-CoV-2 infection compared to people with other co-morbidities who can isolate at home during regional outbreaks. Furthermore, ICHD patients have a reported 16% to 32% case fatality rate if they contract COVID-19 infection [7].
In January 2021 the Portuguese health authority declared hemodialysis patients as well as healthcare workers to be in a high-risk category for infection and a priority for SARSCoV- 2 vaccination. This presented a unique opportunity to investigate the outcomes of a real-life program of simultaneous vaccination of all patients and healthcare workers in an ICHD unit.
This study aimed to evaluate the antibody response following a mRNA COVID-19 vaccination program in an ICHD population. This is one of the first reports on this topic and, to the best of our knowledge, the largest study reported so far [8].
Methods
Study design and population
This study was carried out in a single DaVita hemodialysis unit in Leiria, Portugal. The study adhered to the Declaration of Helsinki and was approved by the Portuguese Research DaVita Ethics Committee. All participants gave informed written consent.
Two hundred and sixteen adult ICHD patients and fortysix adult healthcare workers (Table 1) in our clinic received the first deltoid muscle injection of the BioNTech/Pfizer vaccine (30 μg, 0.3 ml volume per dose) on January 27th, 28th and 29th with the second dose administered exactly 21 days after the first.
Twenty-one days after each of the first and second doses, blood samples were collected to evaluate antibody response and to exclude SARS-CoV-2 infection. All patients and healthcare workers were under the clinical care of the authors of this manuscript throughout the study period.
| Dialysis Patients (N=216) | Health Workers (N=46) | |
| Age (years) | 72 (34-91) | 52 (23-77) |
| Gender, men (%) | 62.5 | 15.2 |
| Diabetes mellitus (%) | 41.2 | 2.17 |
| Dialysis vintage in months | 46.5 (1-456) | |
| Kt/V | 1.90 (1.1-2.8) | |
| Charlson Comorbidity Index | 5 (2-14) | |
| BMI (Kg/m2) | 24.9 (12.8-39.3) | 26.0 (18.9-35.4) |
| Smoker (%) | 5.6 | 10.9 |
| Under ARB or ACEi treatment (%) | 38.4 | 10.9 |
| Abbreviations: ACEi: Angiotensin Converting Enzyme inhibitors; ARB: Angiotensin Receptor Blocker; BMI: Body Mass Index. Values are expressed in median (range). |
||
Vaccination safety assessments
The clinical criteria for vaccination safety used by BioNTech/Pfizer in their phase 3 clinical trial were followed [9].
All participants were observed for any acute reactions for 30 minutes after each dose of vaccine. For 7 days after each dose all participants were monitored for specific local or systemic adverse events:
Local (injection-site) reactions: Pain: mild, does not interfere with activity; moderate, interferes with activity; severe, prevents daily activity. Redness and Swelling: mild, 2.0 to 5.0 cm in diameter; moderate, >5.0 to 10.0 cm in diameter; severe, >10.0 cm in diameter.
Systemic events: Fever: mild, 38.0 to 38.4°C; moderate, 38.4 to 38.9°C; severe, >38.9 to 40.0°C. Chills, Fatigue, Headache, new or worsened Muscle Pain, new or worsened Joint Pain: mild, does not interfere with activity; moderate, some interference with activity; severe, prevents daily activity. Vomiting: mild, 1-2 times in 24 hours; moderate >2 times in 24 hours; severe, requires intravenous hydration. Diarrhea: mild, 2 to 3 loose stools in 24 hours; moderate, 4 to 5 loose stools in 24 hours; severe, 6 or more loose stools in 24 hours. For all events, emergency, indicated department visit or hospitalization.
Vaccination efficacy assessments
The primary end point was the efficacy of the vaccine on inducing antibody response 21 days after each dose. Also, the occurrence of confirmed COVID-19 with onset at least 7 days after each dose. Patients and healthcare workers were analyzed separately, each in two groups: those who had evidence of prior infection and those who had not.
Laboratory studies
Serum tubes (BD Vacutainer) were used for blood sampling. Samples were transported to the laboratory at 2-8°C within 2 hours. On arrival, specimens were centrifuged at 1300g (RCF) for 10 minutes according to the manufacturers’ instructions, aliquoted and stored at -20°C.
All samples were tested for the presence of IgG antibodies against SARS-CoV-2 with two commercially available serological assays: the Elecsys® Anti-SARSCoV- 2 electrochemiluminescence immunoassay (ECLIA), from Roche Diagnostics, measuring IgG against SARSCoV- 2 N-protein, and the Atellica® IM SARS-CoV-2 IgG chemiluminescence assay (CLIA), from Siemens Healthineers, measuring IgG against SARS-CoV-2 S-protein. To ensure that all participants were correctly allocated to the with and without previous SARS-CoV-2 infection groups, anti-N antibody tests were performed 21 days after each vaccine dose.
SARS-CoV-2 IgG titers (index) were calculated automatically by the immunoassay analyzers on the basis of relative light units (RLU), because the viral antibody titer is positively associated with RLU. According to both manufacturers’ instructions, the cut-off value for a positive SARSCoV-2 IgG result is 1.0 (Index Value). All laboratory tests for this study were performed by the same doctor.
Statistical analysis
Descriptive analysis is presented as median (range). We used Kolmogorov-Smirnov and Shapiro Wilk tests to evaluate for normal distribution in continuous variables. We used Wilcoxon tests to compare continuous paired variable and the Chi-square test to compare categorical variables. Logistic regression was used to evaluate the association of continuous variables with serologic response.
We used p<0.05 to indicate statistical significance. All statistical analysis were performed using IBM SPSS Statistics 27.0.
Results
In the analyzed described population, the following results are reported.
Vaccination safety
Acute reactions after 30 minutes of each vaccine dose: No participants had any acute reaction and all left the clinic after vaccination without event.
Local (injection-site) reactions within 7 days after each dose: Mild pain was the most commonly reported local reaction among patients (31% after the first dose and 38% after the second dose) as well as among healthcare workers (63% after the first dose and 11.6% after the second dose). In both cases this resolved within 1 to 2 days without requiring medication.
Systemic reactions within 7 days after each dose: Systemic reactions were reported more often by healthcare workers than by patients; in both groups, they were more prevalent after the second dose. Headache (8.4% in patients and 26% in healthcare workers), mild muscle pain (12.6% in patients and 39.1% in healthcare workers) and fatigue (8.4% in patients and 34.5% in healthcare workers) were the most commonly reported events (Figure 1).
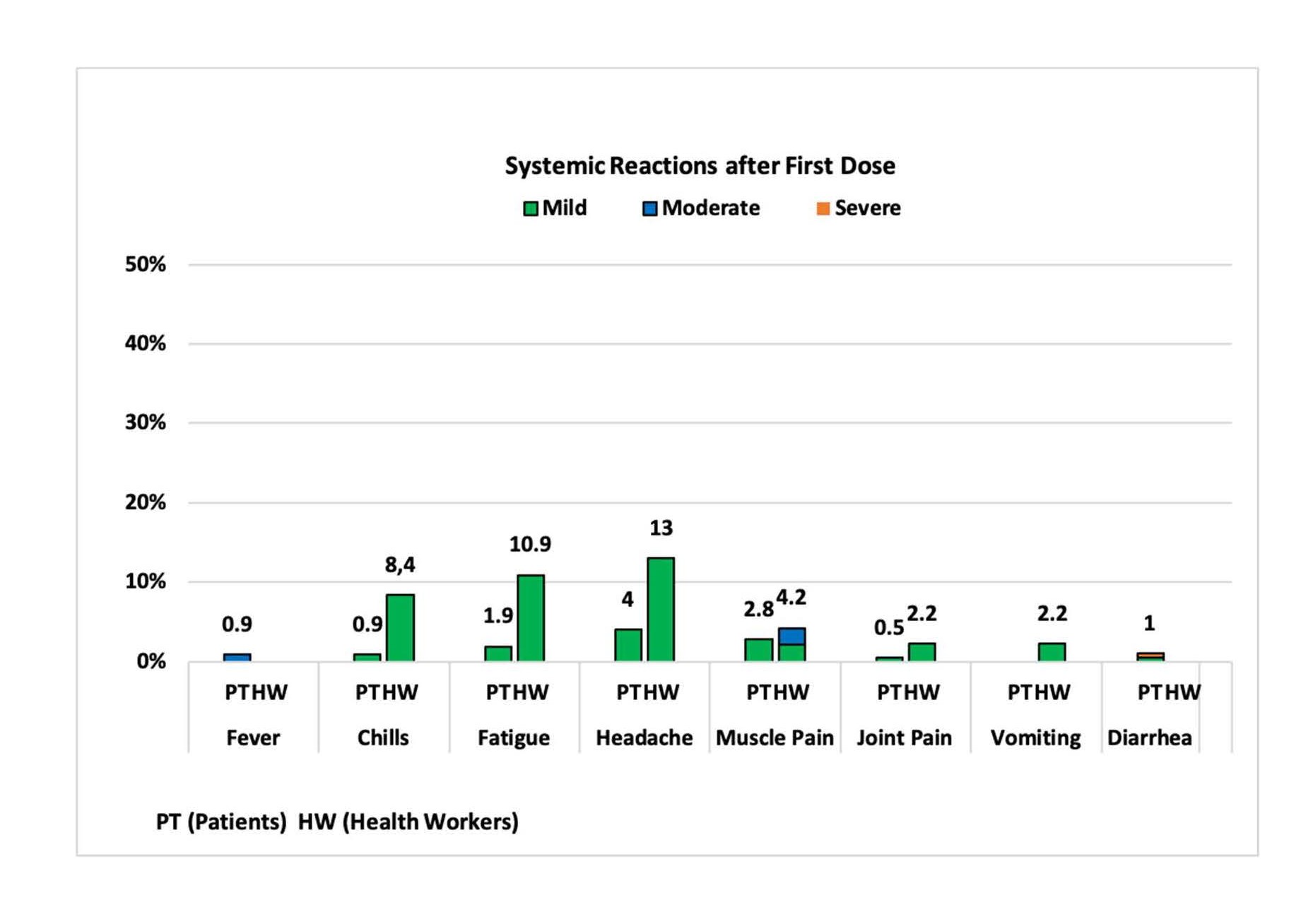
Figure 1A. Percentage of participants with systemic reactions after first dose.
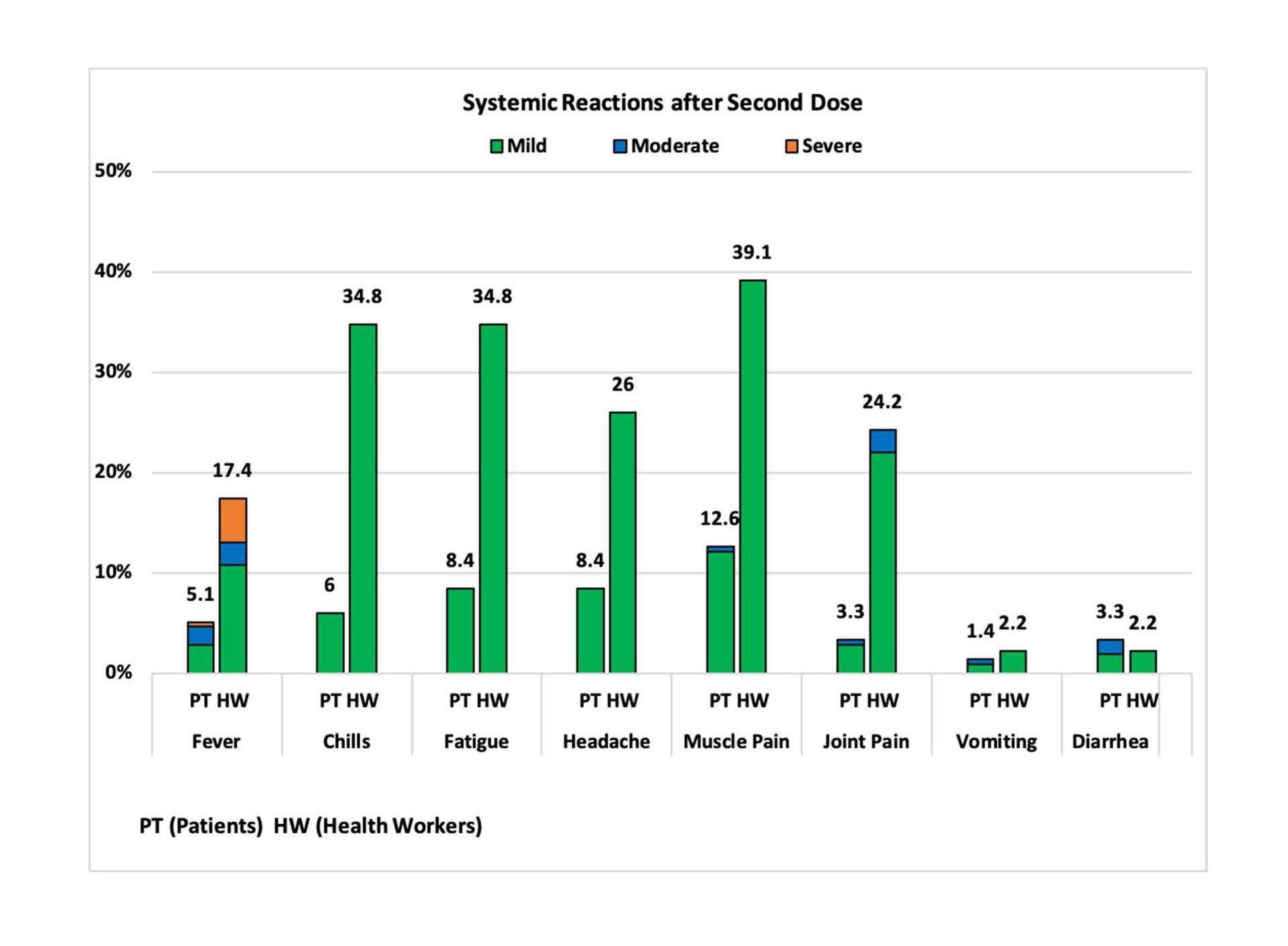
Figure 1B. Percentage of participants with systemic reactions after second dose.
Serious adverse events: None of the patients reported lymphadenopathy along the study period but 8 healthcare workers (1 male and 7 females) reported ipsilateral axillary lymphadenopathy: one female 8 days after the first dose and the others after the second dose. In 2 females it lasted for 3 weeks; in the others it resolved within 1 week. Lymphadenopathy, is likely to have resulted from a robust vaccine-elicited immune response [9].
One patient, 82 years old with Charlson Comorbidity Index of 10, died suddenly at home, eight days after the second dose. This fatality was not considered to be related to vaccination.
Two other patients missed the antibody tests 21 days after the second dose because they were hospitalized for reasons unrelated to vaccination. They remained RT-PCR negative for SARS-CoV-2.
Vaccination efficacy
One patient, arrived for the regular dialysis session on day 21st after the first dose, with fever and mild respiratory symptoms. A nasopharyngeal swab was RT-PCR positive for SARS-CoV-2. The second dose of vaccine was not administered and the patient was dialyzed in a dedicated room for SARS-CoV-2 positive patients. That day, blood tests for anti-N and anti-S were negative, meaning that the patient had not responded to the first dose of vaccine. He did not need hospitalization and recovered completely. No other participant became infected with SARS-CoV-2 along the study period.
IgG anti-N response
All participants considered to be without previous infection remained negative for anti-N throughout the study, except for one patient who become positive for anti-N 21 days after the second vaccine dose. He had no symptoms of SARS-CoV-2 infection and had been anti-N negative 21 days after the first vaccine dose. It is presumed that he contracted SARS-CoV-2 infection via asymptomatic spread in the period between the two anti-N tests.
All patients considered to have had previous infection had positive anti-N in both determinations except for one patient with previous symptomatic and RT-PCR positive SARS-CoV-2 infection who was anti-N negative 21 days after both the first and second vaccine doses. He was anti-S positive after both doses.
There were no statistically significant differences between the levels of antibody anti-N after the first and second doses in all participants, when the groups with or without previous SARS-CoV-2 infection were analyzed separately.
IgG anti-S response
One of the primary end points was to evaluate how participants responded to vaccination through anti-S levels and the factors which may have influenced the magnitude of the responses. Patients and healthcare workers anti-S response to each dose of vaccination are presented in Table 2.
| Anti-S level (relative light units) | No. and % Reactive | |||||
| N | 1st dose | 2nd dose | 1st dose | 2nd dose | ||
| Patients | Median (range) | Median (range) | N (%) | N (%) | ||
| With previous SARS-CoV-2 | 12 | 567.3 (1.3- 3029.2) |
768.9 (22.2- 3983.7) |
p=0.01 | 12 (100%) | 12 (100%) |
| Without previous SARS-CoV-2 | 204 | 0.5 (0.5-17.9) | 34.2 (0.5-487.5) | p<0.001 | 73 (35.8%) | 187 (91.7%) |
| Age ≤55 | 33 | 0.5 (1.0-10.8) | 69.9 (0.5-295.8) | p<0.001 | 19 (57.6%) | 32 (97.0%) |
| Age >55-65 | 34 | 0.5 (0.5-9.5) | 54.8 (0.5-210.7) | p<0.001 | 11 (32.4%) | 29 (85.3%) |
| Age >65-75 | 61 | 0.6 (0.5-17.9) | 39.1 (0.8-487.5) | p<0.001 | 25 (41.0%) | 59 (96.7%) |
| Age >75 | 76 | 0.5 (0.5-11.8) | 23.2 (0.5-148.9) | p<0.001 | 18 (23.7%) | 67 (88.2%) |
| BMI ≥30 | 41 | 0.5 (0.5-8.3) | 17.8 (0.5-138.0) | p<0.001 | 10 (24.4%) | 38 (92.7%) |
| Healthcare Workers | ||||||
| With previous SARS-CoV-2 | 5 | 166.9 (20.2- 210.5) |
194.2 (90.2- 245.6) |
p=0.08 | 5 (100%) | 5 (100%) |
| Without previous SARS-CoV-2 | 41 | 9.3 (0.5-79.2) | 146.3 (18.0- 647.3) |
p<0.001 | 38 (97.0%) | 41 (100%) |
| Age ≤55 | 26 | 14.0 (0.6-79.2) | 163.4 (33.5-647.3) | P<0.001 | 25 (96.2%) | 25 (100%) |
| Age >55 | 15 | 6 (0.5-31.9) | 121.1 (18-579.2) | P<0.001 | 13 (86.7%) | 13 (100%) |
| BMI ≥30 | 7 | 7.2 (0.6-19.8) | 116.4 (33.5-366) | P=0.018 | 6 (85.7%) | 7 (100%) |
| Abbreviation: BMI: Body Mass Index. | ||||||
Influence of age
Among patients there was a significant negative correlation between anti-S levels and age after both the first dose (R= -0.176, p=0.01) and the second dose (R= -0.193, p=0.005). Among healthcare workers there was also a significant negative correlation between anti-S levels and age after the first dose (R= -0.357, p=0.015) but not after the second dose (R= -0284, p=0.056). Healthcare workers always reached higher levels of anti-S in comparison with patients.
Other correlations
Among patients there was also a significant negative correlation between anti-S and Charlson Comorbidity Index adjusted for age after both the first dose (R= -0.150, p= 0.028) and the second dose (R= -0.163, p=0.018). Finally, a negative correlation between anti-S and Body Mass Index was found after the first dose (R= -0.140, p= 0.04) (Table 3).
No correlations were found with dialysis vintage, Kt/V, diabetes, treatment with ACEi or ARB, or smoking.
| Logistic regression of variables associated with being reactive after the 1st vaccine dose | ||
| OR (Odds ratio) | P value | |
| Age (per 1 year) | 0.970 | 0.008 |
| Charlson Comorbidity Index age-adjusted | 0.894 | 0.015 |
| Body Mass Index | 0.938 | 0.044 |
Discussion
Hemodialysis patients and other immunocompromised individuals were excluded from all phase 3 SARS-CoV-2 vaccine trials and, when our study began, there was no published study of the effects of vaccination in ICHD patients. We therefore decided to use the same criteria for safety and efficacy in our study as used by the vaccine manufacturer in its phase 3 study [9] in an attempt to identify possible differences between hemodialysis patients and the general population [10,11]. These differences may be aggravated by immunosenescence associated with ageing [12,13], as ICHD patients in this setting tend to be of older age. From the experience of vaccination for hepatitis B in hemodialysis patients it is known that higher and more frequent doses are needed than in the general population [14]. Is this also true with SARS-CoV-2 vaccination?
All (100%) of our patients with previous SARS-CoV-2 infection had an anti-S response after the first vaccine dose, but only 35.8% of patients without previous infection responded to the first dose. After both doses of vaccine, 91.7% of all patients had responded compared to 100% of healthcare workers. Despite this lower response, the risk of hospitalization and mortality from COVID-19 infection in ICHD patients and the low rate of adverse reactions, suggest that all ICHD and the healthcare workers caring for them should be vaccinated.
Our older ICHD patients appeared to respond less well to vaccination: in patients without previous SARS-CoV-2 infection, 90.5% in those older than 75 years were reactive after two doses of vaccine to 97.0% in those younger than 55 years. (Table 4).
| Probability of bein g reacti ve to the 1st first and 2nd dose (age = 55 and > 75 years) | ||||
| Reactive after 1st dose | Reactive after 2nd dose | |||
| No | Yes | No | Yes | |
| Age ≤ 55 years (n=33) | 14 (42.4%) | 19 (57.6%) | 1 (3.0%) | 32 (97.0%) |
| Age > 75 years (n=76) | 58 (76.3%) | 18 (23.7%) | 7 (9.5%) | 67 (90.5%) |
We found inverse associations between vaccine response and age, Charlson Comorbidity Index and Body Mass Index. All of these are relevant in ICHD patients, who are predominantly of older age, have several comorbidities and have impaired exercise capacity, which can contribute to obesity.
We did not include cellular immunity or neutralizing antibody (nAb) assays in our study. However, a recent study of the immune responses elicited by SARS-CoV-2 infection showed that anti-S IgG (which we did measure) correlates highly with nAb and its response can be used as a marker of neutralizing activity in individuals [15]. This seems likely also to be the case after vaccination.
The main goals of this COVID-19 vaccination program were to reduce morbidity and mortality by reducing disease severity and hospitalization. However, we know that ICHD patients remain a high-risk population for SARS-CoV-2 subsequent infection with variants [16] and efforts to prevent it should be maintained. Further studies are needed to ascertain how long the protection from vaccination will last in this population group and guide timing of booster doses.
Conflict of Interest Statement
All authors have nothing to disclose.
Acknowledgments
To DaVita Portuguese Research Fund for supporting the laboratory costs of this study and to our Head Nurse Mr. Boaventura Cabecinhas for his helpful co-operation.
Grateful thanks to Prof. Jonathan G. Fox (Glasgow, UK) for assistance with language editing.
References
2. Dong E, Du H, Gardner L. An interactive web-based dashboard to track COVID-19 in real time. The Lancet Infectious Diseases. 2020 May 1;20(5):533-534.
3. Merchant RM, South EC, Lurie N. Public health messaging in an era of social media. JAMA. 2021 Jan 19;325(3):223-4.
4. Bambra C, Riordan R, Ford J, Matthews F. The COVID-19 pandemic and health inequalities. J Epidemiol Community Health. 2020 Nov 1;74(11):964-8.
5. Fauci AS. The story behind COVID-19 vaccines. Science 2021; 372: 109.
6. Wu F, Zhao S, Yu B, Chen YM, Wang W, Song ZG, et al. A new coronavirus associated with human respiratory disease in China. Nature. 2020 Mar;579(7798):265-9.
7. Francis A, Baigent C, Ikizler TA, Cockwell P, Jha V. The urgent need to vaccinate dialysis patients against severe acute respiratory syndrome coronavirus 2: a call to action. Kidney International. 2021 Apr 1;99(4):791-3.
8. Grupper A, Sharon N, Finn T, Cohen R, Israel M, Agbaria A, et al. Humoral response to the Pfizer BNT162b2 vaccine in patients undergoing maintenance hemodialysis. Clinical Journal of the American Society of Nephrology. 2021 Apr 6.
9. Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. New England Journal of Medicine. 2020;383: 2603–2615.
10. Espi M, Koppe L, Fouque D, Thaunat O. Chronic kidney disease-associated immune dysfunctions: impact of protein-bound uremic retention solutes on immune cells. Toxins. 2020 May;12(5):300.
11. Lamarche C, Iliuta IA, Kitzler T. Infectious disease risk in dialysis patients: a transdisciplinary approach. Canadian Journal of Kidney Health and Disease. 2019 Apr;6:2054358119839080.
12. Sato Y, Yanagita M. Immunology of the ageing kidney. Nature Reviews Nephrology. 2019 Oct;15(10):625-40.
13. Nikolich-Žugich J. The twilight of immunity: emerging concepts in aging of the immune system. Nature Immunology. 2018 Jan;19(1):10-9.
14. Center for Disease Control and Prevention: Guidelines for Vaccinating Dialysis Patients and Patients with Chronic Kidney Disease Patients. Available at: https://www.cdc. gov/dialysis/pdfs/vaccinating_dialysis_patients_and_ patients_dec2012.pdf Accessed July 9, 2021.
15. Legros V, Denolly S, Vogrig M, Boson B, Siret E, Rigaill J, et al. A longitudinal study of SARS-CoV-2-infected patients reveals a high correlation between neutralizing antibodies and COVID-19 severity. Cellular & Molecular Immunology. 2021 Feb;18(2):318-27.
16. Hacisuleyman E, Hale C, Saito Y, Blachere NE, Bergh M, Conlon EG, et al. Vaccine breakthrough infections with SARS-CoV-2 variants. New England Journal of Medicine. 2021 Jun 10;384(23):2212-8.
