Loading
International Journal of Anesthesia and Critical Care
ISSN: 2834-2887
Latest Articles
Bridging the Gap: Nurse Anesthesia as a Pathway to Global Health Equity
Terrica Durbin
Access to safe anesthesia care remains one of the most profound inequities in global health. Bridging the gap between evidence and policy, this article argues that integrating nurse anesthesia within advanced practice nursing frameworks offers a viable and ethical pathway toward global health equity.
Int J Anaesth Crit Care, 2026, Volume 5, Issue 1, p5-8 | DOI: 10.33696/Anesthesia.5.012
Anesthesia in a Patient with Ehlers-Danlos Syndrome and Mast Cell Activation Syndrome—Case Report
Carlos Henrique Viana de Castro , Fábio Fantazzini Vieira , Felipe Cruz Jorge , Marcelo Figueiredo , Marco Túlio Froes
The concurrent presentation of Hypermobile Ehlers-Danlos Syndrome (hEDS) and Mast Cell Activation Syndrome (MCAS) poses significant anesthetic challenges, balancing the risks of tissue fragility and autonomic dysfunction with potential anaphylaxis. We report the anesthetic management of a female patient with confirmed hEDS and MCAS undergoing mastopexy.
Int J Anaesth Crit Care, 2026, Volume 5, Issue 1, p1-4 | DOI: 10.33696/Anesthesia.5.011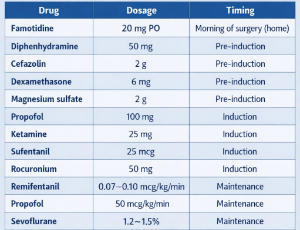
About Scientific Archives
Scientific Archives is a global publisher initiated with the mission of ensuring equal opportunity for accessing science to research community all over the world. Spreading research findings with great relevance to all channels without any barrier is our goal. We want to overcome the challenges of Open Access with ensured quality and transparency.
